Pin1 protein regulates Smad protein signaling and pulmonary fibrosis
- PMID: 22613712
- PMCID: PMC3390608
- DOI: 10.1074/jbc.M111.313684
Pin1 protein regulates Smad protein signaling and pulmonary fibrosis
Abstract
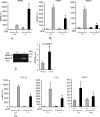
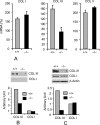
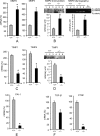
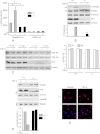
Interstitial pulmonary fibrosis is caused by the excess production of extracellular matrix (ECM) by Fb in response to TGF-β1. Here, we show that the peptidyl-prolyl isomerase Pin1 modulates the production of many pro- and antifibrogenic cytokines and ECM. After acute, bleomycin injury, Pin1(-/-) mice showed reduced, pulmonary expression of collagens, tissue inhibitors of metalloproteinases, and fibrogenic cytokines but increased matrix metalloproteinases, compared with WT mice, despite similar levels of inflammation. In primary fibroblasts, Pin1 was required for TGF-β-induced phosphorylation, nuclear translocation, and transcriptional activity of Smad3. In Pin1(-/-) cells, inhibitory Smad6 was found in the cytoplasm rather than nucleus. Smad6 knockdown in Pin1(-/-) fibroblasts restored TGF-β-induced Smad3 activation, translocation, and target gene expression. Therefore, Pin1 is essential for normal Smad6 function and ECM production in response to injury or TGF-β and thus may be an attractive therapeutic target to prevent excess scarring in diverse lung diseases.
Figures

Similar articles
-
Pin1 induction in the fibrotic liver and its roles in TGF-β1 expression and Smad2/3 phosphorylation.J Hepatol. 2014 Jun;60(6):1235-41. doi: 10.1016/j.jhep.2014.02.004. Epub 2014 Feb 13. J Hepatol. 2014. PMID: 24530597
-
Pin1 down-regulates transforming growth factor-beta (TGF-beta) signaling by inducing degradation of Smad proteins.J Biol Chem. 2009 Mar 6;284(10):6109-15. doi: 10.1074/jbc.M804659200. Epub 2009 Jan 4. J Biol Chem. 2009. PMID: 19122240
-
Prolyl isomerase Pin1 promotes extracellular matrix production in hepatic stellate cells through regulating formation of the Smad3-TAZ complex.Exp Cell Res. 2023 Apr 15;425(2):113544. doi: 10.1016/j.yexcr.2023.113544. Epub 2023 Mar 9. Exp Cell Res. 2023. PMID: 36906101
-
p90RSK Inhibition Ameliorates TGF-β1 Signaling and Pulmonary Fibrosis by Inhibiting Smad3 Transcriptional Activity.Cell Physiol Biochem. 2020 Feb 22;54(2):195-210. doi: 10.33594/000000214. Cell Physiol Biochem. 2020. PMID: 32083406
-
Pin1 promotes transforming growth factor-beta-induced migration and invasion.J Biol Chem. 2010 Jan 15;285(3):1754-64. doi: 10.1074/jbc.M109.063826. Epub 2009 Nov 17. J Biol Chem. 2010. PMID: 19920136 Free PMC article.
Cited by
-
Manipulating PML SUMOylation via Silencing UBC9 and RNF4 Regulates Cardiac Fibrosis.Mol Ther. 2017 Mar 1;25(3):666-678. doi: 10.1016/j.ymthe.2016.12.021. Epub 2017 Jan 28. Mol Ther. 2017. PMID: 28143738 Free PMC article.
-
Peptidyl-prolyl cis-trans isomerase NIMA interacting 1 regulates skeletal muscle fusion through structural modification of Smad3 in the linker region.J Cell Physiol. 2018 Dec;233(12):9390-9403. doi: 10.1002/jcp.26774. Epub 2018 Aug 21. J Cell Physiol. 2018. PMID: 30132832 Free PMC article.
-
Lithium Attenuates TGF-β(1)-Induced Fibroblasts to Myofibroblasts Transition in Bronchial Fibroblasts Derived from Asthmatic Patients.J Allergy (Cairo). 2012;2012:206109. doi: 10.1155/2012/206109. Epub 2012 Sep 3. J Allergy (Cairo). 2012. PMID: 22988467 Free PMC article.
-
The molecular mechanisms of peptidyl-prolyl cis/trans isomerase Pin1 and its relevance to kidney disease.Front Pharmacol. 2024 Apr 22;15:1373446. doi: 10.3389/fphar.2024.1373446. eCollection 2024. Front Pharmacol. 2024. PMID: 38711994 Free PMC article. Review.
-
TGF-β Signaling from Receptors to Smads.Cold Spring Harb Perspect Biol. 2016 Sep 1;8(9):a022061. doi: 10.1101/cshperspect.a022061. Cold Spring Harb Perspect Biol. 2016. PMID: 27449815 Free PMC article. Review.
References
-
- Laurent G. J. (1987) Dynamic state of collagen: pathways of collagen degradation in vivo and their possible role in regulation of collagen mass. Am. J. Physiol. 252, C1–C9 - PubMed
-
- Uhal B. D., Kim J. K., Li X., Molina-Molina M. (2007) Angiotensin-TGF-β1 cross-talk in human idiopathic pulmonary fibrosis. Autocrine mechanisms in myofibroblasts and macrophages. Curr. Pharm. Des. 13, 1247–1256 - PubMed
-
- Zheng L., Walters E. H., Wang N., Whitford H., Orsida B., Levvey B., Bailey M., Williams T. J., Snell G. I. (2004) Effect of inhaled fluticasone propionate on BAL TGF-β1 and bFGF concentrations in clinically stable lung transplant recipients. J. Heart Lung Transplant. 23, 446–455 - PubMed
-
- Halwani R., Al-Muhsen S., Al-Jahdali H., Hamid Q. (2011) Role of transforming growth factor-β in airway remodeling in asthma. Am. J. Respir. Cell Mol. Biol. 44, 127–133 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

