Regulation of lymphocyte function by ORAI and STIM proteins in infection and autoimmunity
- PMID: 22615435
- PMCID: PMC3473275
- DOI: 10.1113/jphysiol.2012.233221
Regulation of lymphocyte function by ORAI and STIM proteins in infection and autoimmunity
Abstract
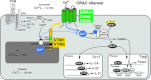
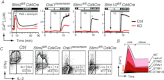
Store-operated Ca(2+) entry (SOCE) in cells of the immune system is mediated by Ca(2+) release-activated Ca(2+) (CRAC) channels that are formed by ORAI1 and its homologues ORAI2 and ORAI3. They are activated by stromal interaction molecules (STIM) 1 and 2 in response to depletion of endoplasmic reticulum Ca(2+) stores. Loss-of-function mutations in the human ORAI1 and STIM1 genes abolish CRAC channel function and SOCE in a variety of non-excitable cells including lymphocytes and other immune cells, resulting in a unique clinical syndrome termed CRAC channelopathy. It is dominated by severe immunodeficiency and autoimmunity due to impaired SOCE and defects in the function of several lymphocyte subsets. These include CD8(+) T cells, CD4(+) effector and regulatory T cells, natural killer (NK) cells and B cells. This review provides a concise discussion of the role of CRAC channels in these lymphocyte populations and the regulation of adaptive immune responses to infection, in autoimmunity and inflammation.
Figures

Similar articles
-
STIM and ORAI proteins in the nervous system.Channels (Austin). 2015;9(5):245-52. doi: 10.1080/19336950.2015.1071747. Epub 2015 Jul 28. Channels (Austin). 2015. PMID: 26218135 Free PMC article. Review.
-
Immunodeficiency due to mutations in ORAI1 and STIM1.Clin Immunol. 2010 May;135(2):169-82. doi: 10.1016/j.clim.2010.01.011. Epub 2010 Mar 1. Clin Immunol. 2010. PMID: 20189884 Free PMC article. Review.
-
Diseases caused by mutations in ORAI1 and STIM1.Ann N Y Acad Sci. 2015 Nov;1356(1):45-79. doi: 10.1111/nyas.12938. Epub 2015 Oct 15. Ann N Y Acad Sci. 2015. PMID: 26469693 Free PMC article. Review.
-
ORAI1 mutations abolishing store-operated Ca2+ entry cause anhidrotic ectodermal dysplasia with immunodeficiency.J Allergy Clin Immunol. 2018 Oct;142(4):1297-1310.e11. doi: 10.1016/j.jaci.2017.10.031. Epub 2017 Nov 16. J Allergy Clin Immunol. 2018. PMID: 29155098 Free PMC article.
-
Ca2+ Signaling but Not Store-Operated Ca2+ Entry Is Required for the Function of Macrophages and Dendritic Cells.J Immunol. 2015 Aug 1;195(3):1202-17. doi: 10.4049/jimmunol.1403013. Epub 2015 Jun 24. J Immunol. 2015. PMID: 26109647 Free PMC article.
Cited by
-
Autophagy, a novel pathway to regulate calcium mobilization in T lymphocytes.Front Immunol. 2013 Jul 4;4:179. doi: 10.3389/fimmu.2013.00179. eCollection 2013. Front Immunol. 2013. PMID: 23847620 Free PMC article.
-
Store-operated Ca2+ Entry Modulates the Expression of Enamel Genes.J Dent Res. 2015 Oct;94(10):1471-7. doi: 10.1177/0022034515598144. Epub 2015 Jul 31. J Dent Res. 2015. PMID: 26232387 Free PMC article.
-
Molecular pharmacology of store-operated CRAC channels.Channels (Austin). 2013 Sep-Oct;7(5):402-14. doi: 10.4161/chan.25292. Epub 2013 Aug 26. Channels (Austin). 2013. PMID: 23807116 Free PMC article. Review.
-
Ca2+ Signaling in Exocrine Cells.Cold Spring Harb Perspect Biol. 2020 May 1;12(5):a035279. doi: 10.1101/cshperspect.a035279. Cold Spring Harb Perspect Biol. 2020. PMID: 31636079 Free PMC article. Review.
-
Identification of Stim1 as a candidate gene for exaggerated sympathetic response to stress in the stroke-prone spontaneously hypertensive rat.PLoS One. 2014 Apr 15;9(4):e95091. doi: 10.1371/journal.pone.0095091. eCollection 2014. PLoS One. 2014. PMID: 24736434 Free PMC article.
References
-
- Baba Y, Nishida K, Fujii Y, Hirano T, Hikida M, Kurosaki T. Essential function for the calcium sensor STIM1 in mast cell activation and anaphylactic responses. Nat Immunol. 2008;9:81–88. - PubMed
-
- Baine I, Abe BT, Macian F. Regulation of T-cell tolerance by calcium/NFATsignaling. Immunol Rev. 2009;231:225–240. - PubMed
-
- Beyersdorf N, Braun A, Vogtle T, Varga-Szabo D, Galdos RR, Kissler S, Kerkau T, Nieswandt B. STIM1-independent T cell development and effector function in vivo. J Immunol. 2009;182:3390–3397. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

