Young HIV-infected children and their adult caregivers prefer tablets to syrup antiretroviral medications in Africa
- PMID: 22567139
- PMCID: PMC3342167
- DOI: 10.1371/journal.pone.0036186
Young HIV-infected children and their adult caregivers prefer tablets to syrup antiretroviral medications in Africa
Abstract
Background: Provision of anti-retroviral therapy (ART) for HIV-infected children is complicated using syrup formulations, which are costlier than tablets, harder to transport and store and difficult for health-workers to prescribe and caregivers to administer. Dispersible/crushable tablets may be more appropriate. We studied the acceptability of syrups and scored tablets among young children who used both in the AntiRetroviral Research fOr Watoto (ARROW) trial.
Methods: ARROW is an ongoing randomized trial of paediatric ART monitoring and treatment strategies in 1206 children in Uganda and Zimbabwe. 405 children initially received syrups of combination ART including Nevirapine, Zidovudine, Abacavir and Lamivudine before changing, when reaching the 12-<15 kg weightband, to scored adult-dose tablets prescribed according to WHO weightband tables. Caregiver expectations and experiences were collected in questionnaires at their last visit on syrups and after 8 and 24 weeks on tablets.
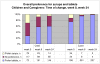
Results: Questionnaires were completed by caregivers of 267 children (median age 2.9 years (IQR 2.5, 3.4)). At last visit on syrups, 79% caregivers reported problems with syrups, mostly related to number, weight, transportation and conspicuousness of bottles. Difficulties taking tablets were expected by 127(48%) caregivers; however, after 8 and 24 weeks, only 26% and 18% reported their children had problems with tablets and no problems were reported with transportation/conspicuousness. Taste, swallowing or vomiting were reported as problems 'sometimes/often' for 14%, 9%, 22% children on syrups and 16%, 9%, 8% on tablets. At last visit on syrups, 74% caregivers expected to prefer tablets but only 27% thought their child would. After 8/24 weeks, 94%/97% caregivers preferred tablets and 57%/59% reported their child did.
Conclusions: Most children at about 3 years can take tablets; caregivers and children themselves generally prefer tablets to liquid formulations of HIV medications above this age. Preferences of caregivers and children should be considered when designing and licensing paediatric drug formulations.
Conflict of interest statement
Figures
References
-
- Bangsberg DR, Perry S, Charlebois ED, Clark RA, Roberston M, et al. Non-adherence to highly active antiretroviral therapy predicts progression to AIDS. AIDS. 2001;15:181–1183. - PubMed
-
- Gross R, Yip B, Lo Re V, Wood E, Alexander CS, et al. A simple, dynamic measure of antiretroviral therapy adherence predicts failure to maintain HIV-1 suppression. J Infect Dis. 2006;194:1108–1114. - PubMed
-
- Nachega JB, Hislop M, Dowdy DW, Lo M, Omer SB, et al. Adherence to highly active antiretroviral therapy assessed by pharmacy claims predicts survival in HIV-infected South African adults. J Acquir Immune Defic Syndr. 2006;43:78–84. - PubMed
-
- Sohn A, Ananworanich J. How can we simplify antiretroviral therapy in children? Curr Opin HIV AIDS. 2007;2:426–430. - PubMed
-
- Havens PL, Gibb DM. Increasing antiretroviral drug access for children with HIV infection. Pediatrics. 2007;119:838–845. (DOI:10.1542/peds.2007-0273) - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical