IL-17 and TNF-α sustain neutrophil recruitment during inflammation through synergistic effects on endothelial activation
- PMID: 22566565
- PMCID: PMC3370121
- DOI: 10.4049/jimmunol.1200385
IL-17 and TNF-α sustain neutrophil recruitment during inflammation through synergistic effects on endothelial activation
Abstract
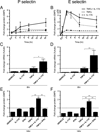
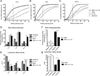
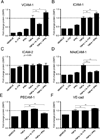
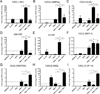
IL-17A (IL-17) is the signature cytokine produced by Th17 cells and has been implicated in host defense against infection and the pathophysiology of autoimmunity and cardiovascular disease. Little is known, however, about the influence of IL-17 on endothelial activation and leukocyte influx to sites of inflammation. We hypothesized that IL-17 would induce a distinct pattern of endothelial activation and leukocyte recruitment when compared with the Th1 cytokine IFN-γ. We found that IL-17 alone had minimal activating effects on cultured endothelium, whereas the combination of TNF-α and IL-17 produced a synergistic increase in the expression of both P-selectin and E-selectin. Using intravital microscopy of the mouse cremaster muscle, we found that TNF-α and IL-17 also led to a synergistic increase in E-selectin-dependent leukocyte rolling on microvascular endothelium in vivo. In addition, TNF-α and IL-17 enhanced endothelial expression of the neutrophilic chemokines CXCL1, CXCL2, and CXCL5 and led to a functional increase in leukocyte transmigration in vivo and CXCR2-dependent neutrophil but not T cell transmigration in a parallel-plate flow chamber system. By contrast, endothelial activation with TNF-α and IFN-γ preferentially induced the expression of the integrin ligands ICAM-1 and VCAM-1, as well as the T cell chemokines CXCL9, CXCL10, and CCL5. These effects were further associated with a functional increase in T cell but not neutrophil transmigration under laminar shear flow. Overall, these data show that IL-17 and TNF-α act in a synergistic manner to induce a distinct pattern of endothelial activation that sustains and enhances neutrophil influx to sites of inflammation.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures



References
-
- Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, Weaver CT. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat. Immunol. 2005;6:1123–1132. - PubMed
-
- Cua JD, Tato CM. Innate IL-17-producing cells: the sentinels of the immune system. Nat. Rev. Immunol. 2010;10:479–489. - PubMed
-
- Lockhart E, Green AM, Flynn JL. IL-17 production is dominated by gammadelta T cells rather than CD4 T cells during Mycobacterium tuberculosis infection. J. Immunol. 2006;177:4662–4669. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

