Comparative computational modeling of airflows and vapor dosimetry in the respiratory tracts of rat, monkey, and human
- PMID: 22584687
- PMCID: PMC3524950
- DOI: 10.1093/toxsci/kfs168
Comparative computational modeling of airflows and vapor dosimetry in the respiratory tracts of rat, monkey, and human
Abstract
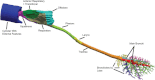
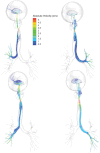
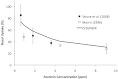
Computational fluid dynamics (CFD) models are useful for predicting site-specific dosimetry of airborne materials in the respiratory tract and elucidating the importance of species differences in anatomy, physiology, and breathing patterns. We improved the imaging and model development methods to the point where CFD models for the rat, monkey, and human now encompass airways from the nose or mouth to the lung. A total of 1272, 2172, and 135 pulmonary airways representing 17±7, 19±9, or 9±2 airway generations were included in the rat, monkey and human models, respectively. A CFD/physiologically based pharmacokinetic model previously developed for acrolein was adapted for these anatomically correct extended airway models. Model parameters were obtained from the literature or measured directly. Airflow and acrolein uptake patterns were determined under steady-state inhalation conditions to provide direct comparisons with prior data and nasal-only simulations. Results confirmed that regional uptake was sensitive to airway geometry, airflow rates, acrolein concentrations, air:tissue partition coefficients, tissue thickness, and the maximum rate of metabolism. Nasal extraction efficiencies were predicted to be greatest in the rat, followed by the monkey, and then the human. For both nasal and oral breathing modes in humans, higher uptake rates were predicted for lower tracheobronchial tissues than either the rat or monkey. These extended airway models provide a unique foundation for comparing material transport and site-specific tissue uptake across a significantly greater range of conducting airways in the rat, monkey, and human than prior CFD models.
Figures







References
-
- Andersen M., Sarangapani R., Gentry R., Clewell H., Covington T., Frederick C. B. (2000). Application of a hybrid CFD-PBPK nasal dosimetry model in an inhalation risk assessment: An example with acrylic acid. Toxicol. Sci. 57 312–325 - PubMed
-
- Anderson J. C., Babb A. L.,, Hlastala M. P. (2003). Modeling soluble gas exchange in the airways and alveoli. Ann. Biomed. Eng. 31 1402–1422 - PubMed
-
- Anjilvel S., Asgharian B. (1995). A multiple-path model of particle deposition in the rat lung. Fundam. Appl. Toxicol. 28 41–50 - PubMed
-
- Asgharian B., Hofman W.,, Bergmann R. (2001). Particle deposition in a multiple-path model of the human lung. Aerosol Sci. Technol. 34 332–339
-
- Asgharian B., Price O. T., Schroeter J. D., Kimbell J. S.,, Singal M. (2012). A lung dosimetry model of vapor uptake and tissue disposition. Inhal. Toxicol. 24 182–193 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

