Activation of JNK and high expression level of CD133 predict a poor response to sorafenib in hepatocellular carcinoma
- PMID: 22596232
- PMCID: PMC3388555
- DOI: 10.1038/bjc.2012.145
Activation of JNK and high expression level of CD133 predict a poor response to sorafenib in hepatocellular carcinoma
Abstract
Background: Hepatocellular carcinoma (HCC) ranks as the third leading cause of cancer deaths worldwide. While sorafenib, a multikinase inhibitor targeting the Raf/extracellular signal-regulated protein kinase (ERK) pathway, has been shown recently to provide a survival advantage to patients with advanced HCC, a predictive biomarker has not been developed. We studied whether c-Jun N-terminal kinase (JNK), which promotes liver carcinogenesis in mice, affects therapeutic response to sorafenib in HCC patients.
Methods: We collected pathological specimens from 39 patients with advanced HCC before starting sorafenib treatment, and measured JNK activity in HCCs.
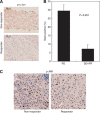
Results: In patients treated with sorafenib, the expression of phospho-c-Jun in HCC, as a read out of JNK activity, was significantly higher (P<0.001) in the non-responder group than in the responder group. c-Jun N-terminal kinase activation in HCC was associated with a decreased time to progression and a poor overall survival (P=0.0028 and P=0.0008, respectively).
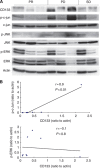
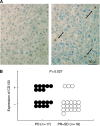
Conclusion: In addition, JNK activity was significantly correlated with CD133 expression level. Correspondingly, high expression level of CD133 was linked to a poor response to sorafenib. Furthermore, D-JNKi, a specific JNK inhibitor, reduced the growth of xenografted CD133(+) cells in athymic mice. In conclusion, JNK activation was positively correlated with CD133 expression level and inversely correlated with the therapeutic response to sorafenib, suggesting that JNK activity may be considered as a new predictive biomarker for response to sorafenib treatment.
Figures



References
-
- Abou-Alfa GK, Schwartz L, Ricci S, Amadori D, Santoro A, Figer A, De Greve J, Douillard JY, Lathia C, Schwartz B, Taylor I, Moscovici M, Saltz LB (2006) Phase II study of sorafenib in patients with advanced hepatocellular carcinoma. J Clin Oncol 24: 4293–4300 - PubMed
-
- Calvisi DF, Ladu S, Gorden A, Farina M, Conner EA, Lee JS, Factor VM, Thorgeirsson SS (2006) Ubiquitous activation of Ras and Jak/Stat pathways in human HCC. Gastroenterology 130: 1117–1128 - PubMed
-
- Chang L, Karin M (2001) Mammalian MAP kinase signalling cascades. Nature 410: 37–40 - PubMed
-
- Dean M, Fojo T, Bates S (2005) Tumour stem cells and drug resistance. Nat Rev Cancer 5: 275–284 - PubMed
-
- Dhillon AS, Hagan S, Rath O, Kolch W (2007) MAP kinase signalling pathways in cancer. Oncogene 26: 3279–3290 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

