Oncogene-induced telomere dysfunction enforces cellular senescence in human cancer precursor lesions
- PMID: 22569128
- PMCID: PMC3395091
- DOI: 10.1038/emboj.2012.132
Oncogene-induced telomere dysfunction enforces cellular senescence in human cancer precursor lesions
Abstract
In normal human somatic cells, telomere dysfunction causes cellular senescence, a stable proliferative arrest with tumour suppressing properties. Whether telomere dysfunction-induced senescence (TDIS) suppresses cancer growth in humans, however, is unknown. Here, we demonstrate that multiple and distinct human cancer precursor lesions, but not corresponding malignant cancers, are comprised of cells that display hallmarks of TDIS. Furthermore, we demonstrate that oncogenic signalling, frequently associated with initiating cancer growth in humans, dramatically affected telomere structure and function by causing telomeric replication stress, rapid and stochastic telomere attrition, and consequently telomere dysfunction in cells that lack hTERT activity. DNA replication stress induced by drugs also resulted in telomere dysfunction and cellular senescence in normal human cells, demonstrating that telomeric repeats indeed are hypersensitive to DNA replication stress. Our data reveal that TDIS, accelerated by oncogene-induced DNA replication stress, is a biological response of cells in human cancer precursor lesions and provide strong evidence that TDIS is a critical tumour suppressing mechanism in humans.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures


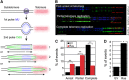
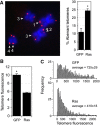


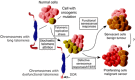
Comment in
-
Telomere dysfunction puts the brakes on oncogene-induced cancers.EMBO J. 2012 Jun 29;31(13):2833-4. doi: 10.1038/emboj.2012.162. Epub 2012 May 29. EMBO J. 2012. PMID: 22643218 Free PMC article.
References
-
- Arlt MF, Durkin SG, Ragland RL, Glover TW (2006) Common fragile sites as targets for chromosome rearrangements. DNA Repair (Amst) 5: 1126–1135 - PubMed
-
- Augereau A, T'Kint de Roodenbeke C, Simonet T, Bauwens S, Horard B, Callanan M, Leroux D, Jallades L, Salles G, Gilson E, Poncet D (2011) Telomeric damage in early stage of chronic lymphocytic leukemia correlates with shelterin dysregulation. Blood 118: 1316–1322 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

