Lack of T cells in Act1-deficient mice results in elevated IgM-specific autoantibodies but reduced lupus-like disease
- PMID: 22585710
- PMCID: PMC3712797
- DOI: 10.1002/eji.201142238
Lack of T cells in Act1-deficient mice results in elevated IgM-specific autoantibodies but reduced lupus-like disease
Abstract
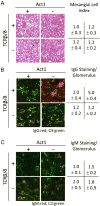
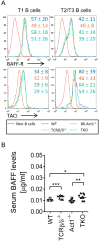
Act1 is a negative regulator of B-cell activation factor of the TNF family (BAFF) and CD40L-induced signaling. BALB/C mice lacking Act1 develop systemic autoimmunity resembling systemic lupus erythematosus (SLE) and Sjögren's syndrome (SjS). SLE and SjS are characterized by anti-nuclear IgG autoantibody (ANA-IgG) production and inflammation of peripheral tissues. As autoantibody production can occur in a T-cell dependent or T-cell independent manner, we investigated the role of T-cell help during Act1-mediated autoimmunity. Act1-deficiency was bred onto C57Bl/6 (B6.Act1(-/-) ) mice and B6.TCRβ(-/-) TCRδ(-/-) Act1(-/-) (TKO) mice were generated. While TCRβ/δ-sufficient B6.Act1(-/-) mice developed splenomegaly and lymphadenopathy, hypergammaglobulinemia, elevated levels of ANA-IgG, and kidney pathology, TKO mice failed to develop any such signs of disease. Neither B6.Act1(-/-) nor TKO mice developed SjS-like disease, suggesting that epigenetic interactions on the BALB/C background are responsible for this phenotype in BALB/C.Act1(-/-) mice. Interestingly, BAFF-driven transitional B-cell abnormalities, previously reported in BALB/C.Act1(-/-) mice, were intact in B6.Act1(-/-) mice and largely independent of T cells. In conclusion, T cells are necessary for the development of SLE-like disease in B6.Act1(-/-) mice, but not BAFF-driven transitional B-cell differentiation.
© 2012 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
Figures



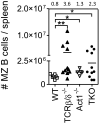
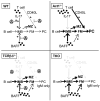
 ) negatively affecting BAFF and CD40L and positively affecting IL-17A induced signaling (reviewed in [9]). TCRβ/δ−/− mice (lower left panel) express increased levels of BAFF by a yet unknown mechanism, resulting in elevated IMB survival and differentiation into marginal zone (MZ) and follicular mature (FM) B cells. Due to the lack of T cells (CD40L), however, no Ig class switching takes place and antibodies present are IgM only. B6.Act1−/− mice (upper right quadrant) have normal levels of BAFF, but increased responsiveness due to the lack of Act1 [1]. This drives IMB survival and differentiation into MZ and FM B cells [2]. Furthermore, as Act1-mediated negative regulation of CD40 signaling is lacking, the formation of IgG-secreting autoreactive plasma cells (PC) is increased. Combined deficiency of T cells and Act1 (lower right quadrant) results in both high levels of BAFF and BAFF hyperresponsiveness [1] driving the accumulation of MZ and FM B cells, but due to the lack of CD40L-expressing T cells, still inhibited Ig class switching. IL-17 is induced during inflammatory conditions and further enhances B-cell survival, proliferation and differentiation in WT mice [37]. Lack of T cells or Act1 will each separately abrogate this pathway.
) negatively affecting BAFF and CD40L and positively affecting IL-17A induced signaling (reviewed in [9]). TCRβ/δ−/− mice (lower left panel) express increased levels of BAFF by a yet unknown mechanism, resulting in elevated IMB survival and differentiation into marginal zone (MZ) and follicular mature (FM) B cells. Due to the lack of T cells (CD40L), however, no Ig class switching takes place and antibodies present are IgM only. B6.Act1−/− mice (upper right quadrant) have normal levels of BAFF, but increased responsiveness due to the lack of Act1 [1]. This drives IMB survival and differentiation into MZ and FM B cells [2]. Furthermore, as Act1-mediated negative regulation of CD40 signaling is lacking, the formation of IgG-secreting autoreactive plasma cells (PC) is increased. Combined deficiency of T cells and Act1 (lower right quadrant) results in both high levels of BAFF and BAFF hyperresponsiveness [1] driving the accumulation of MZ and FM B cells, but due to the lack of CD40L-expressing T cells, still inhibited Ig class switching. IL-17 is induced during inflammatory conditions and further enhances B-cell survival, proliferation and differentiation in WT mice [37]. Lack of T cells or Act1 will each separately abrogate this pathway.Similar articles
-
Act1 is a negative regulator in T and B cells via direct inhibition of STAT3.Nat Commun. 2018 Jul 16;9(1):2745. doi: 10.1038/s41467-018-04974-3. Nat Commun. 2018. PMID: 30013031 Free PMC article.
-
Global T cell dysregulation in non-autoimmune-prone mice promotes rapid development of BAFF-independent, systemic lupus erythematosus-like autoimmunity.J Immunol. 2008 Jul 1;181(1):833-41. doi: 10.4049/jimmunol.181.1.833. J Immunol. 2008. PMID: 18566449 Free PMC article.
-
Constitutive overexpression of BAFF in autoimmune-resistant mice drives only some aspects of systemic lupus erythematosus-like autoimmunity.Arthritis Rheum. 2010 Aug;62(8):2432-42. doi: 10.1002/art.27502. Arthritis Rheum. 2010. PMID: 20506216 Free PMC article.
-
B cells flying solo.Immunol Cell Biol. 2008 Jan;86(1):40-6. doi: 10.1038/sj.icb.7100142. Immunol Cell Biol. 2008. PMID: 18172443 Review.
-
Act1 modulates autoimmunity through its dual functions in CD40L/BAFF and IL-17 signaling.Cytokine. 2008 Feb;41(2):105-13. doi: 10.1016/j.cyto.2007.09.015. Epub 2007 Dec 3. Cytokine. 2008. PMID: 18061473 Review.
Cited by
-
Act1 is a negative regulator in T and B cells via direct inhibition of STAT3.Nat Commun. 2018 Jul 16;9(1):2745. doi: 10.1038/s41467-018-04974-3. Nat Commun. 2018. PMID: 30013031 Free PMC article.
-
Polygenic autoimmune disease risk alleles impacting B cell tolerance act in concert across shared molecular networks in mouse and in humans.Front Immunol. 2022 Aug 24;13:953439. doi: 10.3389/fimmu.2022.953439. eCollection 2022. Front Immunol. 2022. PMID: 36090990 Free PMC article. Review.
-
Suppression of lupus nephritis and skin lesions in MRL/lpr mice by administration of the topoisomerase I inhibitor irinotecan.Arthritis Res Ther. 2016 Oct 22;18(1):243. doi: 10.1186/s13075-016-1144-5. Arthritis Res Ther. 2016. PMID: 27770825 Free PMC article.
-
The value of animal models to study immunopathology of primary human Sjögren's syndrome symptoms.Expert Rev Clin Immunol. 2014 Apr;10(4):469-81. doi: 10.1586/1744666X.2014.883920. Epub 2014 Feb 10. Expert Rev Clin Immunol. 2014. PMID: 24506531 Free PMC article. Review.
-
Recent Advances in Mouse Models of Sjögren's Syndrome.Front Immunol. 2020 Jun 30;11:1158. doi: 10.3389/fimmu.2020.01158. eCollection 2020. Front Immunol. 2020. PMID: 32695097 Free PMC article. Review.
References
-
- Qian Y, Qin J, Cui G, Naramura M, Snow EC, Ware CF, Fairchild RL, et al. Act1, a negative regulator in CD40- and BAFF-mediated B cell survival. Immunity. 2004;21:575–587. - PubMed
-
- Higuchi T, Aiba Y, Nomura T, Matsuda J, Mochida K, Suzuki M, Kikutani H, et al. Cutting Edge: Ectopic expression of CD40 ligand on B cells induces lupus-like autoimmune disease. J Immunol. 2002;168:9–12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

