Skin and soft tissue concentrations of tedizolid (formerly torezolid), a novel oxazolidinone, following a single oral dose in healthy volunteers
- PMID: 22584101
- PMCID: PMC3789129
- DOI: 10.1016/j.ijantimicag.2012.03.006
Skin and soft tissue concentrations of tedizolid (formerly torezolid), a novel oxazolidinone, following a single oral dose in healthy volunteers
Abstract
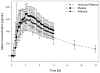
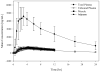
Plasma concentrations of antimicrobial drugs have long been used to correlate exposure with effect, yet one cannot always assume that unbound plasma and tissue concentrations are similar. Knowledge about unbound tissue concentrations is important in the development of antimicrobial drugs, since most infections are localised in tissues. Therefore, a clinical microdialysis study was conducted to evaluate the distribution of tedizolid (TR-700), the active moiety of the antimicrobial prodrug tedizolid phosphate (TR-701), into interstitial fluid (ISF) of subcutaneous adipose and skeletal muscle tissues following a single oral 600 mg dose of tedizolid phosphate in fasting conditions. Twelve healthy adult subjects were enrolled. Two microdialysis probes were implanted into the thigh of each subject, one into the vastus medialis muscle and one into subcutaneous adipose tissue. Probes were calibrated using retrodialysis. Dialysate samples were collected every 20 min for 12h following a single oral dose of 600 mg tedizolid phosphate, and blood samples were drawn over 24h. Unbound tedizolid levels in plasma were similar to those in muscle and adipose tissue. The ratios of unbound (free) AUC in tissues over unbound AUC in plasma (fAUC(tissue)/fAUC(plasma)) were 1.1 ± 0.2 and 1.2 ± 0.2 for adipose and muscle tissue, respectively. The median half-life was 8.1, 9.2 and 9.6h for plasma, adipose tissue and muscle tissue, respectively. Mean protein binding was 87.2 ± 1.8%. The study drug was very well tolerated. The results of this study show that tedizolid distributes well into ISF of adipose and muscle tissues. Unbound levels of tedizolid in plasma, adipose tissue and muscle tissue were well correlated. Free plasma levels are indicative of unbound levels in the ISF of muscle and adipose tissues.
Copyright © 2012 Elsevier B.V. and the International Society of Chemotherapy. All rights reserved.
Conflict of interest statement
CdA and PP are employees of Trius Therapeutics, Inc. (San Diego, CA). All other authors declare no competing interests.
Figures
Similar articles
-
Pulmonary disposition of tedizolid following administration of once-daily oral 200-milligram tedizolid phosphate in healthy adult volunteers.Antimicrob Agents Chemother. 2012 May;56(5):2627-34. doi: 10.1128/AAC.05354-11. Epub 2012 Feb 13. Antimicrob Agents Chemother. 2012. PMID: 22330925 Free PMC article.
-
Comparative Assessment of Tedizolid Pharmacokinetics and Tissue Penetration between Diabetic Patients with Wound Infections and Healthy Volunteers via In Vivo Microdialysis.Antimicrob Agents Chemother. 2017 Dec 21;62(1):e01880-17. doi: 10.1128/AAC.01880-17. Print 2018 Jan. Antimicrob Agents Chemother. 2017. PMID: 29109162 Free PMC article.
-
Penetration of doripenem into skeletal muscle and subcutaneous adipose tissue in healthy volunteers.Antimicrob Agents Chemother. 2012 Jan;56(1):532-5. doi: 10.1128/AAC.05506-11. Epub 2011 Nov 14. Antimicrob Agents Chemother. 2012. PMID: 22083466 Free PMC article.
-
Tedizolid (TR-701): a new oxazolidinone with enhanced potency.Expert Opin Investig Drugs. 2012 Apr;21(4):515-22. doi: 10.1517/13543784.2012.660250. Epub 2012 Feb 13. Expert Opin Investig Drugs. 2012. PMID: 22324973 Review.
-
Tedizolid: a novel oxazolidinone with potent activity against multidrug-resistant gram-positive pathogens.Drugs. 2015 Feb;75(3):253-70. doi: 10.1007/s40265-015-0352-7. Drugs. 2015. PMID: 25673021 Review.
Cited by
-
Tedizolid is a promising antimicrobial option for the treatment of Staphylococcus aureus infections in cystic fibrosis patients.J Antimicrob Chemother. 2020 Jan 1;75(1):126-134. doi: 10.1093/jac/dkz418. J Antimicrob Chemother. 2020. PMID: 31617901 Free PMC article.
-
Economic burden of skin and skin structure infections due to Gram-positive bacteria in patients on hospital at home-based outpatient parenteral antibiotic therapy (OPAT).Rev Esp Quimioter. 2023 Jun;36(3):291-301. doi: 10.37201/req/134.2022. Epub 2023 Apr 5. Rev Esp Quimioter. 2023. PMID: 37017118 Free PMC article.
-
Dynamic distribution of systemically administered antibiotics in orthopeadically relevant target tissues and settings.APMIS. 2024 Dec;132(12):992-1025. doi: 10.1111/apm.13490. Epub 2024 Nov 12. APMIS. 2024. PMID: 39530161 Free PMC article.
-
Pharmacokinetics and Pharmacodynamics of Tedizolid.Clin Pharmacokinet. 2022 Apr;61(4):489-503. doi: 10.1007/s40262-021-01099-7. Epub 2022 Feb 7. Clin Pharmacokinet. 2022. PMID: 35128625 Free PMC article. Review.
-
Pharmacokinetics of tedizolid following oral administration: single and multiple dose, effect of food, and comparison of two solid forms of the prodrug.Pharmacotherapy. 2014 Mar;34(3):240-50. doi: 10.1002/phar.1337. Epub 2013 Aug 7. Pharmacotherapy. 2014. PMID: 23926058 Free PMC article. Clinical Trial.
References
-
- National Nosocomial Infections Surveillance System. National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2004, issued October 2004. Am J Infect Control. 2004;32:470–85. - PubMed
-
- Kallen AJ, Mu Y, Bulens S, Reingold A, Petit S, Gershman K, et al. Active Bacterial Core surveillance (ABCs) MRSA Investigators of the Emerging Infections Program. Health care-associated invasive MRSA Infections, 2005–2008. JAMA. 2010;304:641–7. - PubMed
-
- MacGowan AP. Pharmacokinetic and pharmacodynamic profile of linezolid in healthy volunteers and patients with Gram-positive infections. J Antimicrob Chemother. 2003;51(Suppl 2):ii17–25. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical