Normal leptin expression, lower adipogenic ability, decreased leptin receptor and hyposensitivity to Leptin in Adolescent Idiopathic Scoliosis
- PMID: 22615788
- PMCID: PMC3352937
- DOI: 10.1371/journal.pone.0036648
Normal leptin expression, lower adipogenic ability, decreased leptin receptor and hyposensitivity to Leptin in Adolescent Idiopathic Scoliosis
Abstract
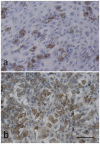
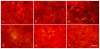
Leptin has been suggested to play a role in the etiology of Adolescent Idiopathic Scoliosis (AIS), however, the leptin levels in AIS girls are still a discrepancy, and no in vitro study of leptin in AIS is reported. We took a series of case-control studies, trying to understand whether Leptin gene polymorphisms are involved in the etiology of the AIS or the change in leptin level is a secondary event, to assess the level of leptin receptor, and to evaluate the differences of response to leptin between AIS cases and controls. We screened all exons of Leptin gene in 45 cases and 45 controls and selected six tag SNPs to cover all the observed variations. Association analysis in 446 AIS patients and 550 healthy controls showed no association between the polymorphisms of Leptin gene and susceptibility/severity to AIS. Moreover, adipogenesis assay of bone mesenchymal stem cells (MSCs) suggested that the adipogenic ability of MSCs from AIS girls was lower than controls. After adjusting the differentiation rate, expressions of leptin and leptin receptor were similar between two groups. Meanwhile, osteogenesis assay of MSC showed the leptin level was similar after adjusting the differentiation rate, but the leptin receptor level was decreased in induced AIS osteoblasts. Immunocytochemistry and western blot analysis showed less leptin receptors expressed in AIS group. Furthermore, factorial designed studies with adipogenesis and osteogenesis revealed that the MSCs from patients have no response to leptin treatment. Our results suggested that Leptin gene variations are not associated with AIS and low serum leptin probably is a secondary outcome which may be related to the low capability of adipogenesis in AIS. The decreased leptin receptor levels may lead to the hyposensitivity to leptin. These findings implied that abnormal peripheral leptin signaling plays an important role in the pathological mechanism of AIS.
Conflict of interest statement
Figures






References
-
- Wang WJ, Yeung HY, Chu WC, Tang NL, Lee KM, et al. Top theories for the etiopathogenesis of adolescent idiopathic scoliosis. J Pediatr Orthop. 2011;31:S14–S27. - PubMed
-
- Kouwenhoven JW, Castelein RM. The pathogenesis of adolescent idiopathic scoliosis: review of the literature. Spine (Phila Pa 1976) 2008;33:2898–2908. - PubMed
-
- Lombardi G, Akoume MY, Colombini A, Moreau A, Banfi G. Biochemistry of adolescent idiopathic scoliosis. Adv Clin Chem. 2011;54:165–182. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

