Anti-leukotriene agents compared to inhaled corticosteroids in the management of recurrent and/or chronic asthma in adults and children
- PMID: 22592685
- PMCID: PMC4164381
- DOI: 10.1002/14651858.CD002314.pub3
Anti-leukotriene agents compared to inhaled corticosteroids in the management of recurrent and/or chronic asthma in adults and children
Abstract
Background: Anti-leukotrienes (5-lipoxygenase inhibitors and leukotriene receptors antagonists) serve as alternative monotherapy to inhaled corticosteroids (ICS) in the management of recurrent and/or chronic asthma in adults and children.
Objectives: To determine the safety and efficacy of anti-leukotrienes compared to inhaled corticosteroids as monotherapy in adults and children with asthma and to provide better insight into the influence of patient and treatment characteristics on the magnitude of effects.
Search methods: We searched MEDLINE (1966 to Dec 2010), EMBASE (1980 to Dec 2010), CINAHL (1982 to Dec 2010), the Cochrane Airways Group trials register, and the Cochrane Central Register of Controlled Trials (Dec 2010), abstract books, and reference lists of review articles and trials. We contacted colleagues and the international headquarters of anti-leukotrienes producers.
Selection criteria: We included randomised trials that compared anti-leukotrienes with inhaled corticosteroids as monotherapy for a minimum period of four weeks in patients with asthma aged two years and older.
Data collection and analysis: Two review authors independently assessed the methodological quality of trials and extracted data. The primary outcome was the number of patients with at least one exacerbation requiring systemic corticosteroids. Secondary outcomes included patients with at least one exacerbation requiring hospital admission, lung function tests, indices of chronic asthma control, adverse effects, withdrawal rates and biological inflammatory markers.
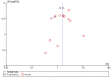
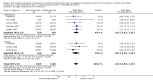
Main results: Sixty-five trials met the inclusion criteria for this review. Fifty-six trials (19 paediatric trials) contributed data (representing total of 10,005 adults and 3,333 children); 21 trials were of high methodological quality; 44 were published in full-text. All trials pertained to patients with mild or moderate persistent asthma. Trial durations varied from four to 52 weeks. The median dose of inhaled corticosteroids was quite homogeneous at 200 µg/day of microfine hydrofluoroalkane-propelled beclomethasone or equivalent (HFA-BDP eq). Patients treated with anti-leukotrienes were more likely to suffer an exacerbation requiring systemic corticosteroids (N = 6077 participants; risk ratio (RR) 1.51, 95% confidence interval (CI) 1.17, 1.96). For every 28 (95% CI 15 to 82) patients treated with anti-leukotrienes instead of inhaled corticosteroids, there was one additional patient with an exacerbation requiring rescue systemic corticosteroids. The magnitude of effect was significantly greater in patients with moderate compared with those with mild airway obstruction (RR 2.03, 95% CI 1.41, 2.91 versus RR 1.25, 95% CI 0.97, 1.61), but was not significantly influenced by age group (children representing 23% of the weight versus adults), anti-leukotriene used, duration of intervention, methodological quality, and funding source. Significant group differences favouring inhaled corticosteroids were noted in most secondary outcomes including patients with at least one exacerbation requiring hospital admission (N = 2715 participants; RR 3.33; 95% CI 1.02 to 10.94), the change from baseline FEV(1) (N = 7128 participants; mean group difference (MD) 110 mL, 95% CI 140 to 80) as well as other lung function parameters, asthma symptoms, nocturnal awakenings, rescue medication use, symptom-free days, the quality of life, parents' and physicians' satisfaction. Anti-leukotriene therapy was associated with increased risk of withdrawals due to poor asthma control (N = 7669 participants; RR 2.56; 95% CI 2.01 to 3.27). For every thirty one (95% CI 22 to 47) patients treated with anti-leukotrienes instead of inhaled corticosteroids, there was one additional withdrawal due to poor control. Risk of side effects was not significantly different between both groups.
Authors' conclusions: As monotherapy, inhaled corticosteroids display superior efficacy to anti-leukotrienes in adults and children with persistent asthma; the superiority is particularly marked in patients with moderate airway obstruction. On the basis of efficacy, the results support the current guidelines' recommendation that inhaled corticosteroids remain the preferred monotherapy.
Conflict of interest statement
Bhupendrasinh Chauhan ‐ none known.
Prof. Francine Ducharme has received travel support, research funds and fees for speaking from Glaxo SmithKline, Novartis, Nycomed, and Merck Frosst Inc.
Figures











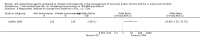




























































Update of
-
Anti-leukotriene agents compared to inhaled corticosteroids in the management of recurrent and/or chronic asthma in adults and children.Cochrane Database Syst Rev. 2004;(2):CD002314. doi: 10.1002/14651858.CD002314.pub2. Cochrane Database Syst Rev. 2004. Update in: Cochrane Database Syst Rev. 2012 May 16;(5):CD002314. doi: 10.1002/14651858.CD002314.pub3. PMID: 15106175 Updated.
References
References to studies included in this review
Abadoglu 2005 {published data only}
-
- Abadoglu O, Mungan D, Aksu O, Erekul S, Misirligil Z. The effect of montelukast on eosinophil apoptosis: induced sputum findings of patients with mild persistent asthma. Allergol Immunopathol 2005;33(2):105‐11. - PubMed
Basyigit 2004 {published data only}
-
- Basyigit I, Yildiz F, Kacar Ozkara S, Boyaci H, Ilgazli A, Ozkarakas O. Effects of different anti‐asthmatic agents on induced sputum and eosinophil cationic protein in mild asthmatics. Respirology 2004;9(4):514‐20. - PubMed
Baumgartner 2003 {published data only}
-
- Baumgartner RA, Martinez G, Edelma JM, Rodriguez Gomez GG, Berstein M, Bird S, et al. Distribution of therapeutic response in asthma control between oral montelukast and inhaled beclomethasone. European Respiratory Journal 2003; Vol. 21:123‐8. - PubMed
Bleecker 2000 {unpublished data only}
-
- Bleecker ER, Welch MJ, Weinstein SF, Kalberg C, Johnson M, Edwards L, et al. Low‐dose inhaled fluticasone propionate versus oral zafirlukast in the treatment of persistent asthma. Journal of Allergy & Clinical Immunology 2000;105(6):1123‐9. - PubMed
Boushey 2005 {published data only}
-
- Boushey HA, Sorkness CA, King TS, Sullivan SD, Fahy JV, Lazarus SC, et al. Daily versus as‐needed corticosteroids for mild persistent asthma. New England Jounal of medicine 2005;352(15):1519‐1528. - PubMed
Bousquet 2005 {published data only}
-
- Bousquet J, Menten J, Tozzi CA, Polos PG. Oral montelukast sodium versus inhaled fluticasone propionate in adults with mild persistent asthma. Journal of Applied Research 2005;5(3):402‐14.
Brabson 2002 {published data only}
-
- Brabson JH, Clifford D, Kerwin E, Raphael G, Pepsin PJ, Edwards LD, et al. Efficacy and safety of low‐dose fluticasone propionate compared with zafirlukast in patients with persistent asthma. American Journal of Medicine 2002;113:15‐21. - PubMed
Busse 2001a {published data only}
-
- Busse W, Wolfe J, Storms W, Srebro S, Edwards L, Johnson M, et al. Fluticasone propionate compared to Zafirlukast in controlling persistent asthma: A randomised double‐blind, placebo‐controlled trial. Journal of Family Practice 2001;50:595‐602. - PubMed
Busse 2001b {published data only}
-
- Busse W, Raphael GD, Galant S, Kalberg C, Goode‐Sellers S, Srebro S, et al. Low‐dose fluticasone propionate compared to montelukast for first‐line treatment of persistent asthma: a randomised controlled trial. Journal of Allergy & Clinical Immunology 2001;107(3):461‐8. - PubMed
Caffey 2005 {published data only}
-
- Caffey LF, Raissy HH, Marshik P, Kelly HW. A crossover comparison of fluticasone propionate and montelukast on inflammatory indices in children with asthma. Pediatric Asthma Allergy & Immunology 2005;18(3):123‐30.
Dempsey 2002a {published data only}
-
- Dempsey OJ, Wilson AM, Lipworth BJ. Comparative efficacy and anti‐inflammatory profile of once daily low dose hfa‐triamcinolone acetonide (taa) and montelukast (ml) in patients with mild persistent atopic asthma. Journal of Allergy and Clinical Immunology 2002;109(1):68‐74. - PubMed
FLTA4030 {unpublished data only}
-
- A randomized, double‐blind, double dummy, placebo‐controlled, parallel‐group, comparative study of inhaled fluticasone propionate (88 mcg bid) versus zafirlukast (20 mg bid) in subjects who are currently receiving beta agonists alone. www.gsk‐clinicalstudyregister.com.
FLTA4031 {unpublished data only}
-
- A randomized, double‐blind, double‐dummy, placebo‐controlled, parallel‐group, comparative study of inhaled fluticasone propionate (88 mcg bid) versus zafirlukast (20 mg bid) in subjects who are currently receiving beta agonists alone. www.gsk‐clinicalstudyregister.com.
FMS40012 {unpublished data only}
-
- Evaluation of the potential anti‐inflammatory action of leukotriene D4 receptor antagonists: comparison of zafirlukast, an LTD4 receptor antagonist with low‐dose fluticasone propionate, an inhaled steroid on sputum eosinophils in mild asthma. GlaxoSmithKline Clinical Trial Register 2005.
FPD40013 {unpublished data only}
-
- FPD40013. A randomized, double‐blind, double dummy, parallel group comparison of fluticasone propionate inhalation powder (50 mcg BID) via discus with oral montelukast (5 mg QD) chewable tablets in children 6‐12 years of age with persistent asthma. GlaxoSmithKline Clinical Trial Register 2005.
Garcia Garcia 2005 {published data only}
-
- Garcia Garcia ML, Wahn U, Gilles L, Swern A, Tozzi CA, Polos P. Montelukast, compared with fluticasone, for control of asthma among 6‐ to 14‐year‐old patients with mild asthma: the MOSAIC study. Pediatrics 2005;116(2):360‐9. - PubMed
Hughes 1999 (BDP) {unpublished data only}
-
- Hughes GL, Edelman JM, Turpin JA, Liss C, Weeks K. Randomized, open‐label pilot study comparing the effects of montelukast sodium tablets, fluticasone aerosol inhaler, and budesonide dry powder inhaler on asthma control in mild asthmatics. American Journal of Respiratory & Critical Care Medicine 1999;159:A641.
Hughes 1999 (FP) {unpublished data only}
-
- Hughes GL, Edelman JM, Turpin JA, Liss C, Weeks K. Randomized, open‐label pilot study comparing the effects of montelukast sodium tablets, fluticasone aerosol inhaler, and budesonide dry powder inhaler on asthma control in mild asthmatics. American Journal of Respiratory & Critical Care Medicine 1999;159:A641.
Israel 2002 {published data only}
-
- Israel E, Chervinsky PS, Friedman B, Bavel J, Skalky CS, Ghannam AF, et al for the Montelukast Beclomethasone Comparison Study Group. Effects of montelukast and beclomethasone on airway function and asthma control. Journal of Allergy and Clinical Immunology 2002;110(6):847‐54. - PubMed
Jayaram 2002 {published data only}
-
- Jayaram L, Pizzichini MMM, Hussack P, Efthimiadis A, Lemiere C, Cartier A, et al. First line anti‐inflammatory treatment for asthma; inhaled steroid or leukotriene antagonist?. Respirology 2002;7(A19):21.
Jayaram 2005 {published data only}
Jenkins 2005 {published data only}
-
- Jenkins CR, Thien FCK, Wheatley JR, Reddel HK. Traditional and patient‐centred outcomes with three classes of asthma medication. European Respiratory Journal 2005;26(1):36‐44. - PubMed
Kanazawa 2007 {published data only}
-
- Kanazawa H, Nomura S, Asai K. Roles of angiopoietin‐1 and angiopoietin‐2 on airway microvascular permeability in asthmatic patients. Chest 2007;131(4):1035‐41. - PubMed
Kanniess 2002 {published data only}
-
- Kanniess F, Richter K, Bohme S, Jorres RA, Magnussen H. Montelukast versus fluticasone: effects on lung function, airway responsiveness and inflammation in moderate asthma. European Respiratory Journal 2002;20:853‐8. - PubMed
Khan 2008 {published data only}
-
- Khan SA, Hashmi ZY. Comparison of therapeutic values between leukotriene receptor antagonist (Montelukast) and inhaled glucocorticoid (Beclomethasone propionate) in bronchial asthma of adults. Pakistan Journal of Medical Sciences 2008;24(3):399‐405.
Kim 2000 {published and unpublished data}
-
- Kim KT, Ginchansky EJ, Friedman BF, Srebro S, Pepsin PJ, Edwards L, et al. Fluticasone propionate versus zafirlukast: effect in patients previously receiving inhaled corticosteroid therapy. Annals of Allergy, Asthma, & Immunology 2000;85:398‐406. - PubMed
Koenig 2008 {published data only}
-
- Koenig SM, Ostrom N, Pearlman D, Waitkus‐Edwards K, Yancey S, Prillaman BA, et al. Deterioration in Asthma Control When Subjects Receiving Fluticasone Propionate/Salmeterol 100/50 mcg Diskus are "Stepped‐Down". Journal of Asthma 2008;45:681‐7. - PubMed
Kooi 2008 {published data only}
-
- Kooi EMW, Schokker S, Marike Boezen H, Vries TW, Vaessen‐Verberne AAPH, Molen T, et al. Fluticasone or montelukast for preschool children with asthma‐like symptoms: Randomized controlled trial. Pulmonary Pharmacology and Therapeutics 2008;21(5):798‐804. - PubMed
Kumar 2007 {published data only}
-
- Kumar V, Ramesh P, Lodha R, Pandey RM, Kabra SK. Montelukast vs. inhaled low‐dose budesonide as monotherapy in the treatment of mild persistent asthma: A randomized double blind controlled trial. Journal of Tropical Pediatrics 2007;53(5):325‐30. - PubMed
Laitinen 1997 {unpublished data only}
-
- Laitinen LA, Naya IP, Binks S, Harris A. Comparative efficacy of zafirlukast & low dose steroids in asthmatics on prn beta2‐agonists. European Respiratory Journal. 1997; Vol. 10, issue Suppl 25:419‐20, Abs. 2716.
Laviolette 1999 {published and unpublished data}
-
- Laviolette M, Malmstrom K, Lu S, Chervinsky P, Pujet J‐C, Peszek I, et al. Montelukast added to inhaled beclomethasone in treatment of asthma. American Journal of Respiratory & Critical Care Medicine 1999;160(6):1862‐8. - PubMed
Lazarus 2007 {published data only}
Lu 2009 {published data only}
-
- Lu S, Liu N, Dass SB, Reiss TF. A randomized study comparing the effect of loratadine added to montelukast with montelukast, loratadine, and beclomethasone monotherapies in patients with chronic asthma. Journal of Asthma 2009;46(5):465‐9. - PubMed
Malmstrom 1999 {published and unpublished data}
-
- Malmstrom K, Rodriguez‐Gomez G, Guerra J, Villaran C, Pineiro A, Lynn X, et al. Oral montelukast, inhaled beclomethasone, and placebo for chronic asthma: a randomized, controlled trial. Annals of Internal Medicine 1999;130(6):487‐95. - PubMed
Maspero 2001 {published data only}
-
- Maspero JF, Duenas‐Meza E, Volovitz B, Daza CP, Kosa L, Vrijens F, et al. Oral montelukast versus inhaled beclomethasone in 6 to 11‐year old children with asthma: results of an open‐label extension study evaluating long‐term safety, satisfaction, and adherence to therapy. Current Medical Research and Opinion 2001; Vol. 17, issue 2:96‐104. - PubMed
Meltzer 2002 {published data only}
-
- Meltzer EO, Lockey RF, Friedman BF, Kalberg C, Goode‐Sellers S, Srebro S, et al for the Fluticasone Propionale Clinical Research Study Group. Efficacy and safety of low‐dose fluticasone propionate compared with montelukast for maintenance treatment of persistent asthma. Mayo Clinc Proceedings 2002;77:437‐5. - PubMed
MK0479‐332 {unpublished data only}
-
- Merck. Montelukast asthmatic smoker study. Clinicaltrials.Gov 2006.
Nathan 2001 {published data only}
-
- Nathan RA, Bleecker ER, Kalberg C and the Fluticasone Propionate Study Group. A comparison of short‐term treatment with inhaled fluticasone propionate and zafirlukast for patients with persistent asthma. American Journal of Medicine 2001;111:195‐202. - PubMed
NCT00442559 {unpublished data only}
-
- Montelukast in mild asthmatic children with allergic rhinitis. Clinicaltrials.gov 2008.
Ng 2007 {published data only}
-
- Ng DKK, Chan CH, Wu S, Chow PY, Wong LSW, Fu YM, et al. Oral montelukast versus inhaled budesonide in children with mild persistent asthma: A pilot study. The Hong Kong Journal of Paediatrics 2007;12(1):3‐10.
Ostrom 2005 {published data only}
-
- Ostrom NK, Decotiis BA, Lincourt WR, Edwards LD, Hanson KM, Carranza Rosenzweig JR, et al. Comparative efficacy and safety of low‐dose fluticasone propionate and montelukast in children with persistent asthma. Journal of Pediatrics 2005;147((2):213‐20. - PubMed
Overbeek 2004 {published data only}
-
- Overbeek SE, O'Sullivan S, Leman K, Mulder PGH, Hoogsteden HC, Prins J‐B. Effect of montelukast compared with inhaled fluticasone on airway inflammation. Clinical & Experimental Allergy 2004;34(9):1388‐94. - PubMed
Peroni 2005 {published data only}
-
- Peroni D, Bodini A, Miraglia Del Giudice M, Loiacono A, Baraldi E, Boner AL, et al. Effect of budesonide and montelukast in asthmatic children exposed to relevant allergens. Allergy 2005;60(2):206‐10. - PubMed
Peters 2007 {published data only}
-
- Peters SP, Anthonisen N, Castro M, Holbrook JT, Irvin CG, Smith LJ, et al. Randomized comparison of strategies for reducing treatment in mild persistent asthma. New England Journal of Medicine 2007;356(20):2027‐39. - PubMed
Riccioni 2001 {published data only}
-
- Riccioni G, Castronuove M, Benedictis M, Pace‐Palitti V, Gioacchino M, Della Vecchia R, et al. Zafirlukast versus budesonide on bronchial reactivity in subjects with mild‐persistent asthma. International Journal of Immunopathology and Pharmacology 2001;14(2):87‐92. - PubMed
Riccioni 2002a {published data only}
-
- Riccioni G, D'Orazio N, Ilio C, Della Vecchia R, Lorenzo A. Effectiveness and safety of montelukast versus budesonide at various doses on bronchial reactivity in subjects with mild persistent asthma [Efficacia e tollerabilita del montelukast verso budesonide a diverso dosaggio sulla reattivita bronchiale in soggetti con asma di grado lieve‐persistente]. La Clinica terapeutica 2002;153(5):317‐21. - PubMed
Riccioni 2002b {published data only}
-
- Riccioni G, Ballone E, D'Orazio N, Sensi S, Nicola M, Mascio R, et al. Effectiveness of montelukast versus budesonide on quality of life and bronchial reactivity in subjects with mild‐persistent asthma. International Journal of Immunopathology & Pharmacology 2002;15(2):149‐55. - PubMed
Riccioni 2003 {published data only}
-
- Riccioni G, Vecchia RD, D'Orazio N, Sensi S, Guagnano MT. Comparison of montelukast and budesonide on bronchial reactivity in subjects with mild‐moderate persistent asthma. Pulmonary Pharmacology & Therapeutics 2003;16(2):111‐4. - PubMed
Sheth 2001 {published data only}
-
- Sheth K, Edwards L, Srebro S, Rickard K. Effects of baseline pulmonary function on treatment response: low‐dose fluticasone versus zafirlukast. Annals of Allergy, Asthma and IImmunology 2001;86:2001.
Sorkness 2007 {published data only}
-
- Sorkness CA, Lemanske Jr RF, Mauger DT, Boehmer SJ, Chinchilli VM, Martinez FD. Long‐term comparison of 3 controller regimens for mild‐moderate persistent childhood asthma: The Pediatric Asthma Controller Trial. Journal of Allergy & Clinical Immunology 2007;119(1):64‐72. - PubMed
Stelmach 2002a {published data only}
-
- Stelmach I, Jerzynska J, Kuna P. A randomized, double‐blind trial of the effect of glucocorticoid, antileukotriene and beta‐agonist treatment on IL‐10 serum levels in children with asthma. Clinical & Experimental Allergy 2002;32(2):264‐9. - PubMed
Stelmach 2002b {published and unpublished data}
-
- Stelmach I, Grzelewski T, Stelmach W, Majak P, Jerzynska J, Gorski P, et al. Effect of triamcinolone acetonide, montelukast, nedocromil sodium and formoterol on eosinophil blood counts, ECP serum levels and clinical progression of asthma in children [Polish]. Polski Merkuriusz Lekarski 2002;12(69):208‐13. - PubMed
Stelmach 2004 {published data only}
-
- Stelmach I, Majak P, Jerzynska J, Stelmach W, Kuna P. Comparative effect of triamcinolone, nedocromil and montelukast on asthma control in children: A randomized pragmatic study. Pediatric Allergy & Immunology 2004;15(4):359‐64. - PubMed
Stelmach 2005 {published data only}
-
- Stelmach I, Bobrowska‐Korzeniowska M, Majak P, Stelmach W, Kuna P. The effect of montelukast and different doses of budesonide on IgE serum levels and clinical parameters in children with newly diagnosed asthma. Pulmonary Pharmacology & Therapeutics 2005;18(5):374‐80. - PubMed
Stelmach 2007 {published data only}
-
- Stelmach I, Grzelewski T, Bobrowska‐Korzeniowska M, Stelmach P, Kuna P. A randomized, double‐blind trial of the effect of anti‐asthma treatment on lung function in children with asthma. Pulmonary Pharmacology and Therapeutics 2007;20(6):691‐700. - PubMed
Stelmach 2008 {published data only}
-
- Stelmach I, Grzelewski T, Majak P, Jerzynska J, Stelmach W, Kuna P. Effect of different antiasthmatic treatments on exercise‐induced bronchoconstriction in children with asthma. Journal of Allergy Clinical Immunology 2008;121(2):383‐9. - PubMed
Szefler 2005 {published data only}
-
- Szefler SJ, Phillips BR, Martinez FD, Chinchilli VM, Lemanske RF, Strunk RC. Characterization of within‐subject responses to fluticasone and montelukast in childhood asthma. Journal of Allergy and Clinical Immunology 2005;115(2):233‐42. - PubMed
Szefler 2007 {published data only}
-
- Szefler SJ, Baker JW, Uryniak T, Goldman M, Silkoff PE. Comparative study of budesonide inhalation suspension and montelukast in young children with mild persistent asthma. Journal of Allergy and Clinical Immunology 2007;120(5):1043‐50. - PubMed
Tamaoki 2008 {published data only}
-
- Tamaoki J, Isono K, Taira M, Tagaya E, Nakata J, Kawatani K, et al. Role of regular treatment with inhaled corticosteroid or leukotriene receptor antagonist in mild intermittent asthma. Allergy and Asthma Proceedings : the Official Journal of Regional and State Allergy Societies 2008;29(2):189‐96. - PubMed
Yamauchi 2001 {published data only}
-
- Yamauchi K, Tanifuji Y, Pan LH, Yoshida T, Sakurai S, Goto SI, et al. Effects of Pranlukast, a Leukotriene receptor antagonist, on airway inflammation in mild asthmatics. Journal of Asthma 2001;38(1):51‐7. - PubMed
Yurdakul 2003 {published data only}
-
- Yurdakul AS, Taci N, Eren A, Sipit T. Comparative efficacy of once‐daily therapy with inhaled corticosteroid, leukotriene antagonist or sustained‐release theophylline in patients with mild persistent asthma. Respiratory Medicine 2003;97(12):1313‐9. - PubMed
Zedan 2009 {published data only}
-
- Zedan M, Gamil N, El‐Chennawi F, Maysara N, Hafeez HA, Nasef N, et al. Evaluation of different asthma phenotype responses to montelukast versus fluticasone Treatment. Pediatric Asthma, Allergy and Immunology 2009;22(2):63‐9.
Zeiger 2005 {published data only}
-
- Zeiger RS, Bird SR, Kaplan MS, Schatz M, Pearlman DS, Orav EJ. Short‐term and long‐term asthma control in patients with mild persistent asthma receiving montelukast or fluticasone: a randomized controlled trial. American Journal of Medicine 2005;118(6):649‐57. - PubMed
Zeiger 2006 {published data only}
-
- Zeiger RS, Szefler SJ, Phillips BR, Schatz M, Martinez FD, Chinchilli VM. Response profiles to fluticasone and montelukast in mild‐to‐moderate persistent childhood asthma. Journal of Allergy & Clinical Immunology 2006;117(1):45‐52. - PubMed
Zielen 2010 {published data only}
-
- Zielen S, Christmann M, Kloska M, Dogan‐Yildiz G, Lieb A, Rosewich M. Predicting short term response to anti‐inflammatory therapy in young children with asthma. Current Medical Research and Opinion 2010;26(2):483‐92. - PubMed
References to studies excluded from this review
Abbott Pharma 1996 {published data only}
-
- Abbott Pharmaceuticals. Zyflo Filmtab (zileuton) product description. Abbott Laboratories 1996.
Al Frayh 2008 {published data only}
-
- Al Frayh A, Abba A, Iskandarani A, Shaker DS, Zaitoun F, Shahrabani L, et al. Establishing therapeutic bioequivalence of a generic salbutamol (Butalin) metered dose inhaler to Ventolin. Biomedical Research 2008;19(1):61‐8.
Allayee 2007 {published data only}
-
- Allayee H, Hartiala J, Lee W, Mehrabian M, Irvin CG, Conti DV, et al. The effect of montelukast and low‐dose theophylline on cardiovascular disease risk factors in asthmatics. Chest 2007;132(3):868‐74. - PubMed
Allen 1997 {published data only}
-
- Allen A, Georgiou P, Compton C, Walls C, Hibell M, Tomson DJ, et al. Lack of pharmacokinetic and pharmacodynamic interactions between pranlukast (Ultair) and terfenadine. American Thoracic Society. 1997:Abs C49.
Allen‐Ramey 2003 {published data only}
-
- Allen‐Ramey FC, Duong PT, Goodman DC, Sajjan SG, Nelsen LM, Santanello NC, et al. Treatment effectiveness of inhaled corticosteroids and leukotriene modifiers for patients with asthma: an analysis from managed care data. Allergy & Asthma Proceedings 2003;24(1):43‐51. - PubMed
Allen‐Ramey 2004 {published data only}
-
- Allen‐Ramey FC, Anstatt DT, Riedel AA, Markson LE. Greater adherence in montelukast patients compared to those on fluticasone [Abstract]. Journal of Allergy and Clinical Immunology 2004;113(2 Suppl):S158.
Allen‐Ramey 2006 {published data only}
-
- Allen‐Ramey FC, Markson LE, Riedel AA, Sajjan S, Weiss KB. Patterns of asthma‐related health care resource use in children treated with montelukast or fluticasone. Current Medical Research & Opinion 2006;22(8):1453‐61. - PubMed
Altman 1998k {published data only}
-
- Altman LC, Munk Z, Seltzer J, Noonan N, Shingo S, Zhang J, et al. A placebo‐controlled, dose‐ranging study of montelukast, a cysteinyl leukotriene‐receptor antagonist. Journal of Allergy & Clinical Immunology 1998;102:50‐6. - PubMed
Anonymous 1997 {published data only}
-
- Anonymous. Zileuton for asthma. Medical Letter on Drugs & Therapeutics 1997;39(995):18‐9. - PubMed
Armour 2007 {published data only}
Bacharier 2008 {published data only}
-
- Bacharier LB, Phillips BR, Zeiger RS, Szefler SJ, Martinez FD, Lemanske RF, et al. Episodic use of an inhaled corticosteroid or leukotriene receptor antagonist in preschool children with moderate‐to‐severe intermittent wheezing. The Journal of Allergy and Clinical Immunology 2008;122(6):1127‐35. - PMC - PubMed
Bai 2010 {published data only}
-
- Bai J, Xu PR. Montelukast in the treatment of bronchiolitis, a multi‐center, randomized, three‐blind, placebo‐controlled trial. Chinese Journal of Evidence‐Based Medicine 2010;10(9):1011‐5.
Balatsouras 2005 {published data only}
-
- Balatsouras DG, Eliopoulos P, Rallis E, Sterpi P, Korres S, Ferekidis E. Improvement of otitis media with effusion after treatment of asthma with leukotriene antagonists in children with co‐existing disease. Drugs Under Experimental & Clinical Research 2005;31(SUPPL):7‐10. - PubMed
Baren 2006 {published data only}
-
- Baren JM, Boudreaux ED, Brenner BE, Cydulka RK, Rowe BH, Clark S, et al. Randomized controlled trial of emergency department interventions to improve primary care follow‐up for patients with acute asthma. Chest 2006;129(2):257‐65. - PubMed
Barnes 1996 {published data only}
-
- Barnes NC, Black B, Syrett N, Cohn J. Reduction of exacerbations of asthma in multi‐national clinical trials with zafirlukast (Accolate). Allergy 1996;51(Suppl 30):84.
Barnes 1997 {published data only}
-
- Barnes NC, Jong B, Miyamoto T. Worldwide clinical experience with the first marketed leukotriene receptor antagonist. Chest 1997;111 Suppl(2):52‐60. - PubMed
Barnes 1997b {published data only}
Barnes 2001 {unpublished data only}
-
- Barnes N, Wei LX, Reiss TF, Leff JA, Shingo S, Yu C, et al. Analysis of montelukast in mild persistent asthmatic patients with near‐normal lung function. Respiratory Medicine 2001;95(5):379‐86. - PubMed
Barnes 2007 {published data only}
-
- Barnes ML, Menzies D, Fardon TC, Burns P, Wilson AM, Lipworth BJ. Combined mediator blockade or topical steroid for treating the unified allergic airway. Allergy 2007;62(1):73‐80. - PubMed
Barnes 2007a {published data only}
-
- Barnes N, Laviolette M, Allen D, Flood‐Page P, Hargreave F, Corris P, et al. Effects of montelukast compared to double dose budesonide on airway inflammation and asthma control. Respiratory Medicine 2007;101(8):1652‐8. - PubMed
Bartoli 2010 {published data only}
-
- Bartoli ML, Dente FL, Bancalari L, Bacci E, Cianchetti S, Franco A, et al. Beclomethasone dipropionate blunts allergen‐induced early increase in urinary LTE4. European Journal of Clinical Investigation 2010;40(6):566‐9. - PubMed
Bateman 1995 {published data only}
-
- Bateman ED, Holgate ST, Binks SM, Tarns IP. A multicentre study to assess the steroid‐sparing potential of accolate (zafirlukast; 20 mg bd). Allergy 1995;50 Suppl(26):320, Abs P‐0709.
Bateman 2003 {published data only}
-
- Bateman ED, Akveld M, Ho M. Greater responder rate to fluticasone propionate/salmeterol combination over montelukast plus fluticasone in asthma [abstract]. American Thoracic Society 99th International Conference 2003;B036:H90.
Baumgartner 1999 {published data only}
-
- Baumgartner RA, Polis A, Angner R, Bird S, Reiss TF. Comparison between montelukast and inhaled beclomethasone therapy in chronic asthma: a double blind, placebo controlled, parallel study in asthmatic patients. Merck Research Laboratories 1999.
Benitez 2005 {published data only}
-
- Benitez HH, Arvizu VM, Gutierrez DJ, Fogelbach GA, Castellanos Olivares A, Vazquez Nava F, et al. Nasal budesonide plus zafirlukast vs nasal budesonide plus loratadine‐pseudoephedrine for controlling the symptoms of rhinitis and asthma. Revista Alergia Mexico 2005;52(2):90‐5. - PubMed
Berger 2006 {published data only}
-
- Berger WE, Milgrom H, Skoner DP, Tripp K, Parsey MV, Baumgartner RA. Evaluation of levalbuterol metered dose inhaler in pediatric patients with asthma: A double‐blind, randomized, placebo‐ and active‐controlled trial. Current Medical Research & Opinion 2006;22(6):1217‐26. - PubMed
Bilancia 2000 {published data only}
-
- Bilancia R, Margiotta D, Balacco D. Low doses of budesonide (B) plus montelukast (M) versus high doses of budesonide in asthmatic patients. American Journal of Respiratory and Critical Care Medicine 2000;161(supp 3):A197.
Bilderback 2004 {published data only}
-
- Bilderback A, Krishnan JA, Riekert KA, Schiller K, Rand CS. Patterns of use for oral montelukast and inhaled fluticasone in a clinical trial [Abstract]. American Thoracic Society 100th International Conference 2004;B39:C18.
Bilderback 2005 {published data only}
-
- Bilderback A, Rand C, Krishnan J, Riekert K, Schiller K, Zieger RS, et al. Adherence with montelukast or fluticasone and outcomes in a 1‐year clinical trial in patients with mild persistent asthma [Abstract]. American Thoracic Society International Conference 2005;D97:Poster 521.
Bisgaard 1999 {published data only}
-
- Bisgaard H, Loland L, Anhoj J. NO in exhaled air of asthmatic children is reduced by the leukotriene receptor antagonist montelukast. American Journal of Respiratory & Critical Care Medicine 1999;160:1227‐31. - PubMed
Bisgaard 2000 {published data only}
-
- Bisgaard H, Nielsen KG. Bronchoprotection with a leukotriene receptor antagonist in asthmatic preschool children. American Journal of Respiratory & Critical Care Medicine 2000;162(1):187‐90. - PubMed
Bisgaard 2005 {published data only}
-
- Bisgaard H, Zielen S, Garcia‐Garcia ML, Johnston SL, Gilles L, Menten J, et al. Montelukast reduces asthma exacerbations in 2‐ to 5‐year‐old children with intermittent asthma. American Journal of Respiratory & Critical Care Medicine 2005;171(1):315‐22. - PubMed
Bjermer 2002 {published data only}
-
- Bjermer L, Greening A, Haahtela T, Bousquet J, Holgate ST, Picado C, et al. IMPACT study group. Addition of montelukast or salmeterol to fluticasone in patients with uncontrolled asthma: results of the IMPACT trial.. Chest. 2002:434.
Bjermer 2003 {published data only}
Bjermer 2004 {published data only}
-
- Bjermer L, Kocevar VS, Zhang Q, Yin DD, Polos PG. Health care resource utilization following addition of montelukast or salmeterol in fluticasone in patients with inadequately controlled asthma (IMPACT trial) [Abstract]. European Respiratory Journal 2004;24(Suppl 48):127s.
Bleecker 2006 {published data only}
-
- Bleecker ER, Yancey SW, Baitinger LA, Edwards LD, Klotsman M, Anderson WH, et al. Salmeterol response is not affected by beta2‐adrenergic receptor genotype in subjects with persistent asthma. Journal of Allergy & Clinical Immunology 2006;118(4):809‐16. - PubMed
Borker 2005 {published data only}
-
- Borker R, Emmett A, Jhingran P, Rickard K, Dorinsky P. Determining economic feasibility of fluticasone propionate‐salmeterol vs montelukast in the treatment of persistent asthma using a net benefit approach and cost‐effectiveness acceptability curves. Annals of Allergy Asthma & Immunology 2005;95(2):181‐9. - PubMed
Bousquet 2005a {published data only}
-
- Bousquet J, Gaugris S, Kocevar VS, Zhang Q, Yin DD, Polos PG, et al. Increased risk of asthma attacks and emergency visits among asthma patients with allergic rhinitis: A subgroup analysis of the improving asthma control trial. Clinical & Experimental Allergy 2005;35(6):723‐7. - PubMed
Bousquet 2007 {published data only}
-
- Bousquet J, Rabe K, Humbert M, Chung KF, Berger W, Fox H, et al. Predicting and evaluating response to omalizumab in patients with severe allergic asthma. Respiratory Medicine 2007;101(7):1483‐92. - PubMed
Brannan 2001 {published data only}
-
- Brannan JD, Anderson SD, Gomes K, King GG, Chan HK, Seale JP. Fenofenadine decreases sensitivity to and montelukast improves recovery from inhaled manitol. American Journal of Respiratory & Critical Care Medicine 2001;163(6):1420‐5. - PubMed
Brocks 1996 {published data only}
-
- Brocks DR, Upward JW, Georgiou P, Stelman G, Doyle E, Allen E, et al. The single and multiple dose pharmacokinetics of pranlukast in healthy volunteers. European Journal of Clinical Pharmacology 1996;51:303‐8. - PubMed
Bronsky 1997 {published data only}
-
- Bronsky E, Grossman J, Nathan RA, Jong B. Pranlukast (Ultair) reduces symptoms of seasonal allergic rhinitis: results of the first US double‐blind, placebo controlled trial in 484 patients. SmithKline Beecham Pharmaceuticals 1997.
Brown 2005 {published data only}
-
- Brown ES, Stuard G, Liggin JDM, Hukovic N, Frol A, Dhanani N, et al. Effect of phenytoin on mood and declarative memory during prescription corticosteroid therapy. Biological Psychiatry 2005;57(5):543‐8. - PubMed
Brown 2007 {published data only}
-
- Brown ES, Frol AB, Khan DA, Larkin GL, Bret ME. Impact of levetiracetam on mood and cognition during prednisone therapy. European Psychiatry 2007;22(7):448‐52. - PubMed
Brown 2010 {published data only}
-
- Brown ES, Denniston D, Gabrielson B, Khan DA, Khanani S, Desai S. Randomized, double‐blind, placebo‐controlled trial of acetaminophen for preventing mood and memory effects of prednisone bursts. Allergy and Asthma Proceedings 2010;31(4):331‐6. - PubMed
Bruce 2002 {published data only}
-
- Bruce C, Palmqvist M, Sjöstrand M, Aronsson B, Arvidsson P, Lotvall J. Greater attenuation of the late asthmatic reaction by fluticasone propionate compared to montelukast. American Journal of Respiratory and Critical Care Medicine 2002;165(Suppl 8):A215.
Buchvald 2002 {published data only}
-
- Buchvald FF, Bisgaard H. Comparison of add‐on of leukotriene receptor antagonist vs. long‐acting beta 2‐agonist of FeNO in asthmatic children on regular inhaled budesonide. European Respiratory Journal 2002;20(Suppl 38):431s.
Buchvald 2002a {published data only}
-
- Buchvald FF, Bisgaard H. Comparison of add‐on of leukotriene receptor antagonist vs. long‐acting beta2‐agonist on FeNO in asthmatic children on regular inhaled budesonide [abstract]. European Respiratory Society Annual Congress. 2002:P2736.
Buchvald 2003 {published data only}
-
- Buchvald F, Bisgaard H. Comparisons of the complementary effect on exhaled nitric oxide of salmeterol vs montelukast in asthmatic children taking regular inhaled budesonide. Annals of Allergy Asthma & Immunology 2003;91(3):309‐13. - PubMed
Buckstein 2003 {published data only}
-
- Bukstein DA, Luskin AT, Bernstein A. "Real‐world" effectiveness of daily controller medicine in children with mild persistent asthma. Annals of Allergy Asthma & Immunology 2003;90(5):543‐9. - PubMed
Burgess 2007 {published data only}
-
- Burgess SW, Sly PD, Cooper DM, Devadason SG. Novel spacer device does not improve adherence in childhood asthma. Pediatric Pulmonology 2007;42(8):736‐9. - PubMed
Busse 1999 {published data only}
-
- Busse W, Nelson H, Wolfe J, Kalberg C, Yancey SW, Rickard KA. Comparison of inhaled salmeterol and oral zafirlukast in patients with asthma. Journal of Allergy & Clinical Immunology 1999;103:1075‐80. - PubMed
Buxton 2004 {published data only}
-
- Buxton MJ, Sullivan SD, Andersson LF, Lamm CJ, Liljas B, Busse WW, et al. Country‐specific cost‐effectiveness of early intervention with budesonide in mild asthma. European Respiratory Journal 2004;24(4):568‐74. - PubMed
Cakmak 2000 {published data only}
-
- Cakmak G, Demir T, Aydemir A, Serdaroglu E, Erginoz E, Donma O, et al. The effect of adding zafirlukast to budesonide treatment on total antoxydant capacity. European Respiratory Journal 2000;16(Supplement 31):457s.
Cakmak 2004 {published data only}
-
- Cakmak G, Demir T, Gemicioglu B, Aydemir A, Serdaroglu E, Donma O. The effects of add‐on zafirlukast treatment to budesonide on bronchial hyperresponsiveness and serum levels of eosinophilic cationic protein and total antioxidant capacity in asthmatic patients. Tohoku Journal of Experimental Medicine 2004;204(4):249‐56. - PubMed
Calhoun 1997 {published data only}
-
- Calhoun WJ, Weisberg SC, Faiferman I, Stober PW. Pranlukast (Ultair) is effective in improving asthma: results of a 12‐week, multicenter, dose‐range study. Journal of Allergy & Clinical Immunology 1997;99:S318, Abs 1305.
Calhoun 2001 {published data only}
-
- Calhoun WJ, Nelson HS, Nathan RA, Pepsin PJ, Kalberg C, Emmett A, et al. Comparison of fluticasone propionate‐salmeterol combination therapy and montelukast in patients who are symptomatic on short‐acting beta(2)‐agonists alone. American Journal of Respiratory & Critical Care Medicine 2001;164(5):759‐63. - PubMed
Calhoun 2004 {published data only}
-
- Calhoun W, Sutton L, Emmett A, Dorinsky P. Asthma control with fluticasone propionate/salmeterol 100/50µg Diskus(r) versus montelukast in patients previously receiving short‐acting beta2‐agonists [Abstract]. Journal of Allergy and Clinical Immunology 2004;113(Suppl 2):S117.
Camargo 2002 {published data only}
-
- Camargo CA, Smithline HA, Marie‐Pierre M, Green SA, Reiss TF. A randomized controlled trial of intravenous montelukast in acute asthma. American Journal of Respiratory and Critical Care Medicine. 2002. - PubMed
Camargo 2003 {published data only}
-
- Camargo CA Jr, Smithline HA, Malice MP, Green SA, Reiss TF. A randomized controlled trial of intravenous montelukast in acute asthma. American Journal of Respiratory & Critical Care Medicine 2003;167(4):528‐33. - PubMed
Canino 2008 {published data only}
Capella 2001 {published data only}
-
- Capella GL, Frigerio E, Altomare G. A randomized trial of leukotriene receptor antagonist montelukast in moderate‐to‐severe atopic dermatitis of adults. European Journal of Dermatology 2001;11(3):209‐13. - PubMed
Ceylan 2004 {published data only}
-
- Ceylan E, Mehmet G, Sahin A. Addition of formoterol or montelukast to low‐dose budesonide: An efficacy comparison in short‐ and long‐term asthma control. Respiration 2004;71(6):594‐601. - PubMed
Chan 2003 {published data only}
Chand 2005 {published data only}
-
- Chand JG, Singh M, Mathew JL. Randomized controlled trial comparing montelukast plus low dose inhaled budesonide with conventional dose inhaled budesonide with conventional dose inhaled budesonide in childhood moderate persistent asthma [Abstract]. Indian Journal of Allergy Asthma and Immunology 2005;19(2):108.
Chanez 2010 {published data only}
-
- Chanez P, Contin‐Bordes C, Garcia G, Verkindre C, Didier A, Blay F, et al. Omalizumab‐induced decrease of FcRI expression in patients with severe allergic asthma. Respiratory Medicine 2010;104(11):1608‐17. - PubMed
Chen 2006 {published data only}
-
- Chen Y‐Z, Busse WW, Pedersen S, Tan W, Lamm C‐J, O'Byrne PM. Early intervention of recent onset mild persistent asthma in children aged under 11 yrs: The Steroid Treatment As Regular Therapy in early asthma (START) trial. Pediatric Allergy & Immunology 2006;17(Suppl 17):7‐13. - PubMed
Chiba 1997 {published data only}
-
- Chiba M, Xu X, Nishime JA, Balani SK, Lin JH. Hepatic microsomal metabolism of montelukast, a potent leukotriene D4 receptor antagonist, in humans. Drug Metabolism and Disposition 1997;25(9):1022‐31. - PubMed
Choi 2003 {published data only}
-
- Choi IS, Koh YI. Effects of BCG revaccination on asthma. Allergy 2003;58(11):1114‐6. - PubMed
Chopra 2005 {published data only}
-
- Chopra N, Williams M, Rimmer M, Kahl L, Jenkins M. Salmeterol HFA is as effective as salmeterol CFC in children and adults with persistent asthma. Respiratory Medicine 2005;99(Suppl 1):S1‐S10. - PubMed
Chuchalin 2002 {published data only}
-
- Chuchalin AG, Ovcharenko SI, Goriachkina LA, Sidorenko IV, Tsoi AN, Alekseev VG, et al. The safety and efficacy of formoterol OxisRTurbuhalerR plus budesonide PulmicortRTurbuhaler in mild to moderate asthma: A comparison with budesonide Turbuhaler alone and current non‐corticosteroid therapy in Russia. International Journal of Clinical Practice 2002;56(1):15‐20. - PubMed
Chuchalin 2007 {published data only}
-
- Chuchalin AG, Tsoi AN, Richter K, Krug N, Dahl R, Luursema PB, et al. Safety and tolerability of indacaterol in asthma: A randomized, placebo‐controlled 28‐day study. Respiratory Medicine 2007;101(10):2065‐75. - PubMed
Chung 2000 {published data only}
-
- Chung HL, Lee JJ, Kim SG. Effect of theophylline on urinary leukotriene B4 and C4 excretion in children with asthma. Korean Journal of Asthma, Allergy and Clinical Immunology 2000;20(5):710‐6.
Ciebiada 2009 {published data only}
-
- Ciebiada M, Gorska‐Ciebiada M, Gorski P. Fexofenadine with either montelukast or a low‐dose inhaled corticosteroid (fluticasone) in the treatment of patients with persistent allergic rhinitis and newly diagnosed asthma. Archives of Medical Science 2009;5(4):564‐9.
Claesson 1998 {published data only}
-
- Claesson HE, Dahlen SE. Asthma and leukotrienes: antileukotrienes as novel anti‐asthmatic drugs. Journal of Internal Medicine 1999;245(3):205‐27. - PubMed
Cloud 1989 {published data only}
-
- Cloud ML, Enas GC, Kemp J, Platts‐Mills T, Altman LC, Townley R, et al. A specific LTD4‐LTE4‐receptor antagonist improves pulmonary function in patients with mild, chronic asthma. American Review of Respiratory Disease 1989;140:1336‐9. - PubMed
Covar 2008 {published data only}
Cowan 2010 {published data only}
-
- Cowan DC, Hewitt RS, Cowan JO, Palmay R, Williamson A, Lucas SJE, et al. Exercise‐induced wheeze: Fraction of exhaled nitric oxide‐directed management. Respirology 2010;15(4):683‐90. - PubMed
Currie 2003 {published data only}
Currie 2003a {published data only}
-
- Currie GP, Lee DK, Haggart K, Bates CE, Lipworth BJ. Effects of montelukast on surrogate inflammatory markers in corticosteroid‐treated patients with asthma. American Journal of Respiratory & Critical Care Medicine 2003;167(9):1232‐8. - PubMed
Currie 2003b {published data only}
-
- Currie GP, Lee DK, Haggart K, Bates CE, Lipworth BJ. Montelukast confers complimentary non‐steroid anti‐inflammatory activity in asthmatics receiving fluticasone alone and fluticasone/salmeterol combination [Abstract]. Journal of Allergy and Clinical Immunology 2003;111(Suppl 2):S146.
Cylly 2003 {published data only}
-
- Cylly A, Kara A, Ozdemir T, Ogus C, Gulkesen KH. Effects of oral montelukast on airway function in acute asthma. Respiratory Medicine 2003;97(5):533‐6. - PubMed
Dahlén 2002 {published data only}
-
- Dahlén SE, Malmstrom K, Nizankowska E, Dahlén B, Kuna P, Kowalski M, et al. Improvement of aspirin‐intolerant asthma by montelukast, a leukotriene antagonist. A randomised, double‐blind, placebo controlled trial. American Journal of Respiratory and Critical Care Medicine 2002;165(1):9‐14. - PubMed
Daikh 2003 {published data only}
-
- Daikh BE, Ryan CK, Schwartz RH. Montelukast reduces peripheral blood eosinophilia but not tissue eosinophilia or symptoms in a patient with eosinophilic gastroenteritis and esophageal stricture. Annals of Allergy, Asthma, & Immunology 2003;90(1):2327. - PubMed
Davies 2004 {published data only}
-
- Davies GM, Dasbach EJ, Santanello NC, Knorr BA, Bratton DL. The effect of montelukast versus usual care on health care resource utilization in children aged 2 to 5 years with asthma. Clinical Therapeutics 2004;26(11):1895‐1904. - PubMed
Daviskas 2007 {published data only}
-
- Daviskas E, Anderson SD, Young IH. Inhaled mannitol changes the sputum properties in asthmatics with mucus hypersecretion. Respirology 2007;12(5):683‐91. - PubMed
Dekhuijzen 2006 {published data only}
-
- Dekhuijzen PNR. Caution recommended in prescribing long‐acting beta2‐ adrenergic agonists to patients with asthma. Nederlands Tijdschrift voor Geneeskunde 2006;150(16):889‐91. - PubMed
Delaronde 2005 {published data only}
-
- Delaronde S, Peruccio DL, Bauer BJ. Improving asthma treatment in a managed care population. American Journal of Managed Care 2005;11(6):361‐8. - PubMed
Dempsey 1999 {published data only}
-
- Dempsey OJ, Wilson AM, Sims EJ, Lipworth BJ. A comparison of once daily topical budesonide (BUD) and oral montelukast (MON) in seasonal allergic rhinitis (SAR) and asthma. European Respiratory Society. 1999. - PubMed
Dempsey 2000 {published data only}
-
- Dempsey OJ, Wilson AM, Sims EJ, Mistry C, Lipworth BJ. Additive bronchoprotective and bronchodilator effects with single doses of salmeterol and montelukast in asthmatic patients receiving inhaled corticosteroids. Chest 2000;117:950‐3. - PubMed
Dempsey 2000a {published data only}
-
- Dempsey OJ, Wilson AM, Sims EJ, Lipworth BJ. Additive anti‐inflammatory effects of montelukast but not salmeterol in asthmatics suboptimally controlled on inhaled steroids [abstract]. American Journal of Respiratory and Critical Care Medicine 2000;161(3 Suppl):A198.
Dempsey 2002b {published data only}
-
- Dempsey OJ, Fowler SJ, Wilson A, Kennedy G, Lipworth BJ. Effects of adding either a leukotriene receptor antagonist or low‐dose theophylline to a low or medium dose of inhaled corticosteroid in patients with persistent asthma. Chest 2002;122(1):151‐9. - PubMed
Demuro‐Mercon 2001 {published data only}
-
- Demuro‐Mercon C, . Turpin J, Santanello N, Firriolo K, Edelman J. Montelukast improves asthma quality of life when added to fluticasone. Journal of Allergy and Clinical Immunology 2001;107(2):S248.
Dessanges 1999 {published data only}
-
- Dessanges JF, Prefaut C, Taytard A, Matran R, Naya I, Compagnon A, et al. The effect of zafirlukast on repetitive exercise‐induced bronchoconstriction: The possible role of leukotrienes in exercise‐induced refractoriness. Journal of Allergy & Clinical Immunology 1999;104(6):1155‐61. - PubMed
Deykin 2007 {published data only}
Diamant 1997 {published data only}
-
- Diamant Z, et al. Effect of oral montelukast on allergen‐induced airway responses in asthmatic subjects. American Journal of Respiratory & Critical Care Medicine 1997.
Diamant 2009 {published data only}
-
- Diamant Z, Ulrik CS. Effect of add‐on montelukast to inhaled corticosteroids on excessive airway narrowing in adult asthmatics [Abstract]. American Thoracic Society International Conference. 2009:A2416.
Dicpinigaitis 2002 {published data only}
-
- Dicpinigaitis PV, Dobkin JB, Reichel J. Antitussive effect of the leukotriene receptor antagonist zafirlukast in subjects with cough‐variant asthma. Journal of Asthma 2002;39(4):291‐7. - PubMed
Djukanovic 2010 {published data only}
-
- Djukanovic R, Wilson SJ, Moore WC, Koenig SM, Laviolette M, Bleecker ER, et al. Montelukast added to fluticasone propionate does not alter inflammation or outcomes. Respiratory Medicine 2010;104(10):1425‐35. - PubMed
Dockhorn 2000 {published data only}
Dorinsky 2001 {published data only}
-
- Dorinsky P, Kalberg C, Pepsin P, Emmett A, Bowers B, Rickard K. The fluticasone/salmeterol combination product is superior to montelukast as first‐line asthma control. European Respiratory Journal 2001;18(Suppl 33):263s.
Dorinsky 2002 {published data only}
-
- Dorinsky PM, Crim C, Yancey S, Edwards L, Rickard K. First line therapy with fluticasone propionate/salmeterol combination product provides superior asthma control versus montelukast or fluticasone propionate alone [Abstract]. Journal of Allergy Asthma and Immunolgy 2002;109(Suppl 1):Abstract No: 767.
Dorinsky 2004 {published data only}
-
- Dorinsky PM, Stauffer J, Waitkus‐Edwards K, Yancey S, Prillaman BA, Sutton L. "Stepping Down" from Fluticasone Propionate/Salmeerol 100/50mcg Diskus(R) Results in Loss of Asthma Control: Lack of Effect of Ethnic Origin [Abstract]. Chest 2004;126(Suppl 4):758S.
Edelman 2000 {published data only}
-
- Edelman JM, Ghannam A, Bird S, Dobbins T. Onset of action of montelukast and inhaled beclomethasone in achieving asthma control [abstract]. American Journal of Respiratory and Critical Care Medicine 2000;161(Suppl 3):A202.
Eliraz 2001 {published data only}
-
- Eliraz A, Raminez‐Rivera A, Ferranti P, et al. Similar efficacy following four weeks treatment of asthmatics with formoterol 12 mcg bid delivered by two different dry powder inhalers: differences in inhaler handling. International Journal of Clinical Practice 2001;55(3):164‐70. - PubMed
El Miedany 2006 {published data only}
-
- Miedany Y, Youssef S, Ahmed I, Gaafary M. Safety of etoricoxib, a specific cyclooxygenase‐2 inhibitor, in asthmatic patients with aspirin‐exacerbated respiratory disease. Annals of Allergy Asthma & Immunology 2006;97(1):105‐9. - PubMed
Ensom 2003 {published data only}
-
- Ensom MHH, Chong G, Beaudin B, Bai TR. Estradiol in severe asthma with premenstrual worsening. Annals of Pharmacotherapy 2003;37(11):1610‐3. - PubMed
Fabbri 1996 {published data only}
-
- Fabbri LM, Piattella M, Caramori G, Ciaccia A. Oral vs inhaled asthma therapy. Drugs 1996;52 Suppl(6):20‐8. - PubMed
Fagerson 2003 {published data only}
-
- Fagerspm GMH. Growth and adrenocortical function in children on high‐dose inhaled steroids versus low‐dose inhaled steroids plus montelukast. Tayside Research Consortium 2003.
Failla 2006 {published data only}
-
- Failla M, Biondi G, Provvidenza Pistorio M, Gili E, Mastruzzo C, Vancheri C, et al. Intranasal steroid reduces exhaled bronchial cysteinyl leukotrienes in allergic patients. Clinical & Experimental Allergy 2006;36(3):325‐30. - PubMed
Fardon 2004 {published data only}
-
- Fardon TC. Does Fluticasone/Salmeterol combination exhibit a putative in vivo anti‐inflammatory action during step‐down?. Tayside Research Consortium 2004.
Fardon 2007 {published data only}
-
- Fardon T, Haggart K, Lee DKC, Lipworth BJ. A proof of concept study to evaluate stepping down the dose of fluticasone in combination with salmeterol and tiotropium in severe persistent asthma. Respiratory Medicine 2007;101(6):1218‐28. - PubMed
Faul 2002 {published data only}
-
- Faul JL, Canfield JC, Gould MK, Wilson SR, Kischner WG. A randomized, double‐blind, placebo‐controlled, crossover study comparing the effects of inhaled fluticasone propionate (880 Micrograms per day) and montelukast (10 MG per day) on glucose control patients with diabetes and asthma. American Journal of Respiratory and Critical Care Medicine 2002;165(Suppl 8):A217.
Findlay 1992 {published data only}
-
- Findlay SR, Barden JM, Easley CB, Glass M. Effect of the oral leukotriene antagonist, ICI 204,219, on antigen‐induced bronchoconstriction in subjects with asthma. Journal of Allergy & Clinical Immunology 1992;89:1040‐5. - PubMed
Finkelstein 2005 {published data only}
Finn 2000 {published data only}
-
- Finn AF, Bonuccelli CM, Traxler BM, Beatty SE. Zafirlukast improves asthma control in children treated with and without inhaled corticosteroids. European Respiratory Journal 2000;16(Supp 31):307.
Fischer 1995 {published data only}
-
- Fischer AR, McFadden CA, Frantz R, Awni WM, Cohn J, Drazen JM, et al. Effect of chronic 5‐lipoxygenase inhibition on airway hyperresponsiveness in asthmatic subjects. American Journal of Respiratory & Critical Care Medicine 1995;152(4 part 1):1203‐07. - PubMed
Fischer 1997 {published data only}
Fish 1997 {published data only}
-
- Fish JE, Kemp JP, Lockey RF, Glass M, Hanby L, Bonuccelli CM. Zafirlukast for symptomatic mild‐to‐moderate asthma: a 13‐week multicenter study. Clinical Therapeutics 1997;19(4):675‐90. - PubMed
Fish 2000 {published data only}
-
- Fish J, Boone R, Emmett A, Yancey S, Knobil K, Rickard K. Salmeterol added to inhaled corticosteroids (ICS) provides greater asthma control compared to montelukast [abstract]. American Journal of Respiratory and Critical Care Medicine 2000;161(Suppl 2):A203.
Fish 2001 {published data only}
-
- Fish JE, Israel E, Murray JJ, Emmett A, Boone R, Yancey SW, et al. Salmeterol powder provides significantly better benefit than montelukast in asthmatic patients receiving concomitant inhaled corticosteroid therapy. Chest 2001;120(2):423‐30. - PubMed
FitzGerald 2009 {published data only}
Fogel 2010 {published data only}
-
- Fogel RB, Rosario N, Aristizabal G, Loeys T, Noonan G, Gaile S, et al. Effect of montelukast or salmeterol added to inhaled fluticasone on exercise‐induced bronchoconstriction in children. Annals of Allergy, Asthma and Immunology 2010;104(6):511‐7. - PubMed
Franzen 1994 {published data only}
-
- Franzen L, Kumlin M, Dahlen SE. Pharmacologic modulation of leukotriene and histamine release in the human lung. Abbott Laboratories 1994.
Fritsch 2006 {published data only}
-
- Fritsch M, Uxa S, Horak Jr F, Putschoegl B, Dehlink E, Szepfalusi Z, et al. Exhaled nitric oxide in the management of childhood asthma: A prospective 6‐months study. Pediatric Pulmonology 2006;41(9):855‐62. - PubMed
Fujimura 1993 {published data only}
-
- Fujimura M, Sakamoto S, Kamio Y, Matsuda T. Effect of a leukotriene antagonist, ONO‐1078, on bronchial hyperresponsiveness in patients with asthma. Respiratory Medicine 1993;87:133‐8. - PubMed
Gabrijelcic 2004 {published data only}
-
- Gabrijelcic J, Casas A, Rabinovich RA, Roca J, Barbera JA, Chung KF, et al. Formoterol protects against platelet‐activating factor‐induced effects in asthma. European Respiratory Journal 2004;23(1):71‐5. - PubMed
Gaddy 1990 {published data only}
-
- Gaddy J, Bush RK, Margolskee D, Williams VC, Busse W. The effects of a leukotriene D4 (LTD4) antagonist (MK‐571) in mild to moderate asthma. Journal of Allergy & Clinical Immunology 1990;85:197, Abs. 216.
Galbreath 2008 {published data only}
-
- Galbreath AD, Smith B, Wood PR, Inscore S, Forkner E, Vazquez M, et al. Assessing the value of disease management: Impact of 2 disease management strategies in an underserved asthma population. Annals of Allergy Asthma and Immunology 2008;101(6):599‐607. - PubMed
Geha 2001 {published data only}
-
- Geha RS. Desloratadine: a new, nonsedating, oral antihistamine. Journal of Allergy & Clinical Immunology 2001;107(4):751‐62. - PubMed
Gelb 2008 {published data only}
-
- Gelb AF, Karpel J, Wise RA, Cassino C, Johnson P, Conoscenti CS. Bronchodilator efficacy of the fixed combination of ipratropium and albuterol compared to albuterol alone in moderate‐to‐severe persistent asthma. Pulmonary Pharmacology and Therapeutics 2008;21(4):630‐6. - PubMed
Georgiou 1997 {published data only}
-
- Georgiou P, Compton C, Allen A, Hust R, Collie H. Pranlukast (Ultair) has no effect on cardiovascular parameters in healthy male subjects. American Thoracic Society. 1997:Abs C49.
Ghiro 2001 {published data only}
-
- Ghiro L, Zanconato S, Rampon O, Piovan V, Scollo M, Baraldi E. Effect of Montelukast added to inhaled steroids on exhaled NO in asthmatic children. European Respiratory Journal 2001;18(Suppl 33):40s. - PubMed
Ghiro 2002 {published data only}
-
- Ghiro L, Zanconato S, Rampon O, Piovan V, Pasquale MF, Baraldi E. Effect of montelukast added to inhaled corticosteroids on fractional exhaled nitric oxide in asthmatic children. European Respiratory Journal 2002;20(3):630‐4. - PubMed
Gold 2001 {published data only}
-
- Gold M, Jõgi R, Mulder PGH, Akveld MLM. Salmeterol/fluticasone propionate combination 50/100µg bid is more effective than fluticasone propionate 100µg bid plus montelukast 10 mg once daily in reducing exacerbations. European Respiratory Journal 2001;18(Supp 33):262s.
Gold 2001a {published data only}
-
- Gold M, Jõgi R, Mulder PGH, Akveld MLM. Salmeterol/fluticasone propionate combination 50/100µg bid is more effective than fluticasone propionate 100µg bid plus montelukast 10 mg once daily in reducing exacerbations. European Respiratory Journal 2001;18(Supp 33):262s.
Green 2002 {published data only}
-
- Green RH, Brightling S, Mckenna B, Hargadon B, Parker D, Wardlaw AJ, et al. A placebo controlled comparison of formoterol, montelukast or higher dose of inhaled corticosteroids in subjects with symptomatic asthma despite treatment with low dose inhaled corticosteroids. Thorax 2002;57(3):11.
Green 2002a {published data only}
-
- Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Bradding P, et al. Asthma exacerbations and sputum eosinophil counts: A randomised controlled trial. Lancet 2002;360(9347):1715‐21. - PubMed
Green 2006 {published data only}
-
- Green RH, Brightling CE, McKenna S, Hargadon B, Neale N, Parker D, et al. Comparison of asthma treatment given in addition to inhaled corticosteroids on airway inflammation and responsiveness. European Respiratory Journal 2006;27(6):1144‐51. - PubMed
Greenberger 2003 {published data only}
-
- Greenberger PA. Therapy in the management of the rhinitis/asthma complex. Allergy & Asthma Proceedings 2003;24(6):403‐7. - PubMed
Grosclaude 2003 {published data only}
-
- Grosclaude M, Cerruti JL, Delannay B, Hérent M, Spilthooren F, Desfougčres JL. A fixed combination of fluticasone and salmeterol permits better control of asthma than a beclomethasone dipropionate and montelukast combination. Allergie et immunologie 2003;35(9):356‐62. - PubMed
Grossman 1995 {published data only}
-
- Grossman J, Bronsky E, Busse W, Montanaro A, Southern L, Tinkelman D, et al. A multicenter, double‐blind, placebo‐controlled study to evaluate the safety, tolerability and clinical activity of oral, twice‐daily LTA, Pranlukast (SB 205312) in patients with mild to moderate asthma. Journal of Allergy & Clinical Immunology 1995;95(1):352, Abs 846.
Grossman 1997 {published data only}
-
- Grossman J, Faiferman I, Dubb JW, Tompson DJ, Busse W, Bronsky E, et al. Results of the first U.S. double‐blind, placebo‐controlled, multicenter clinical study in asthma with pranlukast, a novel leukotriene receptor antagonist. Journal of Asthma 1997;34(4):321‐8. - PubMed
Grzelewska‐Rzymowska 2003 {published data only}
-
- Grzelewska‐Rzymowska I, Malolepszy J, Molina M, Sladek K, Zarkovic J, Siergiejko Z. Equivalent asthma control and systemic safety of inhaled budesonide delivered via HFA‐134a or CFC propellant in a broad range of doses. Respiratory Medicine 2003;97(Suppl D):S10‐S19. - PubMed
Grzelewski 2006 {published data only}
-
- Grzelewski T, Jerzynska J, Stelmach I. Budesonide and montelukast once daily inhibits exercise induced bronchoconstriction in 6 to 18 year old children with asthma. Journal of Allergy and Clinical Immunology 2006;117((2 Suppl 1)):S93. - PubMed
Guilbert 2004 {published data only}
-
- Guilbert TW, Morgan WJ, Krawiec M, Lemanske Jr RF, Sorkness C, Szefler SJ, et al. The Prevention of Early Asthma in Kids study: Design, rationale and methods for the Childhood Asthma Research and Education network. Controlled Clinical Trials 2004;25(3):286‐310. - PubMed
Gupta 1999 {published data only}
-
- Gupta RC, Dixit R, Khilnani G, Gupta N, Joshi N, Saluja M. Comparative efficacy of budesonide and azelastine nasal spray in nasobronchial allergy. Indian Journal of Allergy and Applied Immunology 1999;13(1):11‐6.
Gupta 2007 {published data only}
-
- Gupta S, Kansal AP, Kishan J. Comparative efficacy of combination of fluticasone and salmeterol; fluticasone, salmeterol and montelukast; fluticasone, salmeterol, and levocetirizine in moderate persistent asthma: a study of 120 patients. Chest 2007;132(4):512a.
Gylfors 2005 {published data only}
-
- Gylfors P, Dahlen SE, Larsson K, Kumlin M. Bronchial responsiveness to leukotriene D4 is resistant to inhaled fluticasone propionate [Abstract]. European Respiratory Journal 2005;26(Suppl 49):3687. - PubMed
Gyllfors 2003 {published data only}
-
- Gyllfors P, Bochenek G, Overholt J, Drupka D, Kumlin M, Sheller J, et al. Biochemical and clinical evidence that aspirin‐intolerant asthmatic subjects tolerate the cyclooxygenase 2‐selective analgetic drug celecoxib. Journal of Allergy & Clinical Immunology 2003;111(5):1116‐21. - PubMed
Gyllfors 2006 {published data only}
-
- Gyllfors P, Dahlen SE, Kumlin M, Larsson K, Dahlen B. Bronchial responsiveness to leukotriene D4 is resistant to inhaled fluticasone propionate. Journal of Allergy & Clinical Immunology 2006;118(1):78‐83. - PubMed
Haahtela 1994 {published data only}
-
- Haahtela T, Jarvinen M, Kava T, Kiviranta K, Koskinen S, Lehtonen K, et al. Effects of discontinuing inhaled budesonide in patients with mild asthma. New England Journal of Medicine 1994;331:700‐5. - PubMed
Hakim 2007 {published data only}
-
- Hakim F, Vilozni D, Adler A, Livnat G, Tal A, Bentur L. The effect of montelukast on bronchial hyperreactivity in preschool children. Chest 2007;131(1):180‐6. - PubMed
Hamilton 1998 {published data only}
-
- Hamilton AL, Faiferman I, Stober P, Watson RM, O'Byrne PM. Pranlukast, a cysteinyl leukotriene receptor antagonist, attenuates allergen‐induced early and late phase bronchoconstriction and airway hyperresponsiveness in asthmatic subjects. SmithKline Beecham Pharmaceuticals 1998. - PubMed
Harmanci 2006 {published data only}
-
- Harmanci K, Bakirtas A, Turktas I, Degim T. Oral montelukast treatment of preschool‐aged children with acute asthma. Annals of Allergy Asthma & Immunology 2006;96(5):731‐5. - PubMed
Hartwig 2004 {published data only}
-
- Hartwig KC, DiNella JV, Chiappetta K, Clark MP, Ameredes BT, Calhoun WJ. Progressive improvement in airway hyperresponsiveness (AHR) over one year with fluticasone but not montelukast therapy in asthma: the OFTIRA trial [Abstract]. American Thoracic Society 100th International Conference 2004;A58:Poster K44.
Hassall 1998 {published data only}
-
- Hassell SM, Miller C, Harris A. Zafirlukast (Accolate) reduces the need for oral steroid bursts. American Journal of Respiratory & Critical Care Medicine 1998;157(3):A411.
Havlucu 2005 {published data only}
-
- Havlucu Y, Yilmaz G, Goktan C, Yorgancioglu A. Usefulness of HRCT determining the distal airway inflammation in asthma [Abstract]. European Respiratory Journal 2005;26(Suppl 49):Abstract No. 2072.
Hay 1997 {published data only}
-
- Hay DWP. Pharmacology of leukotriene receptor antagonists: more than inhibitors of bronchoconstriction. Chest 1997;111(2):35S‐45S. - PubMed
Hendeles 2004 {published data only}
-
- Hendeles L, Erb TA, Bird S, Hustard CM, Edelman JM. Post‐exercise response to albuterol after addition of montelukast or salmeterol to inhaled fluticasone [Abstract]. Journal of Allergy and Clinical Immunology 2004;113(Suppl 2):S34.
Henderson 1994 {published data only}
-
- Henderson WR. The role of leukotrienes in inflammation. Annals of Internal Medicine 1994;121(9):684‐97. - PubMed
Hernandez 2002 {published data only}
-
- Hernandez D, Namenyi M, Fiterman J, Price DB, Beeh KM, Fletcher CP, et al. Adding montelukast versus doubling the budesonide dose in persistent asthma: a subgroup analysis of the COMPACT study [abstract]. European Respiratory Society Annual Congress. 2002:P2406.
Hood 1999 {published data only}
Hothersall 2008 {published data only}
-
- Hothersall EJ, Chaudhuri R, McSharry C, Donnelly I, Lafferty J, McMahon AD, et al. Effects of atorvastatin added to inhaled corticosteroids on lung function and sputum cell counts in atopic asthma. Thorax 2008;63(12):1070‐5. - PubMed
Houghton 2004 {published data only}
-
- Houghton CM, Langley SJ, Singh SD, Holden J, Monici Preti AP, Acerbi D, et al. Comparison of bronchoprotective and bronchodilator effects of a single dose of formoterol delivered by hydrofluoroalkane and chlorofluorocarbon aerosols and dry powder in a double blind, placebo‐controlled, crossover study. British Journal of Clinical Pharmacology 2004;58(4):359‐66. - PMC - PubMed
Howland 1994 {published data only}
-
- Howland III W, Segal A, Glass M, Minkwitz MC. 6‐week therapy with the oral leukotriene‐receptor antagonist, ICI 204,219, in the treatment of asthma. Journal of Allergy & Clinical Immunology 1994;93(1 Part 2):259, Abs. 581.
Hozawa 2009 {published data only}
-
- Hozawa S, Haruta Y, Terada M, Yamakido M. Effects of the addition of beta2‐agonist tulobuterol patches to inhaled corticosteroid in patients with asthma. Allergology International 2009;58(4):509‐18. - PubMed
Hsieh 1996 {published data only}
-
- Hsieh, K‐H. Evaluation of efficacy of traditional chinese medicines in the treatment of childhood bronchial asthma: clinical trial, immunological tests and animal study. Pediatric Allergy & Immunology 1996;7:130‐40. - PubMed
Huang 2003 {published data only}
-
- Huang CJ, Wang CH, Liu WT, Yang MC, Lin HC, Yu CT, et al. Zafirlukast Improves Pulmonary Function in Patients with Moderate Persistent Asthma Receiving Regular Inhaled Steroids: A Prospective Randomized Control Study. Chang Gung Medical Journal 2003;26(8):554‐60. - PubMed
Huang 2003a {published data only}
-
- Huang CJ, Wang CH, Lin HC, Yu CT, Shiao JR, Tan CG, et al. Zafirlukast improves pulmonary function in patients with moderate persistent asthma receiving regular inhaled steroids: a randomized control study [abstract]. American Thoracic Society 99th International Conference 2003;B036:Poster H80. - PubMed
Hui K 1991 {published data only}
-
- Hui K, Barnes NC. Lung function improvement in asthma with a cysteinyl‐leukotriene receptor antagonist. Lancet 1991;337:1062‐3. - PubMed
Igde 2009 {published data only}
-
- Igde M, Anlar FY. The efficacy of montelukast monotherapy in moderate persistent asthmatic children. Iranian Journal of Allergy Asthma & Immunology 2009;8(3):169‐70. - PubMed
Ikeda 1997 {published data only}
-
- Ikeda K, Hyashi M, Obata H, Fujita H, Nakanishi T, Izumi T. Two weeks' observation of pranlukast (ONO‐1078), leukotriene receptor antagonist) by peak expiratory flow rate (PEFR) was enough to evaluate clinical efficacies in severe chronic adult asthmatics. American Thoracic Society. 1997:Abs C49.
Ilowite 2004 {published data only}
-
- Ilowite J, Webb R, Friedman B, Kerwin E, Bird SR, Hustad CM, et al. Addition of montelukast or salmeterol to fluticasone for protection against asthma attacks: a randomized, double‐blind, multicenter study. Annals of Allergy Asthma & Immunology 2004;92(6):641‐8. - PubMed
Inoue 2007 {published data only}
-
- Inoue H, Komori M, Matsumoto T, Fukuyama S, Matsumura M, Nakano T, et al. Effects of salmeterol in patients with persistent asthma receiving inhaled corticosteroid plus theophylline. Respiration 2007;74(6):611‐6. - PubMed
Irvin 2003 {published data only}
-
- Irvin C, Anthonisen N, Castro M, Holbrook J, Kaminsky DA, Lima J, et al. Effectiveness of Low‐Dose Theophylline as Add‐on Therapy in the Treatment of Asthma: The LODO Trial [Abstract]. Chest 2003;124(4):3355.
Irvin 2007 {published data only}
-
- Irvin CG, Kaminsky DA, Anthonisen NR, Castro M, Hanania NA, Holbrook JT, et al. Clinical trial of low‐dose theophylline and montelukast in patients with poorly controlled asthma. American Journal of Respiratory & Critical Care Medicine 2007;175(3):235‐42. - PubMed
Israel 1990 {published data only}
-
- Israel E, Dermarkarkian R, Rosenberg M, Sperling R, Taylor G, Rubin P, et al. The effects of a 5‐lipoxygenase inhibitor on asthma induced by cold, dry air. New England Journal of Medicine 1990;323:1740‐4. - PubMed
Israel 1992 {published data only}
-
- Israel E, Drazen J, Pearlman H, Cohn J, Rubin P. A double‐blind multicenter study of zileuton, a potent 5‐lipoxygenase (5‐LO) inhibitor versus placebo in the treatment of spontaneous asthma in adults. Journal of Allergy & Clinical Immunology 1992;89:236, Abs. 368.
Israel 1993 {published data only}
-
- Israel E, Rubin P, Kemp JP, Grossman J, Pierson W, Siegel SC, et al. The effect of inhibition of 5‐lipoxygenase by zileuton in mild‐to‐moderate asthma. Annals of Internal Medicine 1993;119(11):1059‐66. - PubMed
Israel 1996 {published data only}
-
- Israel E, Cohn J, Dube L, Drazen JM. Effect of treatment with zileuton, a 5‐lipoxygenase inhibitor, in patients with asthma. JAMA 1996;275(12):931‐6. - PubMed
Jat 2006 {published data only}
-
- Jat GC, Mathew JL, Singh M. Treatment with 400 mug of inhaled budesonide vs 200 mug of inhaled budesonide and oral montelukast in children with moderate persistent asthma: Randomized controlled trial. Annals of Allergy Asthma & Immunology 2006;97(3):397‐401. - PubMed
Jayaram 2002a {published data only}
-
- Jayaram L, Pizzichini M, Hussack P, Efthmiadis A, Goodwin S, Chaboillez S, et al. First line anti inflammatory treatment for asthma: inhaled steroid or leukotriene antagonist [Abstract]. Journal of Allergy Asthma and Immunolgy 2002;109(Suppl 1):Abstract No: 746.
Jayaram 2005a {published data only}
-
- Jayaram L, Duong M, Pizzichini MM, Pizzichini E, Kamada D, Efthimiadis A, et al. Failure of montelukast to reduce sputum eosinophilia in high‐dose corticosteroid‐dependent asthma. European Respiratory Journal 2005;25(1):41‐6. - PubMed
Jayaram 2006 {published data only}
-
- Jayaram L, Pizzichini MM, Cook RJ, Boulet LP, Lemiere C, Pizzichini E, et al. Determining asthma treatment by monitoring sputum cell counts: Effect on exacerbations. European Respiratory Journal 2006;27(3):483‐94. - PubMed
Johnson 1999 {unpublished data only}
-
- Johnson MC, Srebro S, Edwards L, Bowers B, Rickard K. Physician‐ and patient‐rated assessments correlate well with clinical efficacy measurements in a study comparing fluticasone and zafirlukast. Journal of Allergy & Clinical Immunology. 1999; Vol. 103, issue 1 Part 2:Abs. 882.
Johnston 2007 {published data only}
-
- Johnston NW, Mandhane PJ, Dai J, Duncan JM, Greene JM, Lambert K, et al. Attenuation of the September epidemic of asthma exacerbations in children: A randomized, controlled trial of montelukast added to usual therapy. Pediatrics 2007;120(3):e702‐e712. - PubMed
Jones 2002 {published data only}
-
- Jones C, Allen FC, Duong PT, Sajjan S, Nelsen L, Itzler R, et al. Similar health care outcomes for patients treated with fluticasone propionate (FP) and montelukast sodium (MON) monotherapy [Abstract]. Journal of Allergy Asthma and Immunolgy 2002;109(Suppl 1):Abstract No: 890.
Jonsson 2004 {published data only}
-
- Jonsson B, Berggren F, Svensson K, O'Byrne PM. An economic evaluation of combination treatment with budesonide and formoterol in patients with mild‐to‐moderate persistent asthma. Respiratory Medicine 2004;98(11):1146‐54. - PubMed
Juniper 1995 {published data only}
-
- Juniper EF, Dube L, Swanson LJ, Zileuton Study Group. The effect of zileuton, a 5‐lipoxygenase inhibitor, on asthma quality of life. Asthma. 1995.
Kalberg 1999 {published data only}
-
- Kalberg CJ, Yancey S, Emmett AH, Rickard K. A comparison of salmeterol versus zafirlukast in patients using inhaled corticosteroids. Journal of Allergy & Clinical Immunology 1999; Vol. 103, issue 1 part 2:abs 881.
Kanazawa 2004 {published data only}
-
- Kanazawa H, Yoshikawa T, Hirata K, Yoshikawa J. Effects of pranlukast administration on vascular endothelial growth factor levels in asthmatic patients. Chest 2004;125(5):1700‐05. - PubMed
Kane 1994 {published data only}
-
- Kane G, Pollice M, Tolino M, Cohn J, Dworski R, Murray J, et al. Effect of zileuton on eosinophil influx and leukotrienes in humans undergoing segmental antigen challenge. Abbott Laboratories 1994.
Kanniess 2002a {published and unpublished data}
-
- Kanniess F, Richter K, Janicki S, Schleiss MB, Jorres RA, Magnussen H. Dose reduction of inhaled corticosteroids under concomitant medication with montelukast in patients with asthma. European Respiratory Journal 2002;20:1080‐7. - PubMed
Karaman 2007 {published data only}
-
- Karaman O, Arli O, Uzuner N, Islekel H, Babayigit A, Olmez D, et al. The effectiveness of asthma therapy alternatives and evaluating the effectivity of asthma therapy by interleukin‐13 and interferon gamma levels in children. Allergy & Asthma Proceedings 2007;28(2):204‐9. - PubMed
Karpel 2007 {published data only}
-
- Karpel JP, Nayak A, Lumry W, Craig TJ, Kerwin E, Fish JE, et al. Inhaled mometasone furoate reduces oral prednisone usage and improves lung function in severe persistent asthma. Respiratory Medicine 2007;101(3):628‐37. - PubMed
Katial 2010 {published data only}
-
- Katial RK, Oppenheimer JJ, Ostrom NK, Mosnaim GS, Yancey SW, Waitkus‐Edwards KR, et al. Adding montelukast to fluticasone propionate/salmeterol for control of asthma and seasonal allergic rhinitis. Allergy and Asthma Proceedings 2010;31(1):68‐75. - PubMed
Keith 2009 {published data only}
-
- Keith PK, Koch C, Djandji M, Bouchard J, Psaradellis E, Sampalis JS, et al. Montelukast as add‐on therapy with inhaled corticosteroids alone or inhaled corticosteroids and long‐acting beta‐2‐agonists in the management of patients diagnosed with asthma and concurrent allergic rhinitis (the RADAR trial). Canadian Respiratory Journal 2009;16(Suppl A):17A‐31A. - PMC - PubMed
Kemp 1995 {published data only}
-
- Kemp JP, Glass M, Minkwitz MC. Onset of action of the leukotriene‐receptor antagonist, zafirlukast (Accolate), in patients with asthma. Journal of Allergy & Clinical Immunology 1995;95(1 Part 2):351, Abs. 844.
Kemp 1996 {published data only}
-
- Kemp JP. Zafirlukast, a selective leukotriene D4‐receptor antagonist: a new class of therapy for patients with asthma. Today's Therapeutic Trends 1996;14(2):89‐102.
Kemp 1998 {published data only}
-
- Kemp JP, Tinkelman D, Sublett J, Compton C, Georgiou P. Pranlukast (Ultair) pharmacokinetics in children consistent with that of adults. American Journal of Respiratory & Critical Care Medicine 1998;157(3):A411.
Kemp 1998a {published data only}
-
- Kemp JP, Dockhorn RJ, Shapiro GG, Nguyen HH, Reiss TF, Seidenberg BC, et al. Montelukast once daily inhibits exercise induced bronchoconstriction in 6‐14 year old children with asthma. The Journal of Pediatrics 1998;133(3):424‐28. - PubMed
Kemp 1999 {published data only}
-
- Kemp JP, Minkwitz MC, Bonuccelli CM, Warren MS. Therapeutic effect of zafirlukast as monotherapy in steroid‐naive patients with severe persistent asthma. Chest 1999;115(2):336‐42. - PubMed
Ketchell 2002 {published data only}
Khayyal 2003 {published data only}
-
- Khayyal MT, el‐Ghazaly MA, el‐Khatib AS, Hatem AM, Vries PJ, el‐Shafei S, et al. A clinical pharmacological study of the potential beneficial effects of a propolis food product as an adjuvant in asthmatic patients. Fundamental & Clinical Pharmacology 2003;17(1):93‐102. - PubMed
Kippelen 2010 {published data only}
-
- Kippelen P, Larsson J, Anderson SD, Brannan JD, Delin I, Dahlen B, et al. Acute effects of beclomethasone on hyperpnea‐induced bronchoconstriction. Medicine & Science in Sports & Exercise 2010;42(2):273‐80. - PubMed
Kips 1991 {published data only}
-
- Kips JC, Joos GF, Lepeleire I, Margolskee DJ, Buntinx A, Pauwels RA, et al. MK‐571, a potent antagonist of leukotriene D4‐induced bronchoconstriction in the human. American Review of Respiratory Disease 1991;144:617‐21. - PubMed
Knorr 1998 {published data only}
-
- Knorr B, Matz J, Bernstein JA, Nguyen H, Seidenberg BC, Reiss TF, et al. Montelukast for chronic asthma in 6‐ to 14‐year‐old children. JAMA 1998;279(15):1181‐6. - PubMed
Knorr 1999 {published data only}
-
- Knorr B, Nguycn HH, Seidenberg BC, Reiss TF, Montelukast Pediatric Study Group. Montelukast, a leukotriene receptor antagonist, provides additional clinical benefit in asthmatic children aged 6 to 14 years using inhaled corticosteroids. European Respiratory Society. 1999:P363.
Koenig 2004 {published data only}
-
- Koenig S, Waitkus‐Edwards K, Yancey S, Prillman B, Dorinsky P. Loss of asthma control when patients receiving fluticasone propionate/salmeterol 100/50µg Diskus are "stepped‐down" to fluticasone propionate , salmeterol or montelukast alone [Abstract]. Journal of Allergy and Clinical Immunology 2004;113(Suppl 2):S94.
Kohrogi 1997 {published data only}
-
- Kohrogi H, Iwagoe H, Fujii K, Fukuda K, Kawano O, Hamamoto J, et al. The effect of leukotriene antagonist pranlukast on moderate and severe persistent asthma continues more than one year. American Journal of Respiratory & Critical Care Medicine 1997;155:A662.
Kondo 2006 {published data only}
-
- Kondo N, Katsunuma T, Odajima Y, Morikawa A. A randomized open‐label comparative study of montelukast versus theophylline added to inhaled corticosteroid in asthmatic children. Allergology International 2006;55(3):287‐93. - PubMed
Kooi 2006 {published data only}
-
- Kooi EMW, Schokker S, Boezen HM, Vries TW, Vanssen‐Verberne AAP, Molen T, et al. Effect of fluticasone and montelukast in preschool children with asthma like symptoms [Abstract]. European Respiratory Journal 2006;28(Suppl 50):709s [P4079].
Korenblat 1998 {published data only}
-
- Korenblat P, Chervinsky P, Wenzel S, Faiferman I, Bakst A. Pranlukast (Ultair) reduces health care utilization and improves quality of life in adult patients with mild‐to‐moderate asthma. American Journal of Respiratory & Critical Care Medicine. 1998; Vol. 157, issue 3:A411.
Kuna 1997 {published data only}
-
- Kuna P, Malmstrom K, Dahlen SE, Nizankowska E, Kowalski M, Stevenson D, et al. Montelukast (MK‐0476), a cys‐LT1 receptor antagonist, improves asthma control in aspirin‐intolerant asthmatic patients. American Journal of Respiratory & Critical Care Medicine 1997;155(4):A975.
Kylstra 1998 {published data only}
-
- Kylstra JW, Sweitzer DE, Miller CJ, Bonuccelli CM. Zafirlukast (Accolate) in moderate asthma: patient‐reported outcomes and peripheral eosinophil data from a 13‐week trial. American Journal of Respiratory & Critical Care Medicine 1998;157(3):A410.
Laitinen 1995 {published data only}
-
- Laitinen LA, Zetterstrom O, Holgate ST, Binks SM, Whitney JG. Effects of Accolate (Zafirlukast; 20 mg bd) in permitting reduced therapy with inhaled steroids: a multicenter trial in patients with doses of inhaled steroid optimised between 800 and 2000 mcg per day. Allergy 1995;50 Suppl(26):320, Abs. P‐0710.
Leaker 2010 {published data only}
Lee 2004 {published data only}
-
- Lee DKC, Jackson CM, Haggart K, Lipworth BJ. Repeated dosing effects of mediator antagonists in inhaled corticosteroid‐treated atopic asthmatic patients. Chest 2004;125(4):1372‐7. - PubMed
Lee 2004a {published data only}
-
- Lee DK, Haggart K, Currie GP, Bates CE, Lipworth BJ. Effects of hydrofluoroalkane formulations of ciclesonide 400 microg once daily vs fluticasone 250 microg twice daily on methacholine hyper‐responsiveness in mild‐to‐moderate persistent asthma. British Journal of Clinical Pharmacology 2004;58(1):26‐33. - PMC - PubMed
Lee 2004b {published data only}
-
- Lee DKC, Haggart K, Robb FM, Lipworth BJ. Montelukast protects against nasal lysine‐aspirin challenge in patients with aspirin‐induced asthma. European Respiratory Journal 2004;24(2):226‐30. - PubMed
Lee 2004c {published data only}
-
- Lee DKC, Lipworth BJ. Anti‐inflammatory effects of histamine H1‐receptor antagonist and leukotriene CysLT1‐receptor antagonist in patients with atopic asthma maintained on inhaled corticosteroids [Abstract]. European Respiratory Journal 2004;24(Suppl 48):221s.
Lee 2005 {published data only}
-
- Lee DK, Fardon TC, Bates CE, Haggart K, McFarlane LC, Lipworth BJ. Airway and systemic effects of hydrofluoroalkane formulations of high‐dose ciclesonide and fluticasone in moderate persistent asthma. Chest 2005;127(3):851‐60. - PubMed
Lee 2010 {published data only}
-
- Lee RU, White AA, Ding D, Dursun AB, Woessner KM, Simon RA, et al. Use of intranasal ketorolac and modified oral aspirin challenge for desensitization of aspirin‐exacerbated respiratory disease. Annals of Allergy, Asthma and Immunology 2010;105(2):130‐5. - PubMed
Leff 1998 {unpublished data only}
-
- Leff JA, Busse WW, Pearlman D, Bronsky EA, Kemp J, Hendeles L, et al. Montelukast, a leukotriene‐receptor antagonist, for the treatment of mild asthma and exercise‐induced bronchoconstriction. New England Journal of Medicine 1998;339:147‐52. - PubMed
Leibman 2002 {published data only}
-
- Leibman C, Pathak D, Bowers B, Dorinsky PM, Pepsin P, Kalberg C, et al. Cost effectiveness analysis of fluticasone propionate/salmeterol combination versus montelukast in the treatment of adults with asthma [Abstract]. Journal of Allergy Asthma and Immunoly 2002;109(Suppl 1):Abstract No: 549.
Leibman 2002a {published data only}
-
- Leibman CW, Stanford R, Emmett A, Dorinsky PM, Rickard KA. Cost‐effectiveness of fluticasone propionate‐salmeterol combination versus fluticasone + montelukast in the treatment of persistent asthma. American Journal of Respiratory and Critical Care Medicine 2002;165(Suppl 8):B4.
Leigh 2002 {published data only}
-
- Leigh R, Vethanayagam D, Yoshida M, Watson RM, Rerecich T, Inman MD, et al. Effects of montelukast and budesonide on airway responses and airway inflammation in asthma. American Journal of Respiratory and Critical Care Medicine 2002;166(9):1212‐7. - PubMed
Leigh 2002a {published data only}
-
- Leigh R, Vethanayagam D, Yoshida M, Watson RM, Rererich T, Killian K, et al. Effects of montelukast and budesonide alone or in combination on allergen induced early and late asthmatic responses, and post‐allergen airway hyperresponsiveness [abstract]. American Journal of Respiratory and Critical Care Medicine 2002;165(8 Suppl):A216.
Lemanske 2010 {published data only}
Li 2001 {published data only}
-
- Li J, Zhang DH, Yang JY. Evaluation of the therapeutic effects of zafirlukast in asthma patients during reduction of high‐doses inhaled corticosteroid. Journal of Clinical Lung Section 2001;6(4):9‐11.
Liebke 2001 {published data only}
-
- Liebke C, Sommerfeld C, Wahn U, Niggemann B. [Preventive monotherapy with montelukast versus DNCG in children with mild asthma ‐ Results of an explorative pilot study]. Article in German. Pneumologie 2001;55(5):231‐7. - PubMed
Lindemann 2009 {published data only}
-
- Lindemann J, Pampe ED, Peterkin JJ, Orozco‐Cronin P, Belofsky G, Stull D. Clinical study of the effects on asthma‐related QOL and asthma management of a medical food in adult asthma patients. Current Medical Research and Opinion 2009;25(12):2865‐75. - PubMed
Lipworth 1999 {published data only}
-
- Lipworth BJ. Comparative potency and anti‐inflammatory profile of monotherapy with either montelukast or zafirlukast, patients with mild‐moderate asthma. Academic Publications (Ongoing trial) 1999.
Lis 2001 {published data only}
-
- Lis G, Cichocka Jarosz E, Glodzik I, Szczerbinski T, Bialoruska B. Montelukast in treatment mild chronic asthma. Pol Pneumonologia i Alergologia Polska 2001;69(5‐6):257‐64. - PubMed
Liu 1996 {published data only}
-
- Liu MC, Dube LM, Lancaster J, Zileuton Study Group. Acute and chronic effects of a 5‐lipoxygenase inhibitor in asthma: a 6‐month randomized multicenter trial. Journal of Allergy & Clinical Immunology 1996;98:859‐71. - PubMed
Lizaso 2003 {published data only}
-
- Lizaso Bacaicoa M, Garcia B, Gomez B, Zabalegui A, Rodriguez M, Tabar A. Treatment of allergy to mushrooms. Anales Del Sistema Sanitario De Navarra 2003;26(Suppl 2):129‐37. - PubMed
Lockey 1995 {published data only}
-
- Lockey RF, Lavins BJ, Snader L. Effects of 13 weeks of treatment with ICI 204,219 (Accolate) in patients with mild to moderate asthma. Journal of Allergy & Clinical Immunology 1995;95(Part 2):350, Abs. 839.
Lofdahl 1999 {published data only}
Luppo 2005 {published data only}
-
- Luppo A, Haag J, Maschio L, Alzua E, Nine LF, Lazaroni A. Evaluation of the variation of the PC 20 in patients treated with zafirlukast. Prensa Medica Argentina 2005;92(8):548‐9.
Lyseng‐Williamson 2003 {published data only}
-
- Lyseng‐Williamson KA. Plosker GL. Inhaled salmeterol/fluticasone propionate combination: A pharmacoeconomic review of its use in the management of asthma. Pharmacoeconomics 2003;21(13):951‐89. - PubMed
Macfarlane 2000 {published data only}
-
- Macfarlane AJ, Ying S, Smith SJ, Mannings P, Ryan M, Pavord I, et al. Inhibition of allergen‐induced late phase reactions by zafirlukast and beclomethasone may be mediated by attenuation of eotaxin‐ and rantes‐ mediated eosinophil recruitment. American Journal of Respiratory and Critical Care Medicine 2000;161(Suppl 3):A834.
Magnussen 2008 {published data only}
-
- Magnussen H, Bugnas B, Noord J, Schmidt P, Gerken F, Kesten S. Improvements with tiotropium in COPD patients with concomitant asthma. Respiratory Medicine 2008;102(1):50‐6. - PubMed
Majak 2010 {published data only}
-
- Majak P, Rychlik B, Pulaski L, Blauz A, Agnieszka B, Bobrowska‐Korzeniowska M, et al. Montelukast treatment may alter the early efficacy of immunotherapy in children with asthma. Journal of Allergy and Clinical Immunology 2010;125(6):1220‐7. - PubMed
Malerba 2002 {published data only}
-
- Malerba M, Radaeli A, Ceriani L, Amato M, Tomenzoli D, Nicolai P, et al. Comparison of oral montelukast and inhaled fluticasone in the treatment of asthma associated with chronic rhinopolyposis: A single‐blind, randomized, pilot study. Current Therapeutic Research, Clinical & Experimental 2002;63(6):355‐65.
Marchese 1998 {published data only}
-
- Marchese A, McHugh C, Kehler J, Bi H. Determination of pranlukast and its metabolites in human plasma by LC/MS/MS with PROSPEKT on‐line solid‐phase extraction. SmithKline Beecham Pharmaceuticals 1998. - PubMed
Margolskee 1991 {published data only}
-
- Margolskee D, Bodman S, Dockhorn R, Irael E, Kemp J, Mansmann H, et al. The therapeutic effects of MK‐571, a potent and selective leukotriene (LT) D4 receptor antagonist, in patients with chronic asthma. Journal of Allergy & Clinical Immunology 1991;87(1 Part 2):309, Abs. 677.
Marogna 2010 {published data only}
-
- Marogna M, Colombo F, Spadolini I, Massolo A, Berra D, Zanon P, et al. Randomized open comparison of montelukast and sublingual immunotherapy as add‐on treatment in moderate persistent asthma due to birch pollen. Journal of Investigational Allergology & Clinical Immunology 2010;20(2):146‐52. - PubMed
Maspero 2008 {published data only}
-
- Maspero J, Guerra F, Orozco S, Soto M, Gutierrez‐Schwanhauser J, Mechali DG, et al. The efficacy of inhaled salmeterol/fluticasone propionate diskus/accuhaler compared with oral montelukast in children with persistent asthma PEACE (PEdiatric Asthma Control Evaluation ) [Abstract]. American Thoracic Society International Conference. 2008:A709.
Maspero 2008a {published data only}
-
- Maspero J, Soto‐Ramos M, Guerra F, Chan R, Sharma R, Pedersen S. Improved asthma control and fewer exacerbations with inhaled salmeterol/fluticasone propionate compared with oral montelukast in children with persistent asthma: PEACE (Pediatric Asthma Control Evaluation) [Abstract]. Chest 2008;134(4):51001s.
Maspero 2008b {published data only}
-
- Maspero J, Guerra F, Cuevas F, Gutierrez JP, Soto‐Ramos M, Anderton S, et al. Efficacy and tolerability of salmeterol/fluticasone propionate versus montelukast in childhood asthma: A prospective, randomized, double‐blind, double‐dummy, parallel‐group study. Clinical Therapeutics 2008;30(8):1492‐504. - PubMed
Mastruzzo 2010 {published data only}
-
- Mastruzzo C, Contrafatto MR, Crimi C, Palermo F, Vancheri C, Crimi N. Acute additive effect of montelukast and beclomethasone on AMP induced bronchoconstriction. Respiratory Medicine 2010;104(10):1417‐24. - PubMed
Matsunaga 2004 {published data only}
-
- Matsunaga K, Nishimoto T, Hirano T, Nakanishi M, Yamagata T, Minakata Y, et al. Effect of a leukotriene receptor antagonist on the prevention of recurrent asthma attacks after an emergency room visit. Allergology International 2004;53(4):341‐7.
McCarthy 2003 {published data only}
-
- McCarthy TP, Woodcock AA, Pavord ID, Allen DJ, Parker D, Rice L. A comparison of the anti‐inflammatory and clinical effects of salmeterol 25mcg/fluticasone propionate 50mcg combination (SFC 50) with fluticasone propionate (FP) plus montelukast (M) in patients with mild to moderate asthma [abstract]. American Thoracic Society 99th International Conference 2003;B036:H89.
McGill 1996 {published data only}
-
- McGill K, Busse WW. Zileuton. Lancet 1996;348:519‐24. - PubMed
Mclvor 2009 {published data only}
Mehuys 2008 {published data only}
-
- Mehuys E, Bortel L, Bolle L, Tongelen I, Remon JP, Annemans L, et al. Does pharmacist intervention lead to appropriate use of asthma medication and improved asthma control?. Farmaceutisch Tijdschrift Voor Belgie 2008;85(1):1‐9.
Mendes 2004 {published data only}
-
- Mendes ES, Campos MA, Hurtado A, Wanner A. Effect of montelukast and fluticasone propionate on airway mucosal blood flow in asthma. American Journal of Respiratory & Critical Care Medicine 2004;169(10):1131‐4. - PubMed
Mendes 2004a {published data only}
-
- Mendes ES, Campos MA, Hurtado A, Wanner A. Anti‐inflammatory actions of montelukast and fluticasone propionate as assessed by airway blood flow [Abstract]. American Thoracic Society 100th International Conference 2004;D31:Poster C6.
Menendez 2001 {published data only}
-
- Menendez R, Standford RH, Edwards L, Kalberg C, Rickard K. Cost‐efficacy analysis of fluticasone propionate versus zafirlukast in patients with persistent asthma. Pharmacoeconomics 2001;19(8):865‐74. - PubMed
Meyer 2003 {published data only}
-
- Meyer KA, Arduino JM, Santanello NC, Knorr BA, Bisgaard H. Response to montelukast among subgroups of children aged 2 to 14 years with asthma. Journal of Allergy & Clinical Immunology 2003;111(4):757‐62. - PubMed
Micheletto 1997 {published data only}
-
- Micheletto C, Turco P, Dal Negro R. Accolate 20 mg works as steroid sparing in moderate asthma. American Journal of Respiratory & Critical Care Medicine 1997;155(4):A664.
Miraglia 2007 {published data only}
-
- Miraglia del Giudice M, Piacentini GL, Capasso M, Capristo C, Maiello N, Boner AL, et al. Formoterol, montelukast, and budesonide in asthmatic children: Effect on lung function and exhaled nitric oxide. Respiratory Medicine 2007;101(8):1809‐13. - PubMed
Mitchell 2005 {published data only}
-
- Mitchell EA, Didsbury PB, Kruithof N, Robinson E, Milmine M, Barry M, et al. A randomized controlled trial of an asthma clinical pathway for children in general practice. Acta Paediatrica 2005;94(2):226‐33. - PubMed
Miyamoto 1999 {published data only}
-
- Miyamoto T, Accolate™ clinical trial committee. Effects of zafirlukast on symptoms and pulmonary function of asthmatic patients with and without corticosteroids. European Respiratory Society. 1999:P835.
Molitor 2005 {published data only}
-
- Molitor S, Liefring E, Traytmann M. Asthma control with the salmeterol‐fluticasone‐combination disc compared to standard treatment. Pneumologie 2005;59(3):167‐73. - PubMed
Montani 2007 {published data only}
-
- Montani D. Randomized comparison of strategies for reducing treatment in mild persistent asthma. Revue De Pneumologie Clinique 2007;63(6):390.
Moreira 2008 {published data only}
-
- Moreira A, Delgado L, Haahtela T, Fonseca J, Moreira P, Lopes C, et al. Physical training does not increase allergic inflammation in asthmatic children. European Respiratory Journal 2008;32(6):1570‐5. - PubMed
Morris 2010 {published data only}
-
- Morris CR, Becker AB, Pineiro A, Massaad R, Green SA, Smugar SS, et al. A randomized, placebo‐controlled study of intravenous montelukast in children with acute asthma. Annals of Allergy, Asthma and Immunology 2010;104(2):161‐71. - PubMed
Mosnaim 2002 {published data only}
-
- Mosnaim GS. The role of montelukast in the prevention of acute episodes of asthma in children ages 2 to 14 years [Dissertation]. Rush University 2002:75.
Mosnaim 2008 {published data only}
-
- Mosnaim GS, Cohen MS, Rhoads CH, Rittner SS, Powell LH. Use of MP3 players to increase asthma knowledge in inner‐city African‐American adolescents. International Journal of Behavioral Medicine 2008;15(4):341‐6. - PubMed
Murphy 2006 {published data only}
-
- Murphy K, Ververeli K, Harvey BM, Duke AL, Chapas‐Crilly J, Uryniak T, et al. Antibody response after varicella vaccination in children treated with budesonide inhalation suspension or non‐steroidal conventional asthma therapy. International Journal of Clinical Practice 2006;60(12):1548‐57. - PubMed
Najberg 2008 {published data only}
-
- Najberg E, Smorczewska‐Kiljan A, Ksiayk J, Kaczmarewicz E, Pludowski P, Jaworski M, et al. Effect of one year treatment with inhaled budesonide on bone density and mineralization in children with bronchial asthma. Alergia Astma Immunologia 2008;13(4):217‐26.
Nakagawa 1992 {published data only}
-
- Nakagawa N, Obata T, Kobayashi T, Okada Y, Nambu F, Terawaki T, et al. In vivo pharmacologic profile of ONO‐1078: a potent, selective and orally active peptide leukotriene (LT) antagonist. Japanese Journal of Pharmacology 1992;60(3):217‐25. - PubMed
Nakajima 2001 {published data only}
-
- Nakajima S, Tohda Y, Fujimura M, Taniguchi H, Takagi K, Igarashi T, et al. Effects of montelukast on tapering inhaled corticosteroids in patients with asthma. European Respiratory Journal 2001;18(Suppl 33):260s.
Nakazono 2004 {published data only}
-
- Nakazono H, Mukoyama T, Shinomiya N. The effects of the leukotriene receptor antagonist, pranlukast, on long‐term treatment in children with persistent asthma. Journal of the Medical Society of Toho University 2004;51(6):339‐46.
Nathan 1998 {published data only}
-
- Nathan RA, Bernstein JA, Bielory L, Bonuccelli CM, Calhoun WJ, Galant SP, et al. Zafirlukast improves asthma symptoms and quality of life in patients with moderate reversible airflow obstruction. Journal of Allergy & Clinical Immunology 1998;102:935‐42. - PubMed
Nathan 2000 {published data only}
-
- Nathan BA, Boone RI, Emmett AH, Knobil K, Yancey SW, Rickard KA. Salmeterol and inhaled corticosteroids provide greater asthma control than montelukast and inhaled corticosteroids [Abstract]. Chest 2000;118(Suppl 4):85S.
Nathan 2004 {published data only}
-
- Nathan RA, Philpot E, Faris M, Prillaman B, Yancey S, Dorinsky P. In patients taking fluticasone propionate/salmeterol 100/50µg Diskus(r) for asthma, the addition of fluticasone propionate nasal spray 200µg QD to treat concomitant allergic rhinitis has a safety profile comparable to the addition of montelukast 10mg QD or placebo [Abstract]. Journal of Allergy and Clinical Immunology 2004;113(Suppl 2):S202.
Nathan 2005 {published data only}
-
- Nathan RA, Yancey SW, Waitkus‐Edwards K, Prillaman BA, Stauffer JL, Philpot E, et al. Fluticasone propionate nasal spray is superior to montelukast for allergic rhinitis while neither affects overall asthma control. Chest 2005;128(4):1910‐20. - PubMed
Nayak 1998 {published data only}
-
- Nayak AS, Anderson P, Charous BL, Williams K, Simonson S. Equivalence of adding Zafirlukast versus double‐dose inhaled corticosteroids in asthmatic patients symptomatic on low‐dose inhaled corticosteroids. Journal of Allergy & Clinical Immunology 1998;101(1 pt 2):S233, Abs. 965.
NCT00096954 {unpublished data only}
-
- A Study to Evaluate the Efficacy of Xolair in Atopic Asthmatics (EXACT). ClinicalTrials.Gov 2004.
NCT00140881 {unpublished data only}
-
- A Study to Determine the Effect of Montelukast Sodium as an Episode Modifier in the Treatment of Infrequent Episodic Asthma in Children. ClinicalTrials.Gov.
NCT00196547 {unpublished data only}
-
- Montelukast in Modulating Exacerbations of Asthma in Children. ClinicalTrials.Gov 2005.
NCT00213252 {unpublished data only}
-
- Use of Montelukast to Treat Children With Mild to Moderate Acute Asthma. ClinicalTrials.Gov 2005.
NCT00299065 {unpublished data only}
-
- Safety Study of Zileuton Injection in Patients With Asthma. ClinicalTrials.Gov 2006.
NCT00319488 {unpublished data only}
-
- Childhood Asthma Research and Education (CARE) Network Trial ‐ Acute Intervention Management Strategies (AIMS). ClinicalTrial.Gov 2006.
NCT00395408 {unpublished data only}
-
- Linear Growth Study. ClinicalTrials.Gov 2006.
NCT00421018 {unpublished data only}
-
- Optimization of Asthma Treatment Through Exhaled NO for Increased Asthma‐Related Quality of Life (NOAK). ClinicalTrials.gov 2007.
NCT00462592 {unpublished data only}
-
- Comparison of Combination Therapy: Montelukast and Inhaled Steroid on Exercise Induced Bronchoconstriction. ClinicalTrials.Gov 2007.
NCT00471809 {unpublished data only}
-
- Childhood Asthma Research and Education (CARE) Network Trial ‐ Montelukast or Azithromycin for Reduction of Inhaled Corticosteroids in Childhood Asthma (MARS). ClinicalTrials.Gov 2007.
NCT00486343 {unpublished data only}
-
- Zileuton CR vs Placebo in Poorly Controlled Asthma Patients on Moderate Dose ICS. ClinicalTrials.Gov 2007.
NCT00504946 {unpublished data only}
-
- Pharmacological Modulations of Allergen‐Specific Immunotherapy. ClinicalTrials.Gov 2007.
NCT00545324 {unpublished data only}
-
- First Step With Singulair® Therapy (FIRST). ClinicalTrials.Gov 2007.
NCT00545844 {unpublished data only}
-
- Singulair(R) In Asthma And Allergic Rhinitis. ClinicalTrials.Gov 2007.
NCT00575861 {unpublished data only}
-
- Zileuton and Exhaled Nitric Oxide in Asthmatics. ClinicalTrials.Gov 2007.
NCT00666679 {unpublished data only}
-
- Study of Inhaled Corticosteroid Plus Montelukast Compared With Inhaled Corticosteroid Therapy Alone in Patients With Chronic Asthma. ClinicalTrials.Gov 2008.
NCT00699062 {unpublished data only}
-
- Effect of Montelukast on the Airway Remodeling. ClinicalTrials.Gov 2008.
NCT00755794 {unpublished data only}
-
- The Singulair® Add‐on Study Effectiveness of Adding Montelukast to Inhaled Corticosteroids in Adult Subjects With Uncontrolled Asthma (SAS). ClincalTrials.Gov 2008.
NCT00756418 {unpublished data only}
-
- Montelukast Post‐Marketing Comparative Study With Theophyline Added to Inhaled Corticosteroid. ClinicalTrials.Gov 2008.
NCT00913328 {unpublished data only}
-
- Effect of Add‐on Montelukast to Inhaled Corticosteroids on Airway Responsiveness (SINGDEN). ClinicalTrials.Gov 2009.
NCT00943397 {unpublished data only}
-
- Effect of Add‐on Montelukast to Inhaled Corticosteroids on Airway Responsiveness (SINGDEN). ClinicalTrials.Gov 2009.
NCT01055041 {unpublished data only}
-
- Controller Medications in the Management of Bronchial Asthma. ClinicalTrials.Gov 2010.
NCT01241084 {unpublished data only}
-
- Beneficial Effects of Lactobacillus Reuteri DSM 17938 Supplementation on Asthmatic Children. ClinicalTrials.Gov 2010.
Negro 1997 {published data only}
-
- Negro JM, Miralles JC, Ortiz JL, Funes E, Garcia A. Leukotrienes and its antagonists in allergic disorders. Allergologia et Immunopathologia 1997;25(2):104‐12. - PubMed
Neki 2006 {published data only}
-
- Neki NS, Kazal HL. Comparative efficacy and safety profile of montelukast v/s beclomethasone in mild persistent asthma [Abstract]. Indian Journal of Allergy Asthma and Immunology 2006;20(2):124.
Nelson 2001 {published data only}
-
- Nelson HS, Nathan RA, Kalberg C, Yancey SW, Rickard KA. Comparison of inhaled salmeterol and oral zafirlukast in asthmatic patients using concomitant inhaled corticosteroids. Medgenmed Computer File: Medscape General Medicine 2001;3(4):3. - PubMed
Nelson 2004 {published data only}
-
- Nelson H, Stauffer J, Yancey S, Prillaman B, Sutton L, Dorinsky. In patients with both uncontrolled asthma and allergic rhinitis, montelukast added to fluticasone propionate/salmeterol provides no additional clinical improvements in overall asthma control regardless of baseline asthma severity [Abstract]. American Thoracic Society 100th International Conference 2004;D36:A49.
Nelson 2004a {published data only}
-
- Nelson HS, Yancey S, Waitkus‐Edwards K, Prillaman B, Philpot E, Dorinsky P. In patients taking fluticasone propionate/salmeterol 100/50µg Diskus(r) for asthma, fluticasone propionate nasal spray 200µg QD is superior to montelukast 10mg QD in the treatment of allergic rhinitis in patients with coexistent allergic rhinitis: implication for the one airway hypothesis [Abstract]. Journal of Allergy and Clinical Immunology 2004;113(Suppl 2):S200.
Nelson 2006 {published data only}
-
- Nelson HS, Weiss ST, Bleecker EK, Yancey SW, Dorinsky PM. The salmeterol multicenter asthma research trial: A comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. chest 2006;129(1):15‐26. - PubMed
Nishima 2005 {published data only}
-
- Nishima S, Furusho K, Morikawa A, Mochizuki H, Akasaka T, Sugimoto H, et al. Pranlukast inhibits exercise‐induced bronchospasm in asthmatic children: A randomized, multicenter, double‐blind, placebo‐controlled two‐period crossover trial. Pediatric Asthma Allergy & Immunology 2005;18(1):5‐11.
Nishimura 1999 {published data only}
-
- Nishimura K, Hajiro T, Ishihara K, Hasegawa T, Taniguchi H, Katayama S, et al. Additive effect of pranlukast in combination with inhaled corticosteroid in the treatment of patients with chronic asthma. European Respiratory Journal. 1999:P837.
Nishiyama 2006 {published data only}
-
- Nishiyama O, Taniguchi H, Kondoh Y, Kimura T, Kato K, Kume H, et al. Comparison of the effects of tulobuterol patch and salmeterol in moderate to severe asthma. Clinical & Experimental Pharmacology & Physiology 2006;33(11):1016‐21. - PubMed
Nishizawa 2002 {published data only}
-
- Nishizawa Y, Greicy‐Goto H, Tanigaki Y, Fushiki S. Sparing effect of Saibokuto inhalation on inhaled beclomethasone dipropionate to halved of reduction of inhaled beclomethasone dipropionate‐dose: Well‐controlled comparative study of Saiboku‐to‐inhalation and sodium cromoglycate‐inhalation. Japanese. Oto‐Rhino‐Laryngology Tokyo 2002;45(Suppl 1):8‐15.
Noonan 1998 {published data only}
-
- Noonan MJ, Chervinsky P, Brandon M, Zhang J, Kundu S, McBurney J, et al. Montelukast, a potent leukotriene receptor antagonist, causes dose‐related improvements in chronic asthma. European Respiratory Journal 1998;11:1232‐9. - PubMed
Noonan 1999 {unpublished data only}
-
- Noonan G, Reiss TF, Shingo S, Nguyen HH, Knorr B. Montelukast (MK‐0476) maintains long‐term asthma control in adult and pediatric patients (aged >6 years). Merck Research Laboratories 1999.
Nsouli 2000 {published data only}
-
- Nsouli SM, McNutt WJ. The addition of montelukast to a low dose inhaled corticosteroid compared with a double‐dose of an inhaled corticosteroid in patients with persistent asthma. Annals of Allergy, Asthma & Immunology 2000;84:159.
Nsouli 2001 {published data only}
-
- Nsouli SM, McNutt WJ. The additive effects of montelukast and salmeterol in moderate asthmatics who are uncontrolled on a low dose on inhaled corticosteroids. Annals of Allergy, Asthma & Immunology 2001; 86:81.;86:81.
O'Byrne 1997 {published data only}
-
- O'Byrne PM. Leukotrienes in the pathogenesis of asthma. Chest 1997;111:27S‐34S. - PubMed
O'Byrne 1997a {published data only}
-
- O'Byrne PM, Barnes NC. Summary: the future promise of mediator inhibitors in 'New oral preventive therapy in asthma and oral leukotriene receptor antagonism'. European Respiratory Review 1997; Vol. 7, issue Review 46:274‐7.
O'Byrne 1997b {published data only}
-
- O'Byrne PM, Israel E, Drazen JM. Antileukotrienes in the treatment of asthma. Annals of Internal Medicine 1997;127:472‐80. - PubMed
O'Connor 1994 {published data only}
-
- O'Connor GT, Weiss ST. Clinical and symptom measures. American Journal of Respiratory & Critical Care Medicine 1994;149:S21‐8. - PubMed
O'Connor 2004 {published data only}
-
- O'Connor RD, Nelson H, Borker R, Emmett A, Jhingran P, Rickard K, et al. Cost effectiveness of fluticasone propionate plus salmeterol versus fluticasone propionate plus montelukast in the treatment of persistent asthma. Pharmacoeconomics 2004;22(12):815‐25. - PubMed
O'Connor 2006 {published data only}
-
- O'Connor RD, Gilmore AS, Manjunath R, Stanford RH, Legorreta AP, Jhingran PM. Comparing outcomes in patients with persistent asthma: a registry of two therapeutic alternatives. Current Medical Research & Opinion 2006;22(3):453‐61. - PubMed
O'Shaughnessy 1996 {published data only}
O'Sullivan 2002 {published data only}
-
- O'Sullivan S, McWeeney M, Akveld M, Berelowitz KS, Burke CM, Poulter LW. Effect of the addition of montelukast to inhaled fluticasone propionate on immunopathology in bronchial biopsies from mild to moderate asthmatics [abstract]. European Respiratory Society Annual Congress. 2002:P2405.
O'Sullivan 2002a {published data only}
-
- O'Sullivan S, McWeeny M, Akveld M, Berelowitz KS, Burke CM, Poulter LW. Effect of the addition of montelukast to inhaled fluticasone propionate on immunopathology in bronchial biopsies from mild to moderate asthmatics. European Respiratory Journal 2002;20(Suppl 38):388s.
O'Sullivan 2003 {published data only}
-
- O'Sullivan S, Akveld M, Burke CM, Poulter LW. Effect of the addition of montelukast to inhaled fluticasone propionate on airway inflammation. American Journal of Respiratory and Critical Care Medicine 2003;167(5):745‐50. - PubMed
Obase 2001 {published data only}
-
- Obase Y, Shimoda T, Tomari S, Mitsuta K, Fukushima C, Kawano K, et al. Efficacy and safety of long‐term treatment of asthma in patients with pranlukast, a cysteinyl‐leukotriene‐receptor antagonist: a four‐year follow‐up study. Annals of Allergy, Asthma, & Immunology 2001;87(1):43‐7. - PubMed
Obata 1992 {published data only}
-
- Obata T, Okada Y, Motoishi M, Nakagawa N, Terawaki T, Aishita H. In vitro antagonism of ONO‐1078, a newly developed anti‐asthma agent, against peptide leukotrienes in isolated guinea pig tissues. Japanese Journal of Pharmacology 1992;60:227‐37. - PubMed
Odjakova 2000 {published data only}
-
- Odjakova TT, Peneva MA, Kissiova KP, Balev SS, Radkov JD. Changes in some lymphocyte markers after therapy with fluticasone propionate and montelukast in patients with bronchial asthma. European Respiratory Journal 2000;16(Suppl 31):163s.
Ohbayashi 2007 {published data only}
-
- Ohbayashi H, Adachi M, Ichinose M, Ohta K, Kokubu F, Sano Y, et al. A multicenter, open‐label, randomized comparison of suppressive effects on asthmatic inflammation of lower airways and improved effects on health‐related QOL between HFA‐BDP and fluticasone propionate. Japanese Journal of Allergology 2007;56(6):577‐86. - PubMed
Ohbayashi 2009 {published data only}
-
- Ohbayashi H, Shibata N, Adachi M. The evaluation of the additional effect of pranlukast to salmeterol/fluticasone combination therapy using impulse oscillometry system in a randomized crossover study [Abstract]. American Thoracic Society International Conference 2009;A2764:Poster J32.
Ohbayashi 2009a {published data only}
-
- Ohbayashi H, Shibata N, Hirose T, Adachi M. Additional effects of pranlukast in salmeterol/fluticasone combination therapy for the asthmatic distal airway in a randomized crossover study. Pulmonary Pharmacology & Therapeutics 2009;22(6):574‐9. - PubMed
Ohkura 2009 {published data only}
-
- Ohkura N, Fujimura M, Tokuda A, Takato H, Tamori S, Waseda Y, et al. Additional effects of pranlukast on exhaled nitric oxide levels in patients with persistent asthma. Therapeutic Research 2009;30(8):1361‐6.
Ohta 2009 {published data only}
-
- Ohta K, Miyamoto T, Amagasaki T, Yamamoto M. Efficacy and safety of omalizumab in an Asian population with moderate‐to‐severe persistent asthma. Respirology 2009;14(8):1156‐65. - PubMed
Okudaira 1997 {published data only}
-
- Okudaira H. Challenge studies of a leukotriene receptor antagonist. Chest 1997;111:46S‐51S. - PubMed
Oosaki 1997 {published data only}
-
- Oosaki R, Mizushima Y, Kashii T, Kawasaki A, Kobayashi M. Therapeutic effect of pranlukast, a selective cysteinyl leukotriene receptor antagonist, on bronchial asthma. International Archives of Allergy & Immunology 1997;114(1):97‐100. - PubMed
Oosaki 1997a {published data only}
-
- Oosaki R, Mizushima Y, Kawasaki A, Kashii T, Mita H, Shida T, et al. Urinary excretion of leukotriene E4 and 11‐dehydrothromboxane B2 in patients with spontaneous asthma attacks. International Archives of Allergy & Immunology 1997;114:373‐8. - PubMed
Oppenheimer 2008 {published data only}
-
- Oppenheimer J, Mosnaim G, Waitkus‐Edwards K, Prillaman B, Ortega H. Fluticasone propionate/salmeterol via diskus is superior to montelukast in overall asthma control in subjects with both asthma and allergic rhinitis [Abstract]. Chest 2008;134(4):94002s.
Ostrom 2003 {published data only}
-
- Ostrom NK, DeCotiis BA, Lincourt WR, Edwards LD, Crim CC. A comparison of low does fluticasone propionate and montelukast in children 6‐12 years of age with persistent asthma [abstract]. American Thoracic Society 99th International Conference 2003;A117:D70.
Overbeek 2002 {published data only}
-
- Overbeek SE, O'Sullivan S, Leman K, Mulder PGM, Hoogsteden, Prins JB. Treatment with montelukast is less effective in reducing eosinophilic airway inflammation than fluticasone propionate in atopic asthmatics. American Journal of Respiratory and Critical Care Medicine 2002;165(Suppl 8):A215.
Palmqvist 2003 {published data only}
-
- Palmqvist M, Bruce C, Sjostrand M, Arvidsson P, Lotvall J. Inhibiton of allergen‐induced late asthmatics reaction by montelukast vs fluticasone propionate [Abstract]. European Respiratory Journal 2003;22(Suppl 45):2220.
Palmqvist 2005 {published data only}
-
- Palmqvist M, Bruce C, Sjostrand M, Arvidsson P, Lotvall J. Differential effects of fluticasone and montelukast on allergen‐induced asthma. Allergy 2005;60(1):65‐70. - PubMed
Panettieri 1997 {published data only}
-
- Panettieri RA, Tan EML, Ciocca V, Luttman MA, Leonard TB, Hay DWP. Effects of LTD4 on human airways smooth muscle cell proliferation, matrix expression and contraction in vitro: differential sensitivity to cysteinyl leukotriene receptor antagonists. SmithKline Beecham Pharmaceuticals 1997. - PubMed
Papadopoulos 2009 {published data only}
-
- Papadopoulos NG, Philip G, Giezek H, Watkins M, Smugar SS, Polos PG. The efficacy of montelukast during the allergy season in pediatric patients with persistent asthma and seasonal aeroallergen sensitivity. Journal of Asthma 2009;46(4):413‐420. - PubMed
Pasaoglu 2008 {published data only}
-
- Pasaoglu G, Mungan D, Abadoglu O, Misirligil Z. Leukotriene receptor antagonists: a good choice in the treatment of premenstrual asthma?. Journal of Asthma 2008;45(2):95‐99. - PubMed
Patel 2010 {published data only}
-
- Patel YA, Patel P, Bavadia H, Dave J, Tripathi CB. A randomized, open labelled, comparative study to assess the efficacy and safety of controller medications as add on to inhaled corticosteroid and long‐acting beta2 agonist in the treatment of moderate‐to‐severe persistent asthma. Journal of Postgraduate Medicine 2010;56(4):270‐274. - PubMed
Paterson 1999 {published data only}
-
- Paterson MC, Wilson AM, Dempsey OJ, Sims EJ, Lipworth BJ. The effect of combination therapy with salmeterol and montelukast in asthmatic patients receiving inhaled corticosteroids. European Respiratory Society. 1999:P3490.
Pavord 2007 {published data only}
Pearlman 1999 {published data only}
-
- Pearlman DS, Ostrom NK, Bronsky EA, Bonuccelli CM, Hanby LA. The leukotriene D4‐receptor antagonist zafirlukast attenuates exercise‐induced bronchoconstriction in children. Journal of Pediatrics 1999;134:273‐9. - PubMed
Pearlman 2002 {published data only}
-
- Pearlman DS, White MV, Lieberman AK, Pepsin PJ, Kalberg C, Emmett A, et al. Fluticasone propionate/salmeterol combination compared with montelukast for the treatment of persistent asthma. Annals of Allergy, Asthma & Immunology 2002;88(2):227‐35. - PubMed
Pedersen 2007 {published data only}
-
- Pedersen S, Agertoft L, Williams‐Herman D, Kuznetsova O, Reiss TF, Knorr B, et al. Placebo‐controlled study of montelukast and budesonide on short‐term growth in prepubertal asthmatic children. Pediatric Pulmonology 2007;42(9):838‐43. - PubMed
Pereira 1989 {published data only}
-
- Pereira CAdC, Brito VC, Olieveira VMC, Paitl CE, Nery LE, Romaldini H, et al. Effects of corticosteroids administered early by inhalation in stable chronic obstructive pulmonary diseases. American Review of Respiratory Disease 1989;15(52):1348‐53.
Perng 2004 {published data only}
-
- Perng DW, Huang HY, Lee YC, Perng RP. Leukotriene modifier vs inhaled corticosteroid in mild‐to‐moderate asthma: Clinical and anti‐inflammatory effects. Chest 2004;125(5):1693‐9. - PubMed
Peroni 2005a {published data only}
-
- Peroni DG, Piacentini GL, Bodini A, Ress M, Costella S, Boner AL. Montelukast versus formoterol as second‐line therapy in asthmatic children exposed to relevant allergens. Allergy & Asthma Proceedings 2005;26(4):283‐6. - PubMed
Philip 2005 {published data only}
-
- Philip G, Nayak AS, Berger WE, Leynadier F, Vrijens F, Dass SB, et al. The effect of montelukast on rhinitis symptoms in patients with asthma and seasonal allergic rhinitis. Allergologie 2005;28(9):343‐54. - PubMed
Phipatanakul 2003 {published data only}
-
- Phipatanakul W, Greene C, Downes SJ, Cronin B, Eller TJ, Schneider LC, et al. Montelukast improves asthma control in asthmatic children maintained on inhaled corticosteroids. Annals of Allergy Asthma & Immunology 2003;91(1):49‐54. - PubMed
Phipatanakul 2003a {published data only}
-
- Phipatanakul W, Greene C, Downes S, Cronin B, Schneider L, Irani A. The efficacy of montelukast as an addictive and steroid sparing agent in children with asthma previously maintained on inhaled corticosteroid therapy [Abstract]. Journal of Allergy and Clinical Immunology 2003;111(Suppl 2):S146.
Pieters 2005 {published data only}
-
- Pieters WR, Wilson KK, Smith HC, Tamminga JJ, Sondhi S. Salmeterol/fluticasone propionate versus fluticasone propionate plus montelukast: a cost‐effective comparison for asthma. Treatments in Respiratory Medicine 2005;4(2):129‐38. - PubMed
Pizzichini 1999 {unpublished data only}
-
- Pizzichini E, Leff JA, Reiss TF, Hendeles L, Boulet L‐P, Wei LX, et al. Montelukast reduces airway eosinophilic inflammation in asthma: a randomized, controlled trial. European Respiratory Journal 1999;14(1):12‐8. - PubMed
Plaza 2005 {published data only}
-
- Plaza V, Cobos A, Ignacio‐Garcia JM, Molina J, Bergonon S, Garcia‐Alonso F, et al. Cost‐effectiveness of an intervention based on the Global INitiative for Asthma (GINA) recommendations using a computerized clinical decision support system: A physicians randomized trial. Spa Medicina Clinica 2005;124(6):201‐6. - PubMed
Pogson 2008 {published data only}
-
- Pogson ZEK, Antoniak MD, Pacey SJ, Lewis SA, Britton JR, Fogarty AW. Does a low sodium diet improve asthma control?: A randomized controlled trial. American Journal of Respiratory and Critical Care Medicine 2008;178(2):132‐8. - PubMed
Pohl 2006 {published data only}
-
- Pohl WR, Vetter N, Zwick H, Hrubos W. Adjustable maintenance dosing with budesonide/formoterol or budesonide: Double‐blind study. Respiratory Medicine 2006;100(3):551‐60. - PubMed
Polos 2003 {published data only}
-
- Polos PG. Montelukast or salmeterol added to fluticasone in uncontrolled asthma: a subgroup analysis of the IMPACT study [Abstract]. Journal of Allergy and Clinical Immunology 2003;111(Suppl 2):S126.
Polos 2004 {published data only}
-
- Polos P, Garcia Garcia ML, Gilles L, Tozzi CA, Knorr B, Reiss TF. Montelukast vs fluticasone in patients aged 6 to 14 with mild persistent asthma the MOSAIC study [Abstract]. European Respiratory Journal 2004;24(Suppl 48):377s.
Ponce 2009 {published data only}
-
- Ponce Castro H, Rodríguez Espino S, Rodríguez Orozco AR. Administration of budesonide (inhaled steroid) to children to control intermittent asthma. Revista Alergia Mexico 2009;26(1):9‐12. - PubMed
Price 1999 {unpublished data only}
-
- Price DB, Wolfe S. Montelukast: real life effectiveness and cost‐effectiveness in primary care. Thorpe Medical Research 1999.
Price 2002 {published data only}
-
- Price DB, Hernandez D, Magyar P, Fiterman J, Beeh KM, James IG, et al. Adding montelukast is at least as efficacious as doubling the budesonide dose in persistent asthma: Results of the compact study. American Journal of Respiratory and Critical Care Medicine 2002;165(Suppl8):A216.
Price 2003 {published data only}
Price 2004 {published data only}
-
- Price DB, Zhang Q, Kocevar VS, Polos PG, Yin DD. Healthcare resource use the following addition of montelukast to budesonide compared to doubling the dose of budesonide in patients with inadequately controlled asthma (COMPACT trial) [Abstract]. European Respiratory Journal 2004;24(Suppl 48):126s.
Price 2006 {published data only}
-
- Price DB, Swern A, Tozzi CA, Philip G, Polos P. Effect of montelukast on lung function in asthma patients with allergic rhinitis: analysis from the COMPACT trial. Allergy 2006;61(6):737‐42. - PubMed
Pullerits 1999 {published data only}
-
- Pullerits T, Praks L, Skoogh BE, Ani R, Lotvall J. Randomized placebo‐controlled study comparing a leukotriene receptor antagonist and a nasal glucocorticoid in seasonal allergic rhinitis. American Journal of Respiratory & Critical Care Medicine 1999;159(6):1814‐8. - PubMed
Pullerits 2001 {published data only}
-
- Pullerits T, Praks L, Baker R, Ani R, Lotvall J. Comparison of nasal fluticasone propionate, montelukast, and combined montelukast+loratadine in allergic rhinitis [abstract]. American Journal of Respiratory and Critical Care Medicine 2001;163(5 Suppl):A193.
Pullerits 2002 {published data only}
-
- Pullerits T, Praks L, Ristioja V, Lotvall J. Comparison of a nasal glucocorticoid, antileukotriene, and a combination of antileukotriene and antihistamine in the treatment of seasonal allergic rhinitis. Journal of Allergy & Clinical Immunology 2002;109(6):949‐55. - PubMed
Qaqundah 2006 {published data only}
-
- Qaqundah PY, Sugerman RW, Ceruti E, Maspero JF, Kleha JF, Scott CA, et al. Efficacy and safety of fluticasone propionate hydrofluoroalkane inhalation aerosol in pre‐school‐age children with asthma: A randomized, double‐blind, placebo‐controlled study. Journal of Pediatrics 2006;149(5):663‐70. - PubMed
Rachelefsky 1997 {published data only}
-
- Rachelefsky G. Childhood asthma and allergic rhinitis: the role of leukotrienes. Journal of Pediatrics 1997;131:348‐55. - PubMed
Ragab 2001 {published data only}
-
- Ragab S, Parikh A, Darby YC, Scadding GK. An open audit of montelukast, a leukotriene receptor antagonist, in nasal polyposis associated with asthma. Clinical & Experimental Allergy 2001;31(9):1385‐91. - PubMed
Ramsay 1997 {published data only}
-
- Ramsay CF, Kan CI, Nieman RB, Wang J, Krieken JHJM, Willems LNA, et al. The effects of oral pranlukast on airway immunopathology and clinical parameters in patients with asthma. American Thoracic Society. 1997:Abs C21.
Ramsay 1998 {published data only}
-
- Ramsay CF, Kan CI, Sterk PJ, Barnes NC. Pranlukast improves spirometry and bronchial hyperresponsiveness (BHR) in patients with mild asthma. American Journal of Respiratory & Critical Care Medicine 1998;157(3):A411.
Rand 2004 {published data only}
-
- Rand CS, Bilderbank A, Krishnan J, Riekert K, Schiller K, MIAMI Study Research Group. Adherence with oral montelukast sodium or fluticasone propionate in a clinical trial [Abstract]. Journal of Allergy and Clinical Immunology 2004;113(Suppl 2):s157.
Ratner 2003 {published data only}
-
- Ratner PH, Howland WC, Arastu R, Philpot EE, Klein KC, Baidoo CA, et al. Fluticasone propionate aqueous nasal spray provided significantly greater improvement in daytime and nighttime nasal symptoms of seasonal allergic rhinitis compared with montelukast.[comment]. Annals of Allergy Asthma & Immunology 2003;90(5):536‐42. - PubMed
Reiss 1996 {published data only}
-
- Reiss TF, Altman LC, Chervinsky P, Bewtra A, Stricker WE, Noonan GP, et al. Effects of montelukast (MK‐0476), a new potent cysteinyl leukotriene (LTD4) receptor antagonist, in patients with chronic asthma. Journal of Allergy & Clinical Immunology 1996;98:528‐34. - PubMed
Reiss 1997 {unpublished data only}
-
- Reiss TF, White R, Noonan G, Korenblat P, Hess J, Shingo S. Montelukast (MK‐0476) a cys LT1 receptor antagonist improves the signs and symptoms of asthma over one year of treatment. European Respiratory Journal 1997;10 Suppl(25):437.
Reiss 1997b {published data only}
-
- Reiss TF, et al. Montelukast improves asthma outcomes over a 3‐month treatment period. American Thoracic Society. 1997.
Reiss 1997c {published data only}
Reiss 1998 {published data only}
-
- Reiss TF, Chervinsky P, Dockhorn RJ, Shingo S, Seidenberg B, Edwards TB. Montelukast, a once‐daily leukotriene receptor antagonist, in the treatment of chronic asthma. Archives of Internal Medicine 1998;158:1213‐20. - PubMed
Reiss 2008 {published data only}
-
- Reiss TF, Lu S, Liu N. A randomized study comparing concomitant montelukast and loratadine with montelukast, loratadine and beclomethasone in patients with chronic asthma [Abstract]. Chest 2008;134(4):93001s. - PubMed
Riccioni 2001a {published data only}
-
- Riccioni G, Guagnano MT, Castronuovo M, Benedicts M, Della R. Zafirlukast versus budesonide on bronchial reactivity in subjects with mild‐persistent asthma. European Respiratory Journal 2001;18(Suppl 33):263s. - PubMed
Riccioni 2002 {published data only}
-
- Riccioni G, Santilli F, Guagnano MT, Vecchia RD. Montelukast versus budesonide on quality of life in asthmatic subjects [abstract]. European Respiratory Society Annual Congress. 2002:P757.
Riccioni 2003a {published data only}
-
- Riccioni G, D'Orazio N, Castronuovo M, Menna V, Lambo MS, Stefano F, et al. Dosage tapering of inhalatory budesonide in subjects with mild to moderate persistent asthma treated with montelukast [Abstract]. European Respiratory Journal 2003;22(Suppl 45):Abstract No: P713.
Riccioni 2005 {published data only}
-
- Riccioni G, Della Vecchia R, Castronuovo M, Ilio C, D'Orazio N. Tapering dose of inhaled budesonide in subjects with mild‐to‐moderate persistent asthma treated with montelukast: A 16‐week single‐blind randomized study. Annals of Clinical & Laboratory Science 2005;35(3):285‐9. - PubMed
Rickard 1999 {published data only}
-
- Rickard KA, Yancey S, Emmet AH, Kalberg CJ. Salmeterol compared to zafirlukast when added to inhaled corticosteroid therapy in patients with persistent asthma. European Respiratory Society. 1999:P839.
Rickard 2001 {published data only}
-
- Rickard K, Dorinsky PM, Knobil K, Pepsin P, Akveld ML. The salmeterol/fluticasone propionate combination 50/100µg bid is more effective than oral montelukast 10mg od as a first line therapy in mild and moderate asthmatics. European Respiratory Journal 2001;18(Suppl 33):262s.
Ringdal 1997 {published data only}
-
- Ringdal N, Whitney JG, Summerton L. Problems with inhaler technique and patient preference for oral therapy tablet zafirlukast vs. inhaled beclomethasone. European Respiratory Journal 1997;10 Suppl(25):437, abs P2806.
Ringdal 2003 {published data only}
-
- Ringdal N, Eliraz A, Pruzinec R, Weber HH, Mulder PG, Akveld M, et al. The salmeterol/fluticasone combination is more effective than fluticasone plus oral montelukast in asthma. Respiratory Medicine 2003;97(3):234‐41. - PubMed
Robinson 2001 {published data only}
-
- Robinson DS, Campbell D, Barnes PJ. Addition of leukotriene antagonists to therapy in chronic persistent asthma: a randomised double‐blind placebo‐controlled trial. Lancet 2001;357(9273):2007‐11. - PubMed
Rosenhall 2003 {published data only}
-
- Rosenhall L, Elvstrand A, Tilling B, Vinge I, Jemsby P, Stahl E, et al. One‐year safety and efficacy of budesonide/formoterol in a single inhaler (Symbicort Turbuhaler ) for the treatment of asthma. Respiratory Medicine 2003;97(6):702‐8. - PubMed
Rowe 2007 {published data only}
-
- Rowe BH, Wong E, Blitz S, Diner B, Mackey D, Ross S, et al. Adding Long‐acting beta‐agonists to Inhaled Corticosteroids after Discharge from the Emergency Department for Acute Asthma: A Randomized Controlled Trial. Academic Emergency Medicine 2007;14(10):833‐40. - PubMed
Ruggins 2003 {published data only}
-
- Ruggins N. Pragmatic trial of add‐on therapy in Paediatric Asthma. National Research Register 2003.
Sahn 1997 {published data only}
-
- Sahn SA, Galant S, Murray J, Bronsky E, Spector S, Faiferman I, et al. Pranlukast (Ultair) improves FEV in patients with asthma: results of a 12‐week multicenter study vs. nedocromil. American Thoracic Society. 1997:Abs C49.
Sano 2006 {published data only}
-
- Sano Y, Adachi M, Kiuchi T, Miyamoto T. Effects of nebulized sodium cromoglycate on adult patients with severe refractory asthma. Respiratory Medicine 2006;100(3):420‐33. - PubMed
Schneider 2008 {published data only}
Schuh 2009 {published data only}
-
- Schuh S, Willan AR, Stephens D, Dick PT, Coates A. Can montelukast shorten prednisolone therapy in children with mild to moderate acute asthma? A randomized controlled trial. Journal of Pediatrics 2009;155(6):795‐800. - PubMed
Schwartz 1995 {published data only}
-
- Schwartz HJ, Petty T, Reed R, Dube LM, Swanson LJ, Zileuton Study Group. The comparative effects of zileuton, a 5‐lipoxygenase inhibitor, vs. theophylline in patients with moderate asthma: results from a 13‐week multicenter trial. ALA/ATS. 1995.
SD‐004‐0216 {published data only}
-
- Oxis Turbuhaler® (formoterol), Accolate® (zafirlukast) or placebo as add on treatment to Pulmicort Turbuhaler® (budesonide) in asthmatic patients on inhaled steroids. AstraZeneca Clinical Trials 2000.
Shah 2003 {published data only}
-
- Shah AR. Which Is More Steroid Sparing in Persistent Bronchial Asthma: Montelukast Or Theophylline? [Abstract]. Chest 2003;124(4):107.
Shah 2004 {published data only}
-
- Shah A, Solanki R, Shah K. Effect of add on therapy compared with doubling steroid inhalation in bronchial asthma [Abstract]. Thorax 2004;59(Suppl 2):ii70.
Shah 2006 {published data only}
-
- Shah AR, Sharples LD, Solanki RN, Shah KV. Double‐blind, randomised, controlled trial assessing controller medications in asthma. Respiration 2006;73(4):449‐56. - PubMed
Sheth 2002 {published data only}
-
- Sheth K, Borker R, Emmett A, Rickard K, Dorinsky P. Cost‐effectiveness comparison of salmeterol/fluticasone propionate versus montelukast in the treatment of adults with persistent asthma.. Pharmacoeconomics 2002;20(13):909‐18. - PubMed
Shimoda 2005 {published data only}
-
- Shimoda T, Kishikawa R, Shoji S, Nishima S. The efficacy of anti‐inflammatory treatment in mild intermittent bronchial asthma [Abstract]. Journal of Allergy & Clinical Immunology 2005;115(Suppl 2):S1.
Shingo 2001 {unpublished data only}
-
- Shingo S, Zhang J, Noonan N, Reiss TF, Leff JA. A standardized composite clinical score allows safe tapering of inhaled corticosteroids in an asthma clinical trial.. Unpublished data (Personal Communication:Theodore Reiss June 2001) 2001.
Shoji 1999 {published data only}
-
- Shoji T, Yoshida S, Sakamoto H, Hasegawa H, Nakagawa H, Amayasu H. Anti‐inflammatory effect of roxithromycin in patients with aspirin‐intolerant asthma. Clinical & Experimental Allergy 1999;9(7):950‐6. - PubMed
Simons 2001 {published data only}
-
- Simons FER, Villa JR, Lee BW, Teper AM, Lyttle B, Aristizabal G, et al. Montelukast added to budesonide in children with persistent asthma: a randomised, double‐blind, crossover study. Journal of Pediatrics 2001;138(5):694‐8. - PubMed
Simpson 2004 {published data only}
-
- Simpson MD, Burton DL, Burton MA, Gissing PM, Bowman SL. Pharmaceutical Care: Impact on Asthma Medication Use. Journal of Pharmacy Practice & Research 2004;34(1):26‐9.
Sims 2003 {published data only}
Sims 2008 {published data only}
-
- Sims E, Freeman D, Kemp L, Musgrave S, Juniper L, Gilbert R, et al. Should guidelines be revised for add on therapy in asthma? A 2 year randomized pragmatic equivalence trail of leukotriene antagonist (LTRAs) and long acting beta agonists (LABAs) with inhaled corticosteroids (ICS) in primary care [Abstract]. European Respiratory Society Annual Congress. 2008:E233.
Smith 1993 {published data only}
-
- Smith LJ, Glass M, Minkwitz MC. Inhibition of leukotriene D4‐induced bronchoconstriction in subjects with asthma: a concentration‐effect study of ICI 204,219. Clinical Pharmacology & Therapeutics 1993;54(4):430‐6. - PubMed
Smith 1997 {published data only}
-
- Smith LJ, Hanby LA, Simonson MS, Simonson SG. Effect of zafirlukast on leukotriene D4 (LTD4) ‐ induced bronchoconstriction in asthmatic patients receiving inhaled corticosteroids [abstract]. European Respiratory Journal 1997;10(Suppl 25):437S.
Smith 1998 {published data only}
-
- Smith LJ, Hanby LA, Lavins BJ, Simonson SG. A single dose of zafirlukast reduces LTD4‐induced bronchoconstriction in patients on maintenance inhaled corticosteroid therapy. Annals of Allergy, Asthma, & Immunology 1998;81:43‐9. - PubMed
Smugar 2009 {published data only}
-
- Smugar SS, Fogel R, Aristizabal G, Rosario N, Loeys T, Gaile S, et al. Effect of montelukast or salmeterol added to inhaled fluticasone on response to albuterol in children with exercise‐induced bronchoconstriction [Abstract]. European Respiratory Society Annual Congress. 2009:P1221.
Smugar 2009a {published data only}
-
- Smugar SS, Fogel R, Rosario N, Aristizabal G, Loeys T, Gaile S, et al. Attenuation of exercise‐induced bronchoconstriction with montelukast or salmeterol added to inhaled fluticasone in children [Abstract]. European Respiratory Society Annual Congress. 2009:P1217.
Spahn 1996a {published data only}
-
- Spahn JD, Szefler SJ. The etiology and control of bronchial hyperresponsiveness in children. Current Opinion in Pediatrics 1996;8:591‐6. - PubMed
Spector 1992 {published data only}
-
- Spector SL, Glass M, Minkwitz MC, ICI Asthma Trial Group. The effect of six weeks of therapy with oral doses of ICI 204,219 in asthmatics. American Review of Respiratory Disease 1992;145(4):A16.
Spector 1994 {published data only}
-
- Spector SL, Smith LJ, Glass M. Effects of 6 weeks of therapy with oral doses of ICI 204,219, a leukotriene D4 antagonist, in subjects with bronchial asthma. American Journal of Respiratory & Critical Care Medicine 1994;150:618‐23. - PubMed
Spector 1995 {published data only}
-
- Spector S, Miller CJ, Glass M. 13‐week dose‐response study with Accolate (zafirlukast) in patients with mild to moderate asthma. American Journal of Respiratory & Critical Care Medicine 1995;151(4):A379.
Spector 1996 {published data only}
-
- Spector SL. Management of asthma with zafirlukast. Drugs 1996;52 Suppl(6):36‐46. - PubMed
Stanford 2002 {published data only}
-
- Stanford RH, Borker R, Dorinsky P, Pepsin P, Kalberg C, Emmett A, et al. The cost and efficacy of fluticasone propionate/salmeterol combination versus montelukast in the treatment of adults with persistent asthma. Chest. 2002:P422.
Stensrud 2006 {published data only}
-
- Stensrud T, Berntsen S, Carlsen KH. Humidity influences exercise capacity in subjects with exercise‐induced bronchoconstriction (EIB). Respiratory Medicine 2006;100(9):1633‐41. - PubMed
Stevenson 2005 {published data only}
-
- Stevenson DD, Mehra PK, White AA, Gupta S, Woessner KM, Simon RA. Failure of tacrolimus to prevent aspirin‐induced respiratory reactions in patients with aspirin‐exacerbated respiratory disease. Journal of Allergy & Clinical Immunology 2005;116(4):755‐60. - PubMed
Sthoeger 2007 {published data only}
-
- Sthoeger ZM, Eliraz A, Asher I, Berkman N, Elbirt D. The beneficial effects of Xolair (Omalizumab) as add‐on therapy in patients with severe persistent asthma who are inadequately controlled despite best available treatment (GINA 2002 step IV ‐ The Israeli arm of the INNOVATE study). Israel Medical Association Journal 2007;9(6):472‐5. - PubMed
Storms 2001 {published data only}
-
- Storms W, Michele TM, Knorr B, Noonan G, Shapiro G, Zhang J, et al. Clinical safety and tolerability of montelukast, a leukotriene receptor antagonist, in controlled clinical trials in patients aged > or = 6 years. Clinical & Experimental Allergy 2001;31(1):77‐87. - PubMed
Storms 2004 {published data only}
-
- Storms W, Chervinsky P, Ghannam AF, Bird S, Hustad CM, Edelman JM, et al. A comparison of the effects of oral montelukast and inhaled salmeterol on response to rescue bronchodilation after challenge. Respiratory Medicine 2004;98(11):1051‐62. - PubMed
Strauch 2003 {published data only}
-
- Strauch E, Moske O, Thoma S, Van'S Gravesande KS, Ihorst G, Brandis M, et al. A randomized controlled trial on the effect of montelukast on sputum eosinophil cationic protein in children with corticosteroid‐dependent asthma. Pediatric Research 2003;54(2):198‐203. - PubMed
Strunk 2003 {published data only}
-
- Strunk RC, Szefler SJ, Phillips BR, Zeiger RS, Chinchilli VM, Larsen G, et al. Relationship of exhaled nitric oxide to clinical and inflammatory markers of persistent asthma in children. Journal of Allergy & Clinical Immunology 2003;112(5):883‐92. - PubMed
Strunk 2008 {published data only}
Sugihara 2010 {published data only}
-
- Sugihara N, Kanada S, Haida M, Ichinose M, Adachi M, Hosoe M, et al. 24‐h bronchodilator efficacy of single doses of indacaterol in Japanese patients with asthma: A comparison with placebo and salmeterol. Respiratory Medicine 2010;104(11):1629‐37. - PubMed
Suguro 1997 {published data only}
-
- Suguro H, Majima T, Ichimura K, Hashimoto N, Koyama S, Horie T. Effect of a leukotriene antagonist, pranlukast hydrate, on exercise‐induced bronchoconstriction. American Thoracic Society. 1997:Abs C49.
Suissa 1997 {published data only}
-
- Suissa S, Dennis R, Ernst P, Sheehy O, Wood‐Dauphinee S. Effectiveness of the leukotriene receptor antagonist zafirlukast for mild‐to‐moderate asthma: a randomized, double‐blind, placebo‐controlled trial. Annals of Internal Medicine 1997;126(3):177‐83. - PubMed
Sutherland 2010 {published data only}
-
- Sutherland ER, Camargo CA, Busse WW, Meltzer EO, Orgega HG, Yancey SW, et al. Comparative effect of body mass index on response to asthma controller therapy. Allergy and Asthma Proceedings 2010;31:20‐5. - PubMed
Suzuki 1997 {published data only}
-
- Suzuki N, Kudo K, Sano Y, Adachi M, Kanazawa M, Kudo S, et al. Efficacy of oral pranlukast, a leukotriene‐receptor antagonist, in the treatment of asthma: an open study in Tokyo. American Thoracic Society. 1997:Abs C49.
Svensson 1994 {published data only}
-
- Svensson C, Greiff L, Andersson M, Alkner U, Persson CGA. Bradykinin‐, leukotriene D4‐, and histamine‐induced mucosal exudation of plasma in human airways in vivo. Abbott Laboratories 1994.
Swern 2008 {published data only}
-
- Swern AS, Tozzi CA, Knorr B, Bisgaard H. Predicting an asthma exacerbation in children 2 to 5 years of age. Annals of Allergy Asthma and Immunology 2008;101(6):626‐30. - PubMed
Swernson 2003 {published data only}
-
- Swenson C, Fell S, Liu LY, Busse W. Effect of montelukast (mont) or placebo (PL) on asthma exacerbations and markers of airway inflammation with inhaled corticosteroid (ICS) reduction [abstract]. American Thoracic Society 99th International Conference 2003;B034:Poster H47.
Tamaoki 1997 {published data only}
-
- Tamaoki J, Kondo M, Sakai N, Nakata J, Takemura H, Nagai A, et al. Leukotriene antagonist prevents exacerbation of asthma during reduction of high‐dose inhaled corticosteroid. American Journal of Respiratory & Critical Care Medicine 1997;155:1235‐40. - PubMed
Tan 2006 {published data only}
-
- Tan WC, Lamm CJ, Chen YZ, O'Byrne PM, Pedersen S, Busse WW, et al. Effectiveness of early budesonide intervention in Caucasian versus Asian patients with asthma: 3‐Year results of the START study. Respirology 2006;11(6):767‐75. - PubMed
Tashkin 1998 {published data only}
-
- Tashkin DP, Minkwitz MC, Bonuccelli CM. Zafirlukast (Accolate) treatment results in better asthma control in patients with more moderate disease. American Journal of Respiratory & Critical Care Medicine 1998;157(3):A410.
Teper 2009 {published data only}
-
- Teper A, Kofman C, Zaragoza S, Rodriguez V, Lubovich S, Eguiguren C, et al. Comparison of fluticasone propionate + salmeterol (FP+S) and montelukast (MK) on bronchial reactivity (BR), pulmonary function (PF) and clinical outcome in children with mild asthma [Abstract]. European Respiratory Society Annual Congress. 2009:[P1215].
Terzano 2001 {published data only}
-
- Terzano C, Allegra L, Barkai L, Cremonesi G. Beclomethasone dipropionate versus budesonide inhalation suspension in children with mild to moderate persistent asthma. European Review for Medical & Pharmacological Sciences 2001;5(1):17‐24. - PubMed
Thoma 2002 {published data only}
-
- Thoma S. Anti‐inflammatory effect of montelukast on corticosteroid‐dependent children with asthma [Dissertation] [[Reduktion der bronchialen Inflammation bei mildem bis mittelschwerem Asthma bronchiale im Kindesalter unter Additivtherapie mit Montelukast und inhalativem Budesonid]]. Universitat Freiburg 2002.
Todi 2010 {published data only}
-
- Todi VK, Lodha R, Kabra SK. Effect of addition of single dose of oral montelukast to standard treatment in acute moderate to severe asthma in children between 5 and 15 years of age: A randomised, double‐blind, placebo controlled trial. Archives of Disease in Childhood 2010;95(7):540‐3. - PubMed
Tognella 2004 {published data only}
-
- Tognella S, Micheletto C, Visconti MP, Dal Negro RW. Additive Effects of Montelukast on Bronchial Hyperresponsiveness to MCh and LTE4 Urine Levels in Mild‐persistent Atopic Asthmatics Assuming ICS [Abstract]. Chest 2004;126(Suppl 4):814S.
Tohda 2002 {published and unpublished data}
-
- Tohda Y, Fujimura M, Taniguchi H, Takagi K, Igarashi T, Yasuhara H, et al. Leukotriene receptor antagonist, montelukast, can reduce the need for inhaled steroid while maintaining the clinical stability of asthmatic patients. Clinical & Experimental Allergy 2002;32:1180‐6. - PubMed
Tomari 2001 {published data only}
-
- Tomari S, Shimoda T, Kawano T, Mitsuta K, Obase Y, Fukushima C, et al. Effects of pranlukast, a cysteinyl leukotriene receptor 1 antagonist, combined with inhaled beclomethasone in patients with moderate or severe asthma. Annals of Allergy, Asthma & Immunology i 2001;87:156‐61. - PubMed
Tomita 1999 {published data only}
-
- Tomita K, Hashimoto K, Matsumoto S, Nakamoto N, Tokuyasu H, Yamasaki A, et al. [Pranlukast allows reduction of inhaled steroid dose without deterioration in lung function in adult asthmatics]. Article in Japanese. Arerugi ‐ Japanese Journal of Allergology 1999;48:459‐65. - PubMed
Tonelli 2003 {published data only}
-
- Tonelli M, Zingoni M, Bacci E, Dente FL, Franco A, Giannini D, et al. Short‐term effect of the addition of leukotriene receptor antagonists to the current therapy in severe asthmatics. Pulmonary Pharmacology & Therapeutics 2003;16(4):237‐40. - PubMed
Townley 1995 {published data only}
-
- Townley R, Glass M, Minkwitz MC. 6‐week, dose‐escalation study with Accolate (zafirlukast) in patients with mild to moderate asthma. American Journal of Respiratory & Critical Care Medicine 1995;151(4):A379. - PubMed
Trofor 2002 {published data only}
-
- Trofor A, Rascarachi G, Popa E, Chiselita I. Clinical benefits of montelukast sodium treatment in chronic adult asthma. Revista Medico‐Chirurgicala a Societatii De Medici Si Naturalisti Din Iasi 2002;107(2):298‐302. - PubMed
Tsai 2010 {published data only}
-
- Tsai TC, Lu JH, Chen SJ, Tang RB. Clinical efficacy of house dust mite‐specific immunotherapy in asthmatic children. Pediatrics and Neonatology 2010;51(1):14‐8. - PubMed
Tsuchida 2005 {published data only}
-
- Tsuchida T, Matsuse H, Machida I, Kondo Y, Saeki S, Tomari S, et al. Evaluation of theophylline or pranlukast, a cysteinyl leukotriene receptor 1 antagonist, as add‐on therapy in uncontrolled asthmatic patients with a medium dose of inhaled corticosteroids. Allergy & Asthma Proceedings 2005;26(4):287‐91. - PubMed
Tug 2007 {published data only}
-
- Tug T, Godekmerdan A, Sari N, Karatas F, Erdem ES. Effects of supportive treatment such as antioxidant or leukotriene receptor antagonist drugs on inflammatory and respiratory parameters in asthma patients. Clinical Pharmacology & Therapeutics 2007;81(3):371‐6. - PubMed
Tukiainen 2002 {published data only}
-
- Tukiainen H, Rytila P, Hamalainen K M, Silvasti M S L, Keski‐Karhu J. Safety, tolerability and acceptability of two dry powder inhalers in the administration of budesonide in steroid‐treated asthmatic patients. Respiratory Medicine 2002;96(4):221‐9. - PubMed
Uh 2007 {published data only}
-
- Uh ST, Jung KS, Lee YC, Shim JJ, Park SK, Lee YS, et al. A comparison of the clinical efficacy and safety of salmeterol/fluticasone propionate versus leukotriene antagonist in moderate to severe asthmatics ‐ a sub‐analysis of the SUCCESS Study [Abstract]. Respirology 2007;12(Suppl 4):A148.
Ulrik 2009 {published data only}
-
- Ulrik CS, Diamant Z. Effect of add‐on montelukast to inhaled corticosteroids on excessive airway narrowing in adult asthmatics [Abstract]. European Respiratory Society Annual Congress. 2009:P1962.
Ulrik 2009a {published data only}
-
- Ulrik CS, Diamant Z. Effect of montelukast on excessive airway narrowing response to methacholine in adult asthmatic patients not on controller therapy. Allergy and Asthma Proceedings 2009;30(1):64‐8. - PubMed
Ulrik 2010 {published data only}
-
- Ulrik CS, Diamant Z. Add‐on montelukast to inhaled corticosteroids protects against excessive airway narrowing. Clinical and Experimental Allergy 2010;40(4):576‐81. - PubMed
Van Adelsberg 2005 {published data only}
-
- Adelsberg J, Moy J, Wei LX, Tozzi CA, Knorr B, Reiss TF. Safety, tolerability, and exploratory efficacy of montelukast in 6‐ To 24‐month‐old patients with asthma. Current Medical Research & Opinion 2005;21(6):971‐9. - PubMed
Van Der Meer 2009 {published data only}
-
- Meer V, Bakker MJ, Hout WB, Rabe KF, Sterk PJ, Kievit J, et al. Internet‐based self‐management plus education compared with usual care in asthma: A randomized trial. Annals of Internal Medicine 2009;151(2):110‐20. - PubMed
Vaquerizo 2003 {published data only}
Vastagh 2003 {published data only}
-
- Vastagh E, Kuna P, Calistru P, Bogdan MA. Efficacy and safety of inhaled budesonide delivered once or twice daily via HFA‐134a in mild to moderate persistent asthma in adult patients. Comparison with budesonide CFC. Respiratory Medicine 2003;97(Suppl D):S20‐S28. - PubMed
Verhoeven 2001 {published data only}
Verini 2007 {published data only}
-
- Verini M, Peroni DG, Piacentini GL, Nicodemo A, Rossi N, Bodini A, et al. Comparison of add‐on therapy to inhaled fluticasone propionate in children with asthma: residual volume and exhaled nitric oxide as outcome measures. Allergy & Asthma Proceedings 2007;28(6):691‐4. - PubMed
Vermeulen 2007 {published data only}
-
- Vermeulen JH, Gyurkovits K, Rauer H, Engelstatter R. Randomized comparison of the efficacy and safety of ciclesonide and budesonide in adolescents with severe asthma. Respiratory Medicine 2007;101(10):2182‐91. - PubMed
Vethanayagam 2002 {published data only}
-
- Vethanayagam D, Leigh R, Yoshida M, Watson RM, Rerecich T, Killian K, et al. Effects of montelukast and budesonide alone and together on allergen‐induced airway inflammation in asthma [abstract]. American Journal of Respiratory and Critical Care Medicine 2002;165(8 Suppl):A216. - PubMed
Vidal 2001 {published data only}
-
- Vidal C, Fernandez‐Ovide E, Pineiro J, Nuñez R, González‐Quintela A. Comparison of montelukast versus budesonide in the treatment of exercise‐induced bronchoconstriction. Annals of Allergy, Asthma, & Immunology 2001;86(6):655‐8. - PubMed
Vidal 2001a {published data only}
-
- Vidal C, Fernandez‐Ovide E, Pineiro J Nuñez R, González‐Quintela A. Comparison of montelukast versus budesonide in the treatment of exercise‐induced bronchoconstriction. Annals of Allergy, Asthma, & Immunology 2001;86(6):655‐8. - PubMed
Virchow 1997 {published data only}
-
- Virchow JC, Hassall SM, Summerton L, Harris A. Zafirlukast (Accolate) reduces the impact of asthma in patients taking inhaled corticosteroids. Journal of Allergy & Clinical Immunology 1997;9 Suppl(4):243, Abs. 888.
Virchow 1997a {published data only}
-
- Virchow CJ, Hassall SM, Summerton L, Harris A. Improved asthma control over 6 weeks with zafirlukast in patients on high dose inhaled corticosteroids [abstract]. European Respiratory Journal 1997;10(Suppl 25):437S.
Virchow 1997b {published data only}
-
- Virchow CJ, Hassall SM, Summerton L, Klim J, Harris A. Reduction of asthma exacerbations with zafirlukast in patients on inhaled corticosteroids [abstract]. European Respiratory Journal 1997;10(Suppl 25):420S.
Virnig 2008 {published data only}
-
- Virnig C, Gern JE. Attenuation of the september epidemic of asthma exacerbations in children: A randomized, controlled trial of montelukast added to usual therapy. Pediatrics 2008;122(Suppl 4):S218‐S219. - PubMed
Volovitz 1999 {published data only}
-
- Volovitz B, Tabachnik E, Nussinovitch M, Shtaif B, Blau H, Gil‐ad I, et al. Montelukast, a leukotriene receptor antagonist, reduces the concentration of leukotrienes in the respiratory tract of children with persistent asthma. Journal of Allergy & Clinical Immunology 1999;104:1162‐7. - PubMed
Von Berg 2002 {published data only}
-
- Berg A, Albrecht B, Darlath W, Vo H W, Berdel D. Intraindividual, randomised, double‐blind comparison between sodium cromoglycate and reproterol to assess the protective effect of the single drugs and their combination in children with exercise‐induced asthma. Allergologie 2002;25(11):557‐64.
Wada 2000 {published data only}
-
- Wada K, Minoguchi K, Kohno Y, Oda N, Matsuura T, Kawazu K, et al. Effect of a leukotriene receptor antagonist, pranlukast hydrate, on airway inflammation and airway hyperresponsiveness in patients with moderate to severe asthma. Allergology International 2000;49(1):63‐8.
Wada 2009 {published data only}
-
- Wada M, Nagata S, Kudo T, Shimizu T, Yamashiro Y. Effect of suplatast tosilate on antileukotriene non‐responders with mild‐to‐moderate persistent asthma. Allergology International 2009;58(3):389‐93. - PubMed
Wahedna 1999 {published data only}
Warren 2003 {published data only}
-
- Warren L. Management of Asthma in School‐aged Children On Therapy ‐ MASCOT, Contract signed, not started. National Coordinating Centre for Health Technology Assessment (NCCHTA). http://www.hta.ac.uk/project/1599.asp,Ongoing.
Wasserman 2006 {published data only}
-
- Wasserman RL, Baker JW, Kim KT, Blake KV, Scott CA, Wu W, et al. Efficacy and safety of inhaled fluticasone propionate chlorofluorocarbon in 2‐ to 4‐year‐old patients with asthma: Results of a double‐blind, placebo‐controlled study. Annals of Allergy Asthma & Immunology 2006;96(6):808‐18. - PubMed
Wechsler 1998 {published data only}
-
- Wechsler ME, Garpestad E, Flier SR, Kocker O, Weiland DA, Polito AJ, et al. Pulmonary infiltrates, eosinophilia, and cardiomyopathy following corticosteroid withdrawal in patients with asthma receiving zafirlukast. JAMA 1998;279(6):455‐7. - PubMed
Weinberg 1998 {published data only}
-
- Weinberg EG, Summerton L, Harris A. Assessment of preference for oral zafirlukast vs inhaled beclomethasone in adolescent asthmatics. ERS Annual Congress. 1998; Vol. 12, issue 28:abs P0333.
Weiss 2010 {published data only}
-
- Weiss KB, Gern JE, Johnston NW, Sears MR, Jones CA, Jia G, et al. The back to school asthma study: The effect of montelukast on asthma burden when initiated prophylactically at the start of the school year [Abstract]. Annals of Allergy, Asthma and Immunology 2010;105(2):174‐81. - PubMed
Welch 1994 {published data only}
-
- Welch MJ, Nelson HS, Paull BR, Smith JA, Feiss G, Tobey RE. Effect of RG 12525, a new leukotriene antagonist, on pulmonary function of asthmatic adults. Annals of Allergy 1994;72:348‐52. - PubMed
Wen 2008 {published data only}
-
- Wen CJ, Lai ST, Deng YC, Hung CH. Plasma levels of leukotriene E4 and 9alpha, 11 beta‐PGF2 in asthmatic young children during exacerbation and convalescence. Journal of Medical Sciences 2008;28(6):233‐7.
Wenzel 1994 {published data only}
-
- Wenzel S, Cohn J, Trudeau J, Wilson W, Martin R, Westcott J. Zileuton (Leutrol) decreases urine LTE4, BALF LTB4 and improves lung function in nocturnal asthmatics. Abbott Laboratories 1994.
Wenzel 1995 {published data only}
-
- Wenzel SE, Trudeau JB, Kaminsky DA, Cohn J, Martin RJ, Westcott JY. Effect of 5‐lipoxygenase inhibition on bronchoconstriction and airway inflammation in nocturnal asthma. American Journal of Respiratory & Critical Care Medicine 1995;152(3):897‐905. - PubMed
Wenzel 1997 {published data only}
-
- Wenzel S, Chervinsky P, Kerwin E, Silvers W, Faiferman I, Dubb J. Oral Pranlukast (Ultair) vs. inhaled beclomethasone: results of a 12‐week trial in patients with asthma. American Journal of Respiratory & Critical Care Medicine 1997;155:A203.
Westbroek 1997 {published data only}
-
- Westbroek J, Pasma HR. The effect of inhaled fluticasone propionate (FP) 100 µg bd compared with oral zafirlukast 20 mg bd on bronchial hyperresponsiveness in mild to moderate asthmatics [abstract]. European Respiratory Journal 1997;10(Suppl 25):243S.
Westbroek 1998 {published data only}
-
- Westbroek J, Pasma HR, James MH, Thwaites RMA. Inhaled fluticasone propionate and oral zafirlukast in moderate asthma: a clinical and cost comparison [abstract]. American Journal of Respiratory and Critical Care Medicine 1998;157(Suppl 3):A416.
Westbroek 2000 {published data only}
-
- Westbroek J, Pasma HR. Effects of 2 weeks of treatment with fluticasone propionate 100 mcg b.d. by comparison with zafirlukast 20 mg b.d. on bronchial hyper‐responsiveness in patients with mild to moderate asthma. Respiratory Medicine 2000;94(2):112‐8. - PubMed
Williams 2001 {published and unpublished data}
-
- Williams B, Noonan G, Reiss TF, Knorr B, Guerra J, White R, et al. Long‐term asthma control with oral montelukast and inhaled beclomethasone. Clinical & Experimental Allergy 2001;31:1‐10. - PubMed
Wilson 1999 {published data only}
-
- Wilson AM, Dempsey OJ, Sims EJ, Lipworth BJ. A comparison of salmeterol and montelukast as second‐line therapy in asthmatic patients receiving inhaled corticosteroids. European Respiratory Society. 1999:P3486.
Wilson 2000 {published data only}
-
- Wilson AM, Orr LC, Sims EJ, Dempsey OJ. Antiasthmatic effects of mediator block versus topical corticosteroids in allergic rhinitis and asthma. American Journal of Respiratory & Critical Care Medicine 2000;162(4 part 1):1297‐301. - PubMed
Wilson 2001 {published data only}
-
- Wilson AM, Dempsey OJ, Sims EJ, Lipworth BJ. Evaluation of salmeterol or montelukast as second‐line therapy for asthma not controlled with inhaled corticosteroids. Chest 2001;119(4):1021‐6. - PubMed
Wilson 2001a {published data only}
-
- Wilson AM, Orr LC, Sims EJ, Lipworth BJ. Effects of monotherapy with intra‐nasal corticosteroid or combined oral histamine and leukotriene receptor antagonists in seasonal allergic rhinitis. Clinical & Experimental Allergy 2001;31(1):61‐8. - PubMed
Wilson 2001b {published data only}
-
- Wilson AM, Dempsey OJ, Sims EJ, Lipworth BJ. A comparison of topical budesonide and oral montelukast in seasonal allergic rhinitis and asthma. Clinical & Experimental Allergy 2001;31(4):616‐24. - PubMed
Wilson 2001c {published data only}
-
- Wilson AM, Sims EJ, Orr LC, Coutie WJ, White PS, Gardiner Q, et al. Effects of topical corticosteroid and combined mediator blockade on domiciliary and laboratory measurements of nasal function in seasonal allergic rhinitis. Annals of Allergy, Asthma, & Immunology 2001;87(4):344‐9. - PubMed
Wilson 2004 {published data only}
-
- Wilson A. Are antihistamines useful in managing asthma?. Advanced Studies in Medicine 2004;4(7A):S514‐S519.
Wilson 2010 {published data only}
Wilson 2010a {published data only}
-
- Wilson ECF, Price D, Musgrave SD, Sims EJ, Shepstone L, Murdoch J, et al. Cost effectiveness of leukotriene receptor antagonists versus long‐acting beta‐2 agonists as add‐on therapy to inhaled corticosteroids for asthma: A pragmatic trial. Pharmacoeconomics 2010;28(7):597‐608. - PubMed
Wise 2009 {published data only}
Xiang 2001 {published data only}
-
- Xiang X D, Zhou R, Chen Y. Influence of zafirlukast on pulmonary functions and quality of life in patients with asthma. Bulletin of Hunan Medical University 2001;26(3):251‐3. - PubMed
Yaldiz 2000 {published data only}
-
- Yaldiz E, Özkan G, Özdemir A, Gür A, Çamsari G. The role of montelukast in the reduction of high dose inhaled steroid in asthmatic patients. European Respiratory Journal 2000;16(Supp 31):457s.
Yamamoto 1994 {published data only}
-
- Yamamoto H, Nagata M, Kuramitsu K, Tabe K, Kiuchi H, Sakamoto Y, et al. Inhibition of analgesic‐induced asthma by leukotriene receptor antagonist ONO‐1078. American Journal of Respiratory & Critical Care Medicine 1994;150:254‐7. - PubMed
Yildirim 2001 {published data only}
-
- Yildirim Z, Ozlu T, Bulbul Y. Montelukast and budesonide vs double dose budesonide in moderate asthma. European Respiratory Journal 2001;18(Supp 33):261s.
Yildirim 2004 {published data only}
-
- Yildirim Z, Ozlu T, Bulbul Y, Bayram H. Addition of montelukast versus double dose of inhaled budesonide in moderate persistent asthma. Respirology 2004;9(2):243‐8. - PubMed
Yoo 2001 {published data only}
-
- Yoo SH, Park SH, Song JS, Kang KH, Park CS, Yoo JH, et al. Representing Korea Pranlukast Study Group. Clinical effects of pranlukast, an oral leukotriene receptor antagonist, in mild‐to‐moderate asthma: a 4‐week randomized multicentre controlled trial. Respirology 2001;6(1):15‐21. - PubMed
Yoshida 2000 {published data only}
-
- Yoshida S, Sakamoto H, Ishizaki Y, Onuma K, Shoji T, Nakagawa H, et al. Efficacy of leukotriene receptor antagonist in bronchial hyperresponsiveness and hypersensitivity to analgesic in aspirin‐intolerant asthma. Clinical & Experimental Allergy 2000;30(1):64‐70. - PubMed
Yoshida 2002 {published data only}
-
- Yoshida M, Vethanayagam D, Leigh R, Matsumoto K, Watson RM, Reerich T, et al. A comparison montelukast and budesonide effects on interleukin‐10 profiles in peripheral blood lymphocytes. American Journal of Respiratory and Critical Care Medicine 2002;165(Suppl 8):A217.
Zeiger 2004 {published data only}
-
- Zeiger RS, Baker JW, Kaplan MS, Pearlman DS, Schatz M, Bird S, et al. Variability of symptoms in mild persistent asthma: baseline data from the MIAMI study. Respiratory Medicine 2004;98(9):898‐905. - PubMed
Zeiger 2005a {published data only}
-
- Zeiger RS, Edelman JM, Bird SR, Kaplan MS, Schatz M, Pearlman DS, et al. Short‐term and long‐term asthma control in patients with mild persistent asthma receiving montelukast or fluticasone: a randomised controlled trial [Abstract]. Journal of Allergy & Clinical Immunology 2005;115(Suppl 2):S151. - PubMed
Zeneca Accolate 1998 {published data only}
-
- Zeneca Pharmaceuticals. Zafirlukast (Accolate) product monograph. Zeneca Pharmaceuticals 1998.
Zhang 1999 {unpublished data only}
-
- Zhang J, Chang Y, Reiss TF. Predicting future response using patient baseline variables and early responses to montelukast, a potent cysLT1 antagonist. Merck Research Laboratories 1999.
Zorc 2003 {published data only}
-
- Zorc JJ, Scarfone RJ, Li Y, Hong T, Harmelin M, Grunstein L, et al. Scheduled follow‐up after a pediatric emergency department visit for asthma: A randomized trial. Pediatrics 2003;111(3):495‐502. - PubMed
Additional references
Australia 2006
-
- National Asthma Council. Asthma Management Handbook 2006. Asthma Management Handbook 2006. Vol. 6th edition, Melbourne: National Asthma Campaign, 2006 (http://www.nationalasthma.org.au/cms/images/stories/amh2006_web_5.pdf).
Bisgaard 1988
-
- Bisgaard H, Nielsen MD, Andersen B, Andersen P, Foged N, Fuglsang G, et al. Adrenal function in children with bronchial asthma treated with beclomethasone dipropionate or budesonide. Journal of Allergy & Clinical Immunology 1988;81(6):1088‐95. - PubMed
BTS 2011
-
- Anonymous. British Guidelines on the Management of Asthma. Thorax 2011:1‐64 (http://www.brit‐thoracic.org.uk/Portals/0/Clinical%20Information/Asthma/Guidelines/sign1...). - PubMed
Cates 2002
Deeks 2001
-
- Deeks JJ, Altman DG, Bradburn MJ. Statistical methods for examining heterogeneity and combining results from several studies in meta‐analysis. In: Egger M, Smith GD, Altman DG editor(s). Systematic reviews in health care: meta‐analysis in context. London: BMJ Publishing, 2001:285‐312.
Egger 1997
Elwyn 2010
-
- Elwyn G, Crowe S, Fenton M, Firkins L, Versnel J, Walker S, et al. Identifying and prioritizing uncertainties: patient and clinician engagement in the identification of research questions. Journal of Evaluation in Clinical Practice 2010;16(3):627‐31. - PubMed
GINA 2010
-
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. NIH Publication. Bethesda, MD: National Heart, Lung and Blood Institute, 2002 (http://www.ginasthma.org/pdf/GINA_Report_2010.pdf).
Heuck 1997
-
- Heuck C, Wolthers OD, Hansen M, Kollerup G. Short‐term growth and collagen turnover in asthmatic adolescents treated with the inhaled glucocorticoid budesonide. Steroids 1997;62(10):659‐64. - PubMed
Higgins 2008
-
- Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.1 [updated September 2008]. The Cochrane Collaboration, Available from www.cochrane‐handbook.org, 2008.
Lougheed 2010
Murphy 1993
-
- Murphy S, Kelly HW. Asthma, inflammation, and airway hyper responsiveness in children.Asthma, inflammation, and airway hyper responsiveness in children. Current Opinion in Pediatrics 1993;5(3):255‐65. - PubMed
NAEPP 2007
-
- National Heart, Lung and Blood Institute. National Asthma Education and Prevention Program. NAEPP Expert Panel Report Guidelines for the Diagnosis and Management of Asthma. NIH Publication. National Heart, Lung and Blood Institute. Bethesda, MD: National Institute of Health, 2007 (http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.pdf).
Padfield 1993
-
- Padfield PL, Teelucksingh S. Inhaled corticosteroids: the endocrinologist's view. European Respiratory Review 1993;3(15):494‐500.
Peters‐Golden 2007
-
- Peters‐Golden M, Henderson W. Leukotrienes. New England Journal of Medicine 2007;357:1841‐54. - PubMed
Phillip 1992
-
- Phillip M, Aviram M, Leiberman E, Zadik Z, Giat Y, Levy J, et al. Integrating plasma cortisol concentration in children with asthma receiving long‐term inhaled corticosteroids. Pediatric Pulmonology 1992;12:84‐9. - PubMed
RevMan [Computer program]
-
- Copenhagen, The Nordic Cochrane Centre: The Cochrane Collaboration. Review Manager (RevMan) Version 5.1. Copenhagen, The Nordic Cochrane Centre: The Cochrane Collaboration, 2008.
Richard 2006
-
- Irwin RS, Richardson ND. Side effects with inhaled corticosteroids: The physician's perception. Chest 2006;130:41S‐53S. - PubMed
Sharek 2000
Spahn 1996
-
- Spahn JD, Leung DYM. The role of glucocorticoids in the management of asthma. Allergy & Asthma Proceedings 1996;17(6):341‐50. - PubMed
Todd 1996
-
- Todd G, Dunlop K, McNaboe J, Ryan MF, Carson D, Shields MD. Growth and adrenal suppression in asthmatic children treated with high‐dose fluticasone propionate. Lancet 1996;348:27‐9. - PubMed
Zöllner 2007
-
- Zöllner E. Hypothalamic‐pituitary‐adrenal axis suppression in asthmatic children on inhaled corticosteroids (Part 2)‐‐the risk as determined by gold standard adrenal function tests: a systematic review. Pediatric Allergy and Immunology 2007;18:469‐74. - PubMed
References to other published versions of this review
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

