Untethering the nuclear envelope and cytoskeleton: biologically distinct dystonias arising from a common cellular dysfunction
- PMID: 22611399
- PMCID: PMC3352338
- DOI: 10.1155/2012/634214
Untethering the nuclear envelope and cytoskeleton: biologically distinct dystonias arising from a common cellular dysfunction
Abstract
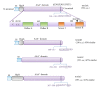
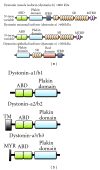
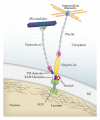
Most cases of early onset DYT1 dystonia in humans are caused by a GAG deletion in the TOR1A gene leading to loss of a glutamic acid (ΔE) in the torsinA protein, which underlies a movement disorder associated with neuronal dysfunction without apparent neurodegeneration. Mutation/deletion of the gene (Dst) encoding dystonin in mice results in a dystonic movement disorder termed dystonia musculorum, which resembles aspects of dystonia in humans. While torsinA and dystonin proteins do not share modular domain architecture, they participate in a similar function by modulating a structural link between the nuclear envelope and the cytoskeleton in neuronal cells. We suggest that through a shared interaction with the nuclear envelope protein nesprin-3α, torsinA and the neuronal dystonin-a2 isoform comprise a bridge complex between the outer nuclear membrane and the cytoskeleton, which is critical for some aspects of neuronal development and function. Elucidation of the overlapping roles of torsinA and dystonin-a2 in nuclear/endoplasmic reticulum dynamics should provide insights into the cellular mechanisms underlying the dystonic phenotype.
Figures
References
-
- Fahn S. Concept and classification of dystonia. Advances in Neurology. 1988;50:1–8. - PubMed
-
- Geyer HL, Bressman SB. The diagnosis of dystonia. The Lancet Neurology. 2006;5(9):780–790. - PubMed
-
- Brüggemann N, Klein C. Genetics of primary torsion dystonia. Current Neurology and Neuroscience Reports. 2010;10(3):199–206. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

