Purification and characterization of a novel hypersensitive response-inducing elicitor from Magnaporthe oryzae that triggers defense response in rice
- PMID: 22624059
- PMCID: PMC3356297
- DOI: 10.1371/journal.pone.0037654
Purification and characterization of a novel hypersensitive response-inducing elicitor from Magnaporthe oryzae that triggers defense response in rice
Abstract
Background: Magnaporthe oryzae, the rice blast fungus, might secrete certain proteins related to plant-fungal pathogen interactions.
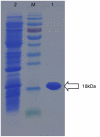
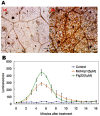
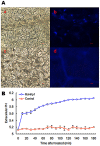
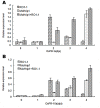
Methodology/principal findings: In this study, we report the purification, characterization, and gene cloning of a novel hypersensitive response-inducing protein elicitor (MoHrip1) secreted by M. oryzae. The protein fraction was purified and identified by de novo sequencing, and the sequence matched the genomic sequence of a putative protein from M. oryzae strain 70-15 (GenBank accession No. XP_366602.1). The elicitor-encoding gene mohrip1 was isolated; it consisted of a 429 bp cDNA, which encodes a polypeptide of 142 amino acids with a molecular weight of 14.322 kDa and a pI of 4.53. The deduced protein, MoHrip1, was expressed in E. coli. And the expression protein collected from bacterium also forms necrotic lesions in tobacco. MoHrip1 could induce the early events of the defense response, including hydrogen peroxide production, callose deposition, and alkalization of the extracellular medium, in tobacco. Moreover, MoHrip1-treated rice seedlings possessed significantly enhanced systemic resistance to M. oryzae compared to the control seedlings. The real-time PCR results indicated that the expression of some pathogenesis-related genes and genes involved in signal transduction could also be induced by MoHrip1.
Conclusion/significance: The results demonstrate that MoHrip1 triggers defense responses in rice and could be used for controlling rice blast disease.
Conflict of interest statement
Figures



Similar articles
-
A novel elicitor identified from Magnaporthe oryzae triggers defense responses in tobacco and rice.Plant Cell Rep. 2014 Nov;33(11):1865-79. doi: 10.1007/s00299-014-1663-y. Epub 2014 Jul 24. Plant Cell Rep. 2014. PMID: 25056480
-
The Secreted Protein MoHrip1 Is Necessary for the Virulence of Magnaporthe oryzae.Int J Mol Sci. 2019 Apr 2;20(7):1643. doi: 10.3390/ijms20071643. Int J Mol Sci. 2019. PMID: 30987045 Free PMC article.
-
The Magnaporthe oryzae Alt A 1-like protein MoHrip1 binds to the plant plasma membrane.Biochem Biophys Res Commun. 2017 Oct 7;492(1):55-60. doi: 10.1016/j.bbrc.2017.08.039. Epub 2017 Aug 12. Biochem Biophys Res Commun. 2017. PMID: 28807829
-
The purification and characterization of a novel hypersensitive-like response-inducing elicitor from Verticillium dahliae that induces resistance responses in tobacco.Appl Microbiol Biotechnol. 2012 Jan;93(1):191-201. doi: 10.1007/s00253-011-3405-1. Epub 2011 Jun 21. Appl Microbiol Biotechnol. 2012. PMID: 21691787
-
Every Coin Has Two Sides: Reactive Oxygen Species during Rice⁻Magnaporthe oryzae Interaction.Int J Mol Sci. 2019 Mar 8;20(5):1191. doi: 10.3390/ijms20051191. Int J Mol Sci. 2019. PMID: 30857220 Free PMC article. Review.
Cited by
-
Apoplastic Cell Death-Inducing Proteins of Filamentous Plant Pathogens: Roles in Plant-Pathogen Interactions.Front Genet. 2020 Jun 26;11:661. doi: 10.3389/fgene.2020.00661. eCollection 2020. Front Genet. 2020. PMID: 32676100 Free PMC article. Review.
-
Enhanced Nicotiana benthamiana immune responses caused by heterologous plant genes from Pinellia ternata.BMC Plant Biol. 2018 Dec 17;18(1):357. doi: 10.1186/s12870-018-1598-5. BMC Plant Biol. 2018. PMID: 30558544 Free PMC article.
-
Inducible and constitutive expression of an elicitor gene Hrip1 from Alternaria tenuissima enhances stress tolerance in Arabidopsis.Transgenic Res. 2015 Feb;24(1):135-45. doi: 10.1007/s11248-014-9824-x. Epub 2014 Aug 14. Transgenic Res. 2015. PMID: 25120219
-
PeBL1, a novel protein elicitor from Brevibacillus laterosporus strain A60, activates defense responses and systemic resistance in Nicotiana benthamiana.Appl Environ Microbiol. 2015 Apr;81(8):2706-16. doi: 10.1128/AEM.03586-14. Epub 2015 Feb 6. Appl Environ Microbiol. 2015. PMID: 25662975 Free PMC article.
-
Secreted Alpha-N-Arabinofuranosidase B Protein Is Required for the Full Virulence of Magnaporthe oryzae and Triggers Host Defences.PLoS One. 2016 Oct 20;11(10):e0165149. doi: 10.1371/journal.pone.0165149. eCollection 2016. PLoS One. 2016. PMID: 27764242 Free PMC article.
References
-
- Panstruga R, Parker JE, Schulze-Lefert P. SnapShot: Plant immune response pathways. Cell. 2009;136:978 e971–973. - PubMed
-
- Cui H, Xiang T, Zhou JM. Plant immunity: a lesson from pathogenic bacterial effector proteins. Cell Microbiol. 2009;11:1453–1461. - PubMed
-
- Nürnberger T, Brunner F. Innate immunity in plants and animals: emerging parallels between the recognition of general elicitors and pathogen-associated molecular patterns. Current Opinion in Plant Biology. 2002;5:318–324. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

