Identification of the minimal region in lipase ABC transporter recognition domain of Pseudomonas fluorescens for secretion and fluorescence of green fluorescent protein
- PMID: 22578275
- PMCID: PMC3430570
- DOI: 10.1186/1475-2859-11-60
Identification of the minimal region in lipase ABC transporter recognition domain of Pseudomonas fluorescens for secretion and fluorescence of green fluorescent protein
Abstract
Background: TliA is a thermostable lipase secreted by the type 1 secretion system (T1SS) of Pseudomonas fluorescens. The secretion is promoted by its secretion/chaperone domain located near the C-terminus, which is composed mainly of four Repeat-in-Toxin (RTX) repeats. In order to identify the minimal region of TliA responsible for its secretion, five different copies of the secretion/chaperone domain, each involving truncated N-terminal residues and a common C-terminus, were acquired and named as lipase ABC transporter recognition domains (LARDs). Each LARD was fused to epidermal growth factor (EGF) or green fluorescent protein (GFP), and the secretion of EGF-LARD or GFP-LARD fusion proteins was assessed in Escherichia coli with ABC transporter.
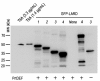
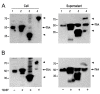
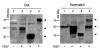
Results: Among the fusion proteins, GFP or EGF with 105-residue LARD3 was most efficiently secreted. In addition, GFP-LARD3 emitted wild type GFP fluorescence. Structurally, LARD3 had the 4 RTX repeats exposed at the N-terminus, while other LARDs had additional residues prior to them or missed some of the RTX repeats. LARD3 was both necessary and sufficient for efficient secretion and maintenance of GFP fluorescence in E. coli, which was also confirmed in P. fluorescens and P. fluorescens ▵tliA, a knock-out mutant of tliA.
Conclusion: LARD3 was a potent secretion signal in T1SS for its fusion flanking RTX motif, which enhanced secretion and preserved the fluorescence of GFP. LARD3-mediated secretion in E. coli or P. fluorescens will enable the development of enhanced protein manufacturing factory and recombinant microbe secreting protein of interest in situ.
Figures




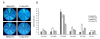

Similar articles
-
Export of recombinant proteins in Escherichia coli using ABC transporter with an attached lipase ABC transporter recognition domain (LARD).Microb Cell Fact. 2009 Jan 29;8:11. doi: 10.1186/1475-2859-8-11. Microb Cell Fact. 2009. PMID: 19178697 Free PMC article.
-
Alleviation of temperature-sensitive secretion defect of Pseudomonas fluorescens ATP-binding cassette (ABC) transporter, TliDEF, by a change of single amino acid in the ABC protein, TliD.J Biosci Bioeng. 2016 Sep;122(3):283-6. doi: 10.1016/j.jbiosc.2016.02.013. Epub 2016 Mar 29. J Biosci Bioeng. 2016. PMID: 27033673
-
A lower isoelectric point increases signal sequence-mediated secretion of recombinant proteins through a bacterial ABC transporter.J Biol Chem. 2017 Dec 1;292(48):19782-19791. doi: 10.1074/jbc.M117.786749. Epub 2017 Sep 29. J Biol Chem. 2017. PMID: 28972172 Free PMC article.
-
Redefining the bacterial Type I protein secretion system.Adv Microb Physiol. 2023;82:155-204. doi: 10.1016/bs.ampbs.2022.10.003. Epub 2022 Dec 5. Adv Microb Physiol. 2023. PMID: 36948654 Review.
-
Type I secretion systems - a story of appendices.Res Microbiol. 2013 Jul-Aug;164(6):596-604. doi: 10.1016/j.resmic.2013.03.011. Epub 2013 Mar 26. Res Microbiol. 2013. PMID: 23541474 Review.
Cited by
-
PySupercharge: a python algorithm for enabling ABC transporter bacterial secretion of all proteins through amino acid mutation.Microb Cell Fact. 2024 Apr 20;23(1):115. doi: 10.1186/s12934-024-02342-z. Microb Cell Fact. 2024. PMID: 38643109 Free PMC article.
-
Type I Protein Secretion-Deceptively Simple yet with a Wide Range of Mechanistic Variability across the Family.EcoSal Plus. 2016 Dec;7(1):10.1128/ecosalplus.ESP-0019-2015. doi: 10.1128/ecosalplus.ESP-0019-2015. EcoSal Plus. 2016. PMID: 28084193 Free PMC article. Review.
-
Generalized Approach towards Secretion-Based Protein Production via Neutralization of Secretion-Preventing Cationic Substrate Residues.Int J Mol Sci. 2022 Jun 15;23(12):6700. doi: 10.3390/ijms23126700. Int J Mol Sci. 2022. PMID: 35743142 Free PMC article.
-
Secretion of recombinant proteins from E. coli.Eng Life Sci. 2018 Apr 14;18(8):532-550. doi: 10.1002/elsc.201700200. eCollection 2018 Aug. Eng Life Sci. 2018. PMID: 32624934 Free PMC article. Review.
-
Lipase and protease double-deletion mutant of Pseudomonas fluorescens suitable for extracellular protein production.Appl Environ Microbiol. 2012 Dec;78(23):8454-62. doi: 10.1128/AEM.02476-12. Epub 2012 Oct 5. Appl Environ Microbiol. 2012. PMID: 23042178 Free PMC article.
References
-
- Delepelaire P. Type I secretion in gram-negative bacteria. Biochim Biophys Acta. 2004;1694(1–3):149–161. - PubMed
-
- Delepelaire P. PrtD, the integral membrane ATP-binding cassette component of the Erwinia chrysanthemi metalloprotease secretion system, exhibits a secretion signal-regulated ATPase activity. J Biol Chem. 1994;269(45):27952–27957. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

