miR-146a regulates mechanotransduction and pressure-induced inflammation in small airway epithelium
- PMID: 22593544
- PMCID: PMC3405275
- DOI: 10.1096/fj.11-199240
miR-146a regulates mechanotransduction and pressure-induced inflammation in small airway epithelium
Abstract
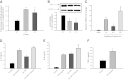
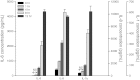
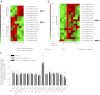
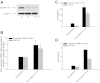
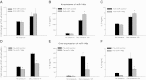
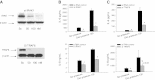
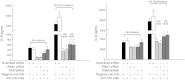
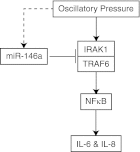
Mechanical ventilation generates biophysical forces, including high transmural pressures, which exacerbate lung inflammation. This study sought to determine whether microRNAs (miRNAs) respond to this mechanical force and play a role in regulating mechanically induced inflammation. Primary human small airway epithelial cells (HSAEpCs) were exposed to 12 h of oscillatory pressure and/or the proinflammatory cytokine TNF-α. Experiments were also conducted after manipulating miRNA expression and silencing the transcription factor NF-κB or toll-like receptor proteins IRAK1 and TRAF6. NF-κB activation, IL-6/IL-8/IL-1β cytokine secretion, miRNA expression, and IRAK1/TRAF6 protein levels were monitored. A total of 12 h of oscillatory pressure and TNF-α resulted in a 5- to 7-fold increase in IL-6/IL-8 cytokine secretion, and oscillatory pressure also resulted in a time-dependent increase in IL-6/IL-8/IL-1β cytokine secretion. Pressure and TNF-α also resulted in distinct patterns of miRNA expression, with miR-146a being the most deregulated miRNA. Manipulating miR-146a expression altered pressure-induced cytokine secretion. Silencing of IRAK1 or TRAF6, confirmed targets of miR-146a, resulted in a 3-fold decrease in pressure-induced cytokine secretion. Cotransfection experiments demonstrate that miR-146a's regulation of pressure-induced cytokine secretion depends on its targeting of both IRAK1 and TRAF6. MiR-146a is a mechanosensitive miRNA that is rapidly up-regulated by oscillatory pressure and plays an important role in regulating mechanically induced inflammation in lung epithelia.
Figures
Similar articles
-
MicroRNA-146a and microRNA-146b regulate human dendritic cell apoptosis and cytokine production by targeting TRAF6 and IRAK1 proteins.J Biol Chem. 2015 Jan 30;290(5):2831-41. doi: 10.1074/jbc.M114.591420. Epub 2014 Dec 11. J Biol Chem. 2015. PMID: 25505246 Free PMC article.
-
Histone deacetylase inhibitors increase microRNA-146a expression and enhance negative regulation of interleukin-1β signaling in osteoarthritis fibroblast-like synoviocytes.Osteoarthritis Cartilage. 2013 Dec;21(12):1987-96. doi: 10.1016/j.joca.2013.09.008. Epub 2013 Oct 6. Osteoarthritis Cartilage. 2013. PMID: 24107356
-
MicroRNA-146a-5p Negatively Regulates Pro-Inflammatory Cytokine Secretion and Cell Activation in Lipopolysaccharide Stimulated Human Hepatic Stellate Cells through Inhibition of Toll-Like Receptor 4 Signaling Pathways.Int J Mol Sci. 2016 Jul 7;17(7):1076. doi: 10.3390/ijms17071076. Int J Mol Sci. 2016. PMID: 27399683 Free PMC article.
-
MiRNA-146a-A Key Player in Immunity and Diseases.Int J Mol Sci. 2023 Aug 14;24(16):12767. doi: 10.3390/ijms241612767. Int J Mol Sci. 2023. PMID: 37628949 Free PMC article. Review.
-
Role of microRNA-146a in normal and malignant hematopoietic stem cell function.Front Genet. 2014 Jul 9;5:219. doi: 10.3389/fgene.2014.00219. eCollection 2014. Front Genet. 2014. PMID: 25071842 Free PMC article. Review.
Cited by
-
MiR-146a alleviates lung injury caused by RSV infection in young rats by targeting TRAF-6 and regulating JNK/ERKMAPK signaling pathways.Sci Rep. 2022 Mar 3;12(1):3481. doi: 10.1038/s41598-022-07346-6. Sci Rep. 2022. PMID: 35241728 Free PMC article.
-
Shear stress upregulates IL-1β secretion by Chlamydia pneumoniae- infected monocytes.Biotechnol Bioeng. 2015 Apr;112(4):838-42. doi: 10.1002/bit.25486. Epub 2015 Feb 20. Biotechnol Bioeng. 2015. PMID: 25336058 Free PMC article.
-
Adipose-derived exosomes protect the pulmonary endothelial barrier in ventilator-induced lung injury by inhibiting the TRPV4/Ca2+ signaling pathway.Am J Physiol Lung Cell Mol Physiol. 2020 Apr 1;318(4):L723-L741. doi: 10.1152/ajplung.00255.2019. Epub 2020 Feb 19. Am J Physiol Lung Cell Mol Physiol. 2020. PMID: 32073873 Free PMC article.
-
Transcriptome-wide analysis of compression-induced microRNA expression alteration in breast cancer for mining therapeutic targets.Oncotarget. 2016 May 10;7(19):27468-78. doi: 10.18632/oncotarget.8322. Oncotarget. 2016. PMID: 27027350 Free PMC article.
-
Effect of miR-146a/bFGF/PEG-PEI Nanoparticles on Inflammation Response and Tissue Regeneration of Human Dental Pulp Cells.Biomed Res Int. 2016;2016:3892685. doi: 10.1155/2016/3892685. Epub 2016 Jan 24. Biomed Res Int. 2016. PMID: 27057540 Free PMC article.
References
-
- Orr A. W., Helmke B. P., Blackman B. R., Schwartz M. A. (2006) Mechanisms of mechanotransduction. Dev. Cell. 10, 11–20 - PubMed
-
- Notter R. H., Finkelstein J. N., Holm B. A., eds. (2005) Lung Injury: Mechanisms, Pathophysiology, and Therapy. Lung Biology in Health and Disease, Vol. 196, Taylor & Francis Group, Boca Raton, FL, USA
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

