Fluorescent nanoprobes dedicated to in vivo imaging: from preclinical validations to clinical translation
- PMID: 22576228
- PMCID: PMC6268987
- DOI: 10.3390/molecules17055564
Fluorescent nanoprobes dedicated to in vivo imaging: from preclinical validations to clinical translation
Abstract
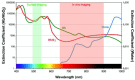
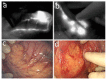
With the fast development, in the last ten years, of a large choice of set-ups dedicated to routine in vivo measurements in rodents, fluorescence imaging techniques are becoming essential tools in preclinical studies. Human clinical uses for diagnostic and image-guided surgery are also emerging. In comparison to low-molecular weight organic dyes, the use of fluorescent nanoprobes can improve both the signal sensitivity (better in vivo optical properties) and the fluorescence biodistribution (passive "nano" uptake in tumours for instance). A wide range of fluorescent nanoprobes have been designed and tested in preclinical studies for the last few years. They will be reviewed and discussed considering the obstacles that need to be overcome for their potential everyday use in clinics. The conjugation of fluorescence imaging with the benefits of nanotechnology should open the way to new medical applications in the near future.
Figures
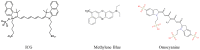



References
-
- Hassan M., Klaunberg B.A. Biomedical applications of fluorescence imaging in vivo. Comp. Med. 2004;54:635–644. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

