Interkingdom signaling: integration, conformation, and orientation of N-acyl-L-homoserine lactones in supported lipid bilayers
- PMID: 22568488
- PMCID: PMC3388113
- DOI: 10.1021/la301241s
Interkingdom signaling: integration, conformation, and orientation of N-acyl-L-homoserine lactones in supported lipid bilayers
Abstract
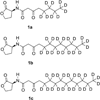
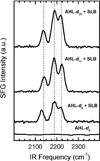
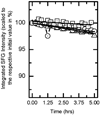
N-Acyl-L-homoserine lactones (AHLs) are small cell-to-cell signaling molecules involved in the regulation of population density and local gene expression in microbial communities. Recent evidence shows that contact of this signaling system, usually referred to as quorum sensing, to living eukaryotes results in interactions of AHL with host cells in a process termed "interkingdom signaling". So far details of this process and the binding site of the AHLs remain unknown; both an intracellular and a membrane-bound receptor seem possible, the first of which requires passage through the cell membrane. Here, we used sum-frequency-generation (SFG) spectroscopy to investigate the integration, conformation, orientation, and translocation of deuterated N-acyl-L-homoserine lactones (AHL-d(n)) with varying chain length (8, 12, and 14 C atoms) in lipid bilayers consisting of a 1:1 mixture of POPC:POPG supported on SiO(2) substrates (prepared by vesicle fusion). We found that all AHL-d(n) derivatives are well-ordered within the supported lipid bilayer (SLB) in a preferentially all-trans conformation of the deuterated alkyl chain and integrated into the upper leaflet of the SLB with the methyl terminal groups pointing downward. For the bilayer system described above, no flip-flop of AHL-d(n) from the upper leaflet to the lower one could be observed. Spectral assignments and interpretations were further supported by Fourier transform infrared and Raman spectroscopy.
Figures


References
-
- Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: A common cause of persistent infections. Science. 1999;284(5418):1318–1322. - PubMed
-
- Krumbein WE, Paterson DM, Zavarzin GA. Fossil and recent biofilms : a natural history of life on Earth. Dordrecht; Boston: Kluwer Academic Publishers; 2003. p. xxi.p. 482.
-
- Costerton JW, Cheng KJ, Geesey GG, Ladd TI, Nickel JC, Dasgupta M, Marrie TJ. Bacterial biofilms in nature and disease. Annu. Rev. Microbiol. 1987;41:435–464. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

