Extracellular nucleotides inhibit insulin receptor signaling, stimulate autophagy and control lipoprotein secretion
- PMID: 22590634
- PMCID: PMC3349634
- DOI: 10.1371/journal.pone.0036916
Extracellular nucleotides inhibit insulin receptor signaling, stimulate autophagy and control lipoprotein secretion
Abstract
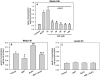
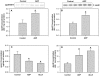
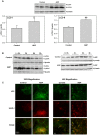
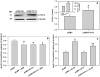
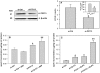
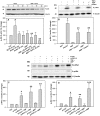
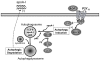
Hyperglycemia is associated with abnormal plasma lipoprotein metabolism and with an elevation in circulating nucleotide levels. We evaluated how extracellular nucleotides may act to perturb hepatic lipoprotein secretion. Adenosine diphosphate (ADP) (>10 µM) acts like a proteasomal inhibitor to stimulate apoB100 secretion and inhibit apoA-I secretion from human liver cells at 4 h and 24 h. ADP blocks apoA-I secretion by stimulating autophagy. The nucleotide increases cellular levels of the autophagosome marker, LC3-II, and increases co-localization of LC3 with apoA-I in punctate autophagosomes. ADP affects autophagy and apoA-I secretion through P2Y(13). Overexpression of P2Y(13) increases cellular LC3-II levels by ~50% and blocks induction of apoA-I secretion. Conversely, a siRNA-induced reduction in P2Y(13) protein expression of 50% causes a similar reduction in cellular LC3-II levels and a 3-fold stimulation in apoA-I secretion. P2Y(13) gene silencing blocks the effects of ADP on autophagy and apoA-I secretion. A reduction in P2Y(13) expression suppresses ERK1/2 phosphorylation, increases the phosphorylation of IR-β and protein kinase B (Akt) >3-fold, and blocks the inhibition of Akt phosphorylation by TNFα and ADP. Conversely, increasing P2Y(13) expression significantly inhibits insulin-induced phosphorylation of insulin receptor (IR-β) and Akt, similar to that observed after treatment with ADP. Nucleotides therefore act through P2Y(13), ERK1/2 and insulin receptor signaling to stimulate autophagy and affect hepatic lipoprotein secretion.
Conflict of interest statement
Figures

Similar articles
-
Hepatic lipase release is inhibited by a purinergic induction of autophagy.Cell Physiol Biochem. 2014;33(4):883-94. doi: 10.1159/000358661. Epub 2014 Mar 28. Cell Physiol Biochem. 2014. PMID: 24713587
-
Adenosine nucleotides acting at the human P2Y1 receptor stimulate mitogen-activated protein kinases and induce apoptosis.J Biol Chem. 2001 May 11;276(19):16379-90. doi: 10.1074/jbc.M006617200. Epub 2001 Jan 25. J Biol Chem. 2001. PMID: 11278310
-
P2 receptor-mediated stimulation of the PI3-K/Akt-pathway in vivo.Glia. 2009 Aug 1;57(10):1031-45. doi: 10.1002/glia.20827. Glia. 2009. PMID: 19115395
-
ATGs: Scaffolds for MAPK/ERK signaling.Autophagy. 2014 Mar;10(3):535-7. doi: 10.4161/auto.27642. Epub 2014 Jan 7. Autophagy. 2014. PMID: 24412893 Free PMC article. Review.
-
The P2 receptors and congenital platelet function defects.Semin Thromb Hemost. 2005 Apr;31(2):168-73. doi: 10.1055/s-2005-869522. Semin Thromb Hemost. 2005. PMID: 15852220 Review.
Cited by
-
The PRKAA1/AMPKα1 pathway triggers autophagy during CSF1-induced human monocyte differentiation and is a potential target in CMML.Autophagy. 2015;11(7):1114-29. doi: 10.1080/15548627.2015.1034406. Autophagy. 2015. PMID: 26029847 Free PMC article.
-
New Potential Pharmacological Functions of Chinese Herbal Medicines via Regulation of Autophagy.Molecules. 2016 Mar 17;21(3):359. doi: 10.3390/molecules21030359. Molecules. 2016. PMID: 26999089 Free PMC article. Review.
-
Testosterone regulates the autophagic clearance of androgen binding protein in rat Sertoli cells.Sci Rep. 2015 Mar 9;5:8894. doi: 10.1038/srep08894. Sci Rep. 2015. PMID: 25745956 Free PMC article.
-
Clodronate, an inhibitor of the vesicular nucleotide transporter, ameliorates steatohepatitis and acute liver injury.Sci Rep. 2021 Mar 4;11(1):5192. doi: 10.1038/s41598-021-83144-w. Sci Rep. 2021. PMID: 33664289 Free PMC article.
-
Extracellular ATP Increases Glucose Metabolism in Skeletal Muscle Cells in a P2 Receptor Dependent Manner but Does Not Contribute to Palmitate-Induced Insulin Resistance.Front Physiol. 2020 Sep 25;11:567378. doi: 10.3389/fphys.2020.567378. eCollection 2020. Front Physiol. 2020. PMID: 33101053 Free PMC article.
References
-
- Grundy SM. Hypertriglyceridemia, atherogenic dyslipidemia, and the metabolic syndrome. Am J Cardiol. 1998;81:18B–25B. - PubMed
-
- Adiels M, Olofsson SO, Taskinen MR, Boren J. Overproduction of very low-density lipoproteins is the hallmark of the dyslipidemia in the metabolic syndrome. Arterioscler Thromb Vasc Biol. 2008;28:1225–1236. - PubMed
-
- Solini A, Iacobini C, Ricci C, Chiozzi P, Amadio L, et al. Purinergic modulation of mesangial extracellular matrix production: role in diabetic and other glomerular diseases. Kidney Int. 2005;67:875–885. - PubMed
-
- Nilsson J, Nilsson LM, Chen YW, Molkentin JD, Erlinge D, et al. High glucose activates nuclear factor of activated T cells in native vascular smooth muscle. Arterioscler Thromb Vasc Biol. 2006;26:794–800. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

