Regulation of calbindin-D(28k) expression by Msx2 in the dental epithelium
- PMID: 22614360
- PMCID: PMC3460364
- DOI: 10.1369/0022155412450641
Regulation of calbindin-D(28k) expression by Msx2 in the dental epithelium
Abstract
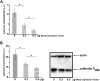
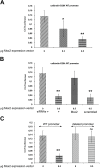
Amelogenesis involves the coordinated expression of a set of molecules that includes enamel matrix proteins and calcium-binding proteins. Msx2 is a member of the divergent homeobox gene family and is instrumental in dental morphogenesis and biomineralization. This study focused on an EF-hand calcium-binding protein, calbindin-D(28k), which is highly expressed in dental epithelium. In vivo data showed that calbindin-D(28k) levels were higher in ameloblasts from Msx2(+/-) mice than Msx2(+/+) mice. Consistent with this finding, calbindin-D(28k) distribution was affected in transgenic mice with ectopic expression in root epithelium in rests of Malassez in Msx2(+/-) and more clearly in Msx2(-/-) mice. In accordance with these in vivo data, calbindin-D(28k) protein and mRNA levels were decreased in LS8 ameloblast-like cells by exogenous Msx2 overexpression. Furthermore, calbindin-D(28k) promoter activity (nt-1075/+34) was specifically diminished in the presence of Msx2 overexpression, showing that Msx2 behave as a transcriptional repressor for calbindin-D(28k) gene expression. In conclusion, Msx2 may control the spatiotemporally restricted frame of calbindin-D(28k) production in the dental epithelium in relation to enamel mineralization, as previously shown for amelogenin.
Conflict of interest statement
Figures

Similar articles
-
MSX2 in ameloblast cell fate and activity.Front Physiol. 2015 Jan 5;5:510. doi: 10.3389/fphys.2014.00510. eCollection 2014. Front Physiol. 2015. PMID: 25601840 Free PMC article. Review.
-
In situ hybridization of calbindin-D 28 k transcripts in undecalcified sections of the rat continuously erupting incisor.Connect Tissue Res. 1995;32(1-4):137-43. doi: 10.3109/03008209509013716. Connect Tissue Res. 1995. PMID: 7554910
-
Cytoprotective effects of calbindin-D(28k) against antimycin-A induced hypoxic injury in proximal tubular cells.Life Sci. 2002 Jun 21;71(5):559-69. doi: 10.1016/s0024-3205(02)01710-1. Life Sci. 2002. PMID: 12052440
-
Calbindin-D28k is expressed in osteoblastic cells and suppresses their apoptosis by inhibiting caspase-3 activity.J Biol Chem. 2000 Aug 25;275(34):26328-32. doi: 10.1074/jbc.M003600200. J Biol Chem. 2000. PMID: 10835428
-
Parvalbumin and calbindin D-28k immunoreactivity in transgenic mice with a G93A mutant SOD1 gene.Brain Res. 2006 Apr 14;1083(1):196-203. doi: 10.1016/j.brainres.2006.01.129. Epub 2006 Mar 20. Brain Res. 2006. PMID: 16546142
Cited by
-
Unraveling the transcriptional regulation of TWIST1 in limb development.PLoS Genet. 2018 Oct 29;14(10):e1007738. doi: 10.1371/journal.pgen.1007738. eCollection 2018 Oct. PLoS Genet. 2018. PMID: 30372441 Free PMC article.
-
Enam expression is regulated by Msx2.Dev Dyn. 2023 Oct;252(10):1292-1302. doi: 10.1002/dvdy.598. Epub 2023 May 16. Dev Dyn. 2023. PMID: 37191055 Free PMC article.
-
Transcriptome sequencing and analysis for the pigmentation of scale and skin in common carp (Cyprinus carpio).Mol Biol Rep. 2021 Mar;48(3):2399-2410. doi: 10.1007/s11033-021-06273-5. Epub 2021 Mar 19. Mol Biol Rep. 2021. PMID: 33742327
-
MSX2 in ameloblast cell fate and activity.Front Physiol. 2015 Jan 5;5:510. doi: 10.3389/fphys.2014.00510. eCollection 2014. Front Physiol. 2015. PMID: 25601840 Free PMC article. Review.
-
Temporospatial expression of neurogranin in the rat molar development.J Mol Histol. 2025 Mar 29;56(2):123. doi: 10.1007/s10735-025-10401-x. J Mol Histol. 2025. PMID: 40156717
References
-
- Aïoub M, Lézot F, Molla M, Castaneda B, Robert B, Goubin G, Nefussi JR, Berdal A. 2007. Msx2 −/– transgenic mice develop compound amelogenesis imperfecta, dentinogenesis imperfecta and periodental osteopetrosis. Bone. 41:851–859 - PubMed
-
- Bei M, Stowell S, Maas R. 2004. Msx2 controls ameloblast terminal differentiation. Dev Dyn. 231:758–765 - PubMed
-
- Bendall AJ, Abate-Shen C. 2000. Roles for Msx and Dlx homeoproteins in vertebrate development. Gene. 247:17–31 - PubMed
-
- Berdal A, Hotton D, Pike JW, Mathieu H, Dupret JM. 1993. Cell- and stage-specific expression of vitamin D receptor and calbindin genes in rat incisor: regulation by 1,25-dihydroxyvitamin D3. Dev Biol. 155:172–179 - PubMed
-
- Berdal A, Hotton D, Saffar JL, Thomasset M, Nanci A. 1996. Calbindin-D9k and calbindin-D28k expression in rat mineralized tissues in vivo. J Bone Min Res. 11:768–779 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

