Water extract from the leaves of Withania somnifera protect RA differentiated C6 and IMR-32 cells against glutamate-induced excitotoxicity
- PMID: 22606332
- PMCID: PMC3351387
- DOI: 10.1371/journal.pone.0037080
Water extract from the leaves of Withania somnifera protect RA differentiated C6 and IMR-32 cells against glutamate-induced excitotoxicity
Abstract
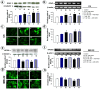
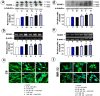
Glutamate neurotoxicity has been implicated in stroke, head trauma, multiple sclerosis and neurodegenerative disorders. Search for herbal remedies that may possibly act as therapeutic agents is an active area of research to combat these diseases. The present study was designed to investigate the neuroprotective role of Withania somnifera (Ashwagandha), also known as Indian ginseng, against glutamate induced toxicity in the retinoic acid differentiated rat glioma (C6) and human neuroblastoma (IMR-32) cells. The neuroprotective activity of the Ashwagandha leaves derived water extract (ASH-WEX) was evaluated. Cell viability and the expression of glial and neuronal cell differentiation markers was examined in glutamate challenged differentiated cells with and without the presence of ASH-WEX. We demonstrate that RA-differentiated C6 and IMR-32 cells, when exposed to glutamate, undergo loss of neural network and cell death that was accompanied by increase in the stress protein HSP70. ASH-WEX pre-treatment inhibited glutamate-induced cell death and was able to revert glutamate-induced changes in HSP70 to a large extent. Furthermore, the analysis on the neuronal plasticity marker NCAM (Neural cell adhesion molecule) and its polysialylated form, PSA-NCAM revealed that ASH-WEX has therapeutic potential for prevention of neurodegeneration associated with glutamate-induced excitotoxicty.
Conflict of interest statement
Figures




Similar articles
-
Withania somnifera water extract as a potential candidate for differentiation based therapy of human neuroblastomas.PLoS One. 2013;8(1):e55316. doi: 10.1371/journal.pone.0055316. Epub 2013 Jan 31. PLoS One. 2013. PMID: 23383150 Free PMC article.
-
Withania somnifera Suppresses Tumor Growth of Intracranial Allograft of Glioma Cells.Mol Neurobiol. 2016 Aug;53(6):4143-4158. doi: 10.1007/s12035-015-9320-1. Epub 2015 Jul 26. Mol Neurobiol. 2016. PMID: 26208698
-
Withania somnifera aqueous extract facilitates the expression and release of GnRH: In vitro and in vivo study.Neurochem Int. 2015 Oct;89:111-9. doi: 10.1016/j.neuint.2015.08.001. Epub 2015 Aug 6. Neurochem Int. 2015. PMID: 26257126
-
Withania somnifera (L.) Dunal - Modern perspectives of an ancient Rasayana from Ayurveda.J Ethnopharmacol. 2021 Jan 10;264:113157. doi: 10.1016/j.jep.2020.113157. Epub 2020 Aug 9. J Ethnopharmacol. 2021. PMID: 32783987 Review.
-
Neurodegenerative diseases and Withania somnifera (L.): An update.J Ethnopharmacol. 2020 Jun 28;256:112769. doi: 10.1016/j.jep.2020.112769. Epub 2020 Mar 30. J Ethnopharmacol. 2020. PMID: 32240781 Review.
Cited by
-
Asymmetrical dimethylarginine antagonizes glutamate-induced apoptosis in PC12 cells.J Mol Neurosci. 2013 Jan;49(1):89-95. doi: 10.1007/s12031-012-9897-z. Epub 2012 Oct 5. J Mol Neurosci. 2013. PMID: 23054590
-
Identification and Functional Characterization of Anti-metastasis and Anti-angiogenic Activities of Triethylene Glycol Derivatives.Front Oncol. 2018 Nov 28;8:552. doi: 10.3389/fonc.2018.00552. eCollection 2018. Front Oncol. 2018. PMID: 30547009 Free PMC article.
-
Indian Ginseng (Withania somnifera) supplementation ameliorates oxidative stress and mitochondrial dysfunctions in experimental model of stroke.Metab Brain Dis. 2018 Aug;33(4):1261-1274. doi: 10.1007/s11011-018-0234-2. Epub 2018 Apr 18. Metab Brain Dis. 2018. PMID: 29671210
-
An updated review on phytochemistry and molecular targets of Withania somnifera (L.) Dunal (Ashwagandha).Front Pharmacol. 2023 Mar 29;14:1049334. doi: 10.3389/fphar.2023.1049334. eCollection 2023. Front Pharmacol. 2023. PMID: 37063285 Free PMC article. Review.
-
Propensity of Withania somnifera to Attenuate Behavioural, Biochemical, and Histological Alterations in Experimental Model of Stroke.Cell Mol Neurobiol. 2016 Oct;36(7):1123-38. doi: 10.1007/s10571-015-0305-4. Epub 2015 Dec 30. Cell Mol Neurobiol. 2016. PMID: 26718711 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

