Intrinsic signal optical imaging evidence for dorsal V3 in the prosimian galago (Otolemur garnettii)
- PMID: 22628051
- PMCID: PMC3593310
- DOI: 10.1002/cne.23154
Intrinsic signal optical imaging evidence for dorsal V3 in the prosimian galago (Otolemur garnettii)
Abstract
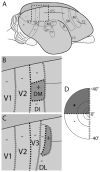
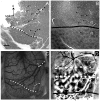
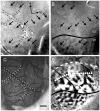
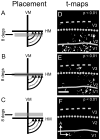
Currently, we lack consensus regarding the organization along the anterior border of dorsomedial V2 in primates. Previous studies suggest that this region could be either the dorsomedial area, characterized by both an upper and a lower visual field representation, or the dorsal aspect of area V3, which only contains a lower visual field representation. We examined these proposals by using optical imaging of intrinsic signals to investigate this region in the prosimian galago (Otolemur garnettii). Galagos represent the prosimian radiation of surviving primates; cortical areas that bear strong resemblances across members of primates provide a strong argument for their early origin and conserved existence. Based on our mapping of horizontal and vertical meridian representations, visuotopy, and orientation preference, we find a clear lower field representation anterior to dorsal V2 but no evidence of any upper field representation. We also show statistical differences in orientation preference patches between V2 and V3. We additionally supplement our imaging results with electrode array data that reveal differences in the average spatial frequency preference, average temporal frequency preference, and sizes of the receptive fields between V1, V2, and V3. The lack of upper visual field representation along with the differences between the neighboring visual areas clearly distinguish the region anterior to dorsal V2 from earlier visual areas and argue against a DM that lies along the dorsomedial border of V2. We submit that the region of the cortex in question is the dorsal aspect of V3, thus strengthening the possibility that V3 is conserved among primates.
Copyright © 2012 Wiley Periodicals, Inc.
Figures





Similar articles
-
Corticocortical connection patterns reveal two distinct visual cortical areas bordering dorsal V2 in marmoset monkey.Vis Neurosci. 2015 Jan;32:E012. doi: 10.1017/S0952523815000097. Vis Neurosci. 2015. PMID: 26423121 Free PMC article.
-
Visual field representation in striate and prestriate cortices of a prosimian primate (Galago garnetti).J Neurophysiol. 1997 Jun;77(6):3193-217. doi: 10.1152/jn.1997.77.6.3193. J Neurophysiol. 1997. PMID: 9212268
-
Second and third visual areas of the cat: interindividual variability in retinotopic arrangement and cortical location.J Physiol. 1980 Feb;299:247-76. doi: 10.1113/jphysiol.1980.sp013123. J Physiol. 1980. PMID: 7381768 Free PMC article.
-
Resolving the organization of the territory of the third visual area: a new proposal.Vis Neurosci. 2015 Jan;32:E016. doi: 10.1017/S0952523815000152. Vis Neurosci. 2015. PMID: 26241553 Review.
-
Visual cortex organization in primates: theories of V3 and adjoining visual areas.Prog Brain Res. 2001;134:285-95. doi: 10.1016/s0079-6123(01)34019-0. Prog Brain Res. 2001. PMID: 11702549 Review.
Cited by
-
Cortical projections to the two retinotopic maps of primate pulvinar are distinct.J Comp Neurol. 2019 Feb 15;527(3):577-588. doi: 10.1002/cne.24515. Epub 2018 Nov 1. J Comp Neurol. 2019. PMID: 30078198 Free PMC article.
-
In vivo evidence of functional and anatomical stripe-based subdivisions in human V2 and V3.Sci Rep. 2017 Apr 7;7(1):733. doi: 10.1038/s41598-017-00634-6. Sci Rep. 2017. PMID: 28389654 Free PMC article.
-
Topographic organization of areas V3 and V4 and its relation to supra-areal organization of the primate visual system.Vis Neurosci. 2015 Jan;32:E014. doi: 10.1017/S0952523815000115. Vis Neurosci. 2015. PMID: 26241035 Free PMC article.
-
Corticocortical connection patterns reveal two distinct visual cortical areas bordering dorsal V2 in marmoset monkey.Vis Neurosci. 2015 Jan;32:E012. doi: 10.1017/S0952523815000097. Vis Neurosci. 2015. PMID: 26423121 Free PMC article.
-
A Comparison of the Efficacy and Cardiopulmonary Effects of 3 Different Sedation Protocols in Otolemur garnettii.J Am Assoc Lab Anim Sci. 2020 Sep 1;59(5):557-566. doi: 10.30802/AALAS-JAALAS-19-000158. Epub 2020 Jul 21. J Am Assoc Lab Anim Sci. 2020. PMID: 32693866 Free PMC article.
References
-
- Adams DL, Zeki S. Functional organization of macaque V3 for stereoscopic depth. J Neurophysiol. 2001;86:2195–2203. - PubMed
-
- Allman JM, Kaas JH. The dorsomedial cortical visual area: a third tier area in the occipital lobe of the owl monkey (Aotus trivirgatus) Brain Res. 1975;100:473–487. - PubMed
-
- Allman J, Campbell CB, McGuinness E. The dorsal third tier area in Galago senegalensis. Brain Res. 1979;179:355–361. - PubMed
-
- Baizer JS. Receptive field properties of V3 neurons in monkey. Invest Ophthalmol Vis Sci. 1982;23:87–95. - PubMed
-
- Beck PD, Kaas JH. Cortical connections of the dorsomedial visual area in new world owl monkeys (Aotus trivir-gatus) and squirrel monkeys (Saimiri sciureus) J Comp Neurol. 1998a;400:18–34. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

