Differential effects of lipids and lyso-lipids on the mechanosensitivity of the mechanosensitive channels MscL and MscS
- PMID: 22586095
- PMCID: PMC3365151
- DOI: 10.1073/pnas.1200051109
Differential effects of lipids and lyso-lipids on the mechanosensitivity of the mechanosensitive channels MscL and MscS
Abstract
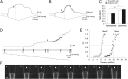
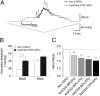
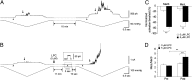
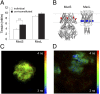
Mechanosensitive (MS) channels of small (MscS) and large (MscL) conductance are the major players in the protection of bacterial cells against hypoosmotic shock. Although a great deal is known about structure and function of these channels, much less is known about how membrane lipids may influence their mechanosensitivity and function. In this study, we use liposome coreconstitution to examine the effects of different types of lipids on MscS and MscL mechanosensitivity simultaneously using the patch-clamp technique and confocal microscopy. Fluorescence lifetime imaging (FLIM)-FRET microscopy demonstrated that coreconstitution of MscS and MscL led to clustering of these channels causing a significant increase in the MscS activation threshold. Furthermore, the MscL/MscS threshold ratio dramatically decreased in thinner compared with thicker bilayers and upon addition of cholesterol, known to affect the bilayer thickness, stiffness and pressure profile. In contrast, application of micromolar concentrations of lysophosphatidylcholine (LPC) led to an increase of the MscL/MscS threshold ratio. These data suggest that differences in hydrophobic mismatch and bilayer stiffness, change in transbilayer pressure profile, and close proximity of MscL and MscS affect the structural dynamics of both channels to a different extent. Our findings may have far-reaching implications for other types of ion channels and membrane proteins that, like MscL and MscS, may coexist in multiple molecular complexes and, consequently, have their activation characteristics significantly affected by changes in the lipid environment and their proximity to each other.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Hamill OP, Martinac B. Molecular basis of mechanotransduction in living cells. Physiol Rev. 2001;81:685–740. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

