Lipoic acid metabolism of Plasmodium--a suitable drug target
- PMID: 22607141
- PMCID: PMC3426790
- DOI: 10.2174/138161212801327266
Lipoic acid metabolism of Plasmodium--a suitable drug target
Abstract
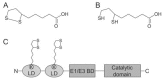
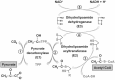
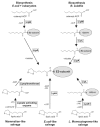
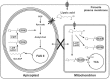
α-Lipoic acid (6,8-thioctic acid; LA) is a vital co-factor of α-ketoacid dehydrogenase complexes and the glycine cleavage system. In recent years it was shown that biosynthesis and salvage of LA in Plasmodium are necessary for the parasites to complete their complex life cycle. LA salvage requires two lipoic acid protein ligases (LplA1 and LplA2). LplA1 is confined to the mitochondrion while LplA2 is located in both the mitochondrion and the apicoplast. LplA1 exclusively uses salvaged LA and lipoylates α-ketoglutarate dehydrogenase, branched chain α-ketoacid dehydrogenase and the H-protein of the glycine cleavage system. LplA2 cannot compensate for the loss of LplA1 function during blood stage development suggesting a specific function for LplA2 that has yet to be elucidated. LA salvage is essential for the intra-erythrocytic and liver stage development of Plasmodium and thus offers great potential for future drug or vaccine development. LA biosynthesis, comprising octanoyl-acyl carrier protein (ACP) : protein N-octanoyltransferase (LipB) and lipoate synthase (LipA), is exclusively found in the apicoplast of Plasmodium where it generates LA de novo from octanoyl-ACP, provided by the type II fatty acid biosynthesis (FAS II) pathway also present in the organelle. LA is the co-factor of the acetyltransferase subunit of the apicoplast located pyruvate dehydrogenase (PDH), which generates acetyl-CoA, feeding into FAS II. LA biosynthesis is not vital for intra-erythrocytic development of Plasmodium, but the deletion of several genes encoding components of FAS II or PDH was detrimental for liver stage development of the parasites indirectly suggesting that the same applies to LA biosynthesis. These data provide strong evidence that LA salvage and biosynthesis are vital for different stages of Plasmodium development and offer potential for drug and vaccine design against malaria.
Figures
Similar articles
-
Conditional knock-out of lipoic acid protein ligase 1 reveals redundancy pathway for lipoic acid metabolism in Plasmodium berghei malaria parasite.Parasit Vectors. 2017 Jun 27;10(1):315. doi: 10.1186/s13071-017-2253-y. Parasit Vectors. 2017. PMID: 28655332 Free PMC article.
-
Apicoplast lipoic acid protein ligase B is not essential for Plasmodium falciparum.PLoS Pathog. 2007 Dec;3(12):e189. doi: 10.1371/journal.ppat.0030189. PLoS Pathog. 2007. PMID: 18069893 Free PMC article.
-
Knockout studies reveal an important role of Plasmodium lipoic acid protein ligase A1 for asexual blood stage parasite survival.PLoS One. 2009;4(5):e5510. doi: 10.1371/journal.pone.0005510. Epub 2009 May 12. PLoS One. 2009. PMID: 19434237 Free PMC article.
-
Plasmodium falciparum possesses organelle-specific alpha-keto acid dehydrogenase complexes and lipoylation pathways.Biochem Soc Trans. 2005 Nov;33(Pt 5):977-80. doi: 10.1042/BST20050977. Biochem Soc Trans. 2005. PMID: 16246025 Review.
-
Plasmodium falciparum: organelle-specific acquisition of lipoic acid.Int J Biochem Cell Biol. 2009 Apr;41(4):748-52. doi: 10.1016/j.biocel.2008.10.028. Epub 2008 Nov 5. Int J Biochem Cell Biol. 2009. PMID: 19027872 Review.
Cited by
-
Cluster analysis of Plasmodium RNA-seq time-course data identifies stage-specific co-regulated biological processes and regulatory elements.F1000Res. 2016 Aug 8;5:ISCB Comm J-1932. doi: 10.12688/f1000research.9093.1. eCollection 2016. F1000Res. 2016. PMID: 27990252 Free PMC article.
-
Roles of Ferredoxin-Dependent Proteins in the Apicoplast of Plasmodium falciparum Parasites.mBio. 2021 Feb 22;13(1):e0302321. doi: 10.1128/mbio.03023-21. Epub 2022 Feb 15. mBio. 2021. PMID: 35164549 Free PMC article.
-
Dynamic Relay of Protein-Bound Lipoic Acid in Staphylococcus aureus.J Bacteriol. 2019 Oct 21;201(22):e00446-19. doi: 10.1128/JB.00446-19. Print 2019 Nov 15. J Bacteriol. 2019. PMID: 31451544 Free PMC article.
-
Biochemical and structural characterization of the apicoplast dihydrolipoamide dehydrogenase of Plasmodium falciparum.Biosci Rep. 2015 Jan 14;35(1):e00171. doi: 10.1042/BSR20140150. Biosci Rep. 2015. PMID: 25387830 Free PMC article.
-
Conditional knock-out of lipoic acid protein ligase 1 reveals redundancy pathway for lipoic acid metabolism in Plasmodium berghei malaria parasite.Parasit Vectors. 2017 Jun 27;10(1):315. doi: 10.1186/s13071-017-2253-y. Parasit Vectors. 2017. PMID: 28655332 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
