Freeze-thaw lysates of Plasmodium falciparum-infected red blood cells induce differentiation of functionally competent regulatory T cells from memory T cells
- PMID: 22585585
- PMCID: PMC3549566
- DOI: 10.1002/eji.201142164
Freeze-thaw lysates of Plasmodium falciparum-infected red blood cells induce differentiation of functionally competent regulatory T cells from memory T cells
Abstract
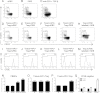
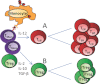
In addition to naturally occurring regulatory T (nTreg) cells derived from the thymus, functionally competent Treg cells can be induced in vitro from peripheral blood lymphocytes in response to TCR stimulation with cytokine costimulation. Using these artificial stimulation conditions, both naïve as well as memory CD4(+) T cells can be converted into induced Treg (iTreg) cells, but the cellular origin of such iTreg cells in vivo or in response to more physiologic stimulation with pathogen-derived antigens is less clear. Here, we demonstrate that a freeze/thaw lysate of Plasmodium falciparum schizont extract (PfSE) can induce functionally competent Treg cells from peripheral lymphocytes in a time- and dose-dependent manner without the addition of exogenous costimulatory factors. The PfSE-mediated induction of Treg cells required the presence of nTreg cells in the starting culture. Further experiments mixing either memory or naïve T cells with antigen presenting cells and CFSE-labeled Treg cells identified CD4(+) CD45RO(+) CD25(-) memory T cells rather than Treg cells as the primary source of PfSE-induced Treg cells. Taken together, these data suggest that in the presence of nTreg cells, PfSE induces memory T cells to convert into iTreg cells that subsequently expand alongside PfSE-induced effector T cells.
© 2012 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures




References
-
- Sakaguchi S. Naturally arising Foxp3-expressing CD25+CD4+ regulatory T cells in immunological tolerance to self and non-self. Nat. Immunol. 2005;6:345–352. - PubMed
-
- Mills KHG. Regulatory T cells: friend or foe in immunity to infection? Nat. Rev. Immunol. 2004;4:841–855. - PubMed
-
- Belkaid Y, Blank RB, Suffia I. Natural regulatory T cells and parasites: a common quest for host homeostasis. Immunol. Rev. 2006;212:287–300. - PubMed
-
- Bluestone JA, Abbas AK. Natural versus adaptive regulatory T cells. Nat. Rev. Immunol. 2003;3:253–257. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

