Tissue interactions between craniosynostotic dura mater and bone
- PMID: 22627405
- PMCID: PMC3360881
- DOI: 10.1097/SCS.0b013e31824e645f
Tissue interactions between craniosynostotic dura mater and bone
Abstract
Background: Cells within the dura mater have been implicated in the determination of suture patency and fusion. Craniosynostosis (CS), the premature fusion of 1 or more of the cranial sutures, could result from abnormal control over the differentiation of osteoprogenitor cells from the dura mater. This study tested whether dura mater cells derived from rabbits with congenital CS were different from cells derived from normal rabbits and investigated the effects that CS dura mater had on osteogenic differentiation in vitro and in vivo.
Methods: Cells were derived from the dura mater from wild-type rabbits (WT; n = 23) or CS rabbits (n = 16). Cells were stimulated with bone morphogenetic protein 4, and alkaline phosphatase (ALP) expression and cell proliferation were assessed. Dura mater-derived cells were also cocultured with primary rabbit bone-derived cells, and ALP was assessed. Finally, interactions between the dura mater and overlying tissues were manipulated in vivo.
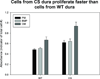
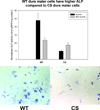
Results: Craniosynostotic dura mater-derived cells proliferated faster than did WT cells but were not more ALP positive. Coculture experiments showed that CS dura mater cells induced increased ALP activity in CS bone-derived cells, but not in WT bone-derived cells. In vivo experiments showed that a physical barrier successfully inhibited dura mater-derived osteogenesis.
Conclusions: Coculture of CS bone- and CS dura mater-derived cells evoked an abnormal phenotype in vitro. Covering the CS dura mater led to decreased bone formation in vivo. Further investigations will focus on the signaling molecules involved in the communication between these 2 CS tissue types in vitro and in vivo.
Conflict of interest statement
All authors have no conflicts of interest.
Figures



References
-
- Kim HJ, Rice DP, Kettunen PJ, Thesleff I. FGF-, BMP- and Shh-mediated signalling pathways in the regulation of cranial suture morphogenesis and calvarial bone development. Development. 1998;125(7):1241–1251. - PubMed
-
- Rice DP, Aberg T, Chan Y, Tang Z, Kettunen PJ, Pakarinen L, Maxson RE, Thesleff I. Integration of FGF and TWIST in calvarial bone and suture development. Development. 2000;127(9):1845–1855. - PubMed
-
- Rice DP, Rice R, Thesleff I. Molecular mechanisms in calvarial bone and suture development, and their relation to craniosynostosis. Eur J Orthod. 2003;25(2):139–148. - PubMed
-
- Opperman LA, Nolen AA, Ogle RC. TGF-beta 1, TGF-beta 2, and TGF-beta 3 exhibit distinct patterns of expression during cranial suture formation and obliteration in vivo and in vitro. J Bone Miner Res. 1997;12(3):301–310. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

