Optimal dosing of miltefosine in children and adults with visceral leishmaniasis
- PMID: 22585212
- PMCID: PMC3393397
- DOI: 10.1128/AAC.00292-12
Optimal dosing of miltefosine in children and adults with visceral leishmaniasis
Abstract
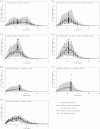
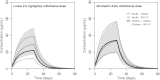
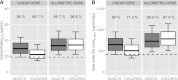
Only anecdotal data are available on the pharmacokinetics (PK) of miltefosine in children suffering from visceral leishmaniasis (VL). While failure rates were higher in children with VL, steady-state concentrations appeared lower than those seen with adults. We hypothesized that the current linear dosage (in milligrams per kilogram of body weight) is too low for children and that a new dosing algorithm based on an appropriate body size model would result in an optimal exposure. A population PK analysis was performed on three historic pooled data sets, including Indian children, Indian adults, and European adults. Linear and allometric scaling of PK parameters by either body weight or fat-free mass (FFM) was evaluated for body size models. Based on the developed PK model, a dosing algorithm for miltefosine in children and adults was proposed and evaluated in silico. The population PK model employing allometric scaling fitted best to the pooled miltefosine data. Allometric scaling by FFM reduced between-subject variability, e.g., for drug clearance, from 49.6% to 32.1%. A new allometric miltefosine dosing algorithm was proposed. Exposure to miltefosine was lower in children than adults receiving 2.5 mg/kg/day: a C(max) of 18.8 μg/ml was reached by 90% of adults and 66.7% of children. The allometric daily dose resulted in similar levels of exposure to miltefosine for adults and children. The use of a new allometric dosing algorithm for miltefosine in VL patients results in optimal exposure to miltefosine in both adults and children and might improve clinical outcome in children.
Figures
References
-
- Ambrose PG, et al. 2009. Use of a clinically derived exposure-response relationship to evaluate potential tigecycline-Enterobacteriaceae susceptibility breakpoints. Diagn. Microbiol. Infect. Dis. 63:38–42 - PubMed
-
- Anderson BJ, Holford NHG. 2008. Mechanism-based concepts of size and maturity in pharmacokinetics. Annu. Rev. Pharmacol. Toxicol. 48:303–332 - PubMed
-
- Anderson BJ, McKee AD, Holford NH. 1997. Size, myths and the clinical pharmacokinetics of analgesia in paediatric patients. Clin. Pharmacokinet. 33:313–327 - PubMed
-
- Anderson BJ, Holford NHG. 2009. Mechanistic basis of using body size and maturation to predict clearance in humans. Drug Metab. Pharmacokinet. 24:25–36 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

