Estrogen protects renal endothelial barrier function from ischemia-reperfusion in vitro and in vivo
- PMID: 22622457
- PMCID: PMC3774263
- DOI: 10.1152/ajprenal.00354.2011
Estrogen protects renal endothelial barrier function from ischemia-reperfusion in vitro and in vivo
Abstract
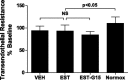
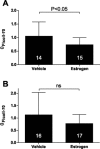
Emerging evidence suggests that renal endothelial function may be altered in ischemia-reperfusion injury. Acute kidney injury is sexually dimorphic, and estrogen protects renal tubular function after experimental ischemic injury. This study tested the hypothesis that during ischemia-reperfusion, estrogen alters glomerular endothelial function to prevent hyperpermeability. Glomerular endothelial cells were exposed to 8-h oxygen-glucose deprivation (OGD) followed by 4- and 8-h reoxygenation-glucose repletion. After 4-h reoxygenation-glucose repletion, transendothelial permeability to Ficoll-70 was reduced, and transendothelial resistance increased, by 17β-estradiol vs. vehicle treatment during OGD (OGD-vehicle: 91.0 ± 11.8%, OGD-estrogen: 102.6 ± 10.8%, P < 0.05). This effect was reversed by coadministration of G protein-coupled receptor 30 (GPR30) antagonist G15 with 17β-estradiol (OGD-estrogen-G15: 89.5 ± 6.9, P < 0.05 compared with 17β-estradiol). To provide preliminary confirmation of this result in vivo, Ficoll-70 was administered to mice 24 h after cardiac arrest and cardiopulmonary resuscitation (CA/CPR). Blood urea nitrogen (BUN) and serum creatinine (SCr) in these mice were elevated within 12 h following CA/CPR and reduced at 24 h by pretreatment with 17β-estradiol (BUN/SCr 17β-estradiol: 34 ± 19/0.2 ± 0.1 vehicle: 92 ± 49/0.5 ± 0.3, n = 8-12, P < 0.05). Glomerular sieving of Ficoll 70 was increased by CA/CPR within 2 h of injury and 17β-estradiol treatment (θ; 17β-estradiol: 0.74 ± 0.26 vs. vehicle: 1.05 ± 0.53, n = 14-15, P < 0.05). These results suggest that estrogen reduces postischemic glomerular endothelial hyperpermeability at least in part through GPR30 and that estrogen may regulate post CA/CPR glomerular permeability in a similar fashion in vivo.
Figures







References
-
- Akis N, Madaio MP. Isolation, culture, and characterization of endothelial cells from mouse glomeruli. Kidney Int 65: 2223–2227, 2004 - PubMed
-
- Andersson M, Nilsson U, Hjalmarsson C, Haraldsson B, Nystrom JS. Mild renal ischemia-reperfusion reduces charge and size selectivity of the glomerular barrier. Am J Physiol Renal Physiol 292: F1802–F1809, 2007 - PubMed
-
- Ardelt AA, McCullough LD, Korach KS, Wang MM, Munzenmaier DH, Hurn PD. Estradiol regulates angiopoietin-1 mRNA expression through estrogen receptor-alpha in a rodent experimental stroke model. Stroke 36: 337–341, 2005 - PubMed
-
- Basile DP. Rarefaction of peritubular capillaries following ischemic acute renal failure: a potential factor predisposing to progressive nephropathy. Curr Opin Nephrol Hypertens 13: 1–7, 2004 - PubMed
-
- Basile DP, Friedrich JL, Spahic J, Knipe N, Mang H, Leonard EC, Changizi-Ashtiyani S, Bacallao RL, Molitoris BA, Sutton TA. Impaired endothelial proliferation and mesenchymal transition contribute to vascular rarefaction following acute kidney injury. Am J Physiol Renal Physiol 300: F721–F733, 2011 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

