Lrp5 is not required for the proliferative response of osteoblasts to strain but regulates proliferation and apoptosis in a cell autonomous manner
- PMID: 22567110
- PMCID: PMC3342322
- DOI: 10.1371/journal.pone.0035726
Lrp5 is not required for the proliferative response of osteoblasts to strain but regulates proliferation and apoptosis in a cell autonomous manner
Abstract
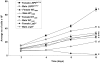
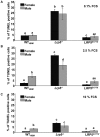
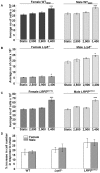
Although Lrp5 is known to be an important contributor to the mechanisms regulating bone mass, its precise role remains unclear. The aim of this study was to establish whether mutations in Lrp5 are associated with differences in the growth and/or apoptosis of osteoblast-like cells and their proliferative response to mechanical strain in vitro. Primary osteoblast-like cells were derived from cortical bone of adult mice lacking functional Lrp5 (Lrp5(-/-)), those heterozygous for the human G171V High Bone Mass (HBM) mutation (LRP5(G171V)) and their WT littermates (WT(Lrp5), WT(HBM)). Osteoblast proliferation over time was significantly higher in cultures of cells from LRP5(G171V) mice compared to their WT(HBM) littermates, and lower in Lrp5(-/-) cells. Cells from female LRP5(G171V) mice grew more rapidly than those from males, whereas cells from female Lrp5(-/-) mice grew more slowly than those from males. Apoptosis induced by serum withdrawal was significantly higher in cultures from Lrp5(-/-) mice than in those from WT(HBM) or LRP5(G171V) mice. Exposure to a single short period of dynamic mechanical strain was associated with a significant increase in cell number but this response was unaffected by genotype which also did not change the 'threshold' at which cells responded to strain. In conclusion, the data presented here suggest that Lrp5 loss and gain of function mutations result in cell-autonomous alterations in osteoblast proliferation and apoptosis but do not alter the proliferative response of osteoblasts to mechanical strain in vitro.
Conflict of interest statement
Figures
Similar articles
-
Analysis of multiple bone responses to graded strains above functional levels, and to disuse, in mice in vivo show that the human Lrp5 G171V High Bone Mass mutation increases the osteogenic response to loading but that lack of Lrp5 activity reduces it.Bone. 2011 Aug;49(2):184-93. doi: 10.1016/j.bone.2011.03.683. Epub 2011 Mar 16. Bone. 2011. PMID: 21419885 Free PMC article.
-
High bone mass in mice expressing a mutant LRP5 gene.J Bone Miner Res. 2003 Jun;18(6):960-74. doi: 10.1359/jbmr.2003.18.6.960. J Bone Miner Res. 2003. PMID: 12817748
-
High Bone Mass-Causing Mutant LRP5 Receptors Are Resistant to Endogenous Inhibitors In Vivo.J Bone Miner Res. 2015 Oct;30(10):1822-30. doi: 10.1002/jbmr.2514. J Bone Miner Res. 2015. PMID: 25808845 Free PMC article.
-
Pathogenic mutations and polymorphisms in the lipoprotein receptor-related protein 5 reveal a new biological pathway for the control of bone mass.Curr Opin Lipidol. 2005 Apr;16(2):207-14. doi: 10.1097/01.mol.0000162326.62419.e4. Curr Opin Lipidol. 2005. PMID: 15767861 Review.
-
LRP5 high bone mass (Worth-type autosomal dominant endosteal hyperostosis): case report and historical review of the literature.Arch Osteoporos. 2023 Sep 2;18(1):112. doi: 10.1007/s11657-023-01319-6. Arch Osteoporos. 2023. PMID: 37659026 Free PMC article. Review.
Cited by
-
Protein kinase Cα (PKCα) regulates bone architecture and osteoblast activity.J Biol Chem. 2014 Sep 12;289(37):25509-22. doi: 10.1074/jbc.M114.580365. Epub 2014 Jul 28. J Biol Chem. 2014. PMID: 25070889 Free PMC article.
-
Aging and Mechanoadaptive Responsiveness of Bone.Curr Osteoporos Rep. 2019 Dec;17(6):560-569. doi: 10.1007/s11914-019-00553-7. Curr Osteoporos Rep. 2019. PMID: 31760583 Free PMC article. Review.
-
The LRP5 high-bone-mass mutation causes alveolar bone accrual with minor craniofacial alteration.J Periodontal Res. 2023 Aug;58(4):723-732. doi: 10.1111/jre.13130. Epub 2023 May 2. J Periodontal Res. 2023. PMID: 37128744 Free PMC article.
-
Remote activation of the Wnt/β-catenin signalling pathway using functionalised magnetic particles.PLoS One. 2015 Mar 17;10(3):e0121761. doi: 10.1371/journal.pone.0121761. eCollection 2015. PLoS One. 2015. PMID: 25781466 Free PMC article.
-
Preclinical models for in vitro mechanical loading of bone-derived cells.Bonekey Rep. 2015 Aug 19;4:728. doi: 10.1038/bonekey.2015.97. eCollection 2015. Bonekey Rep. 2015. PMID: 26331007 Free PMC article.
References
-
- Ehrlich PJ, Lanyon LE. Mechanical strain and bone cell function: a review. Osteoporos Int. 2002;13:688–700. - PubMed
-
- Day TF, Guo X, Garrett-Beal L, Yang Y. Wnt/beta-catenin signaling in mesenchymal progenitors controls osteoblast and chondrocyte differentiation during vertebrate skeletogenesis. Dev Cell. 2005;8:739–750. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

