Use of human amelogenin in molecular encapsulation for the design of pH responsive microparticles
- PMID: 22630169
- PMCID: PMC3403901
- DOI: 10.1186/1472-6750-12-25
Use of human amelogenin in molecular encapsulation for the design of pH responsive microparticles
Abstract
Background: Proteins can be used in drug delivery systems to improve pharmacological properties of an active substance. Differences in pH between tissues can be utilized in order to achieve a targeted drug release at a specific location or tissue, such as a tumor. The enamel matrix protein amelogenin has a pH dependent solubility profile and self-assemble to form aggregates at neutral pH. This could make amelogenin useful in the design of pH responsive drug delivery systems.
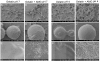
Results: In this study amelogenin was evaluated as a pH responsive component in drug delivery applications. This was achieved by testing the ability of amelogenin to entrap/release other proteins upon changes in pH, and by testing if amelogenin could confer pH responsiveness to an existing and versatile drug delivery system, such as gelatin microparticles. Amelogenin was able to encapsulate bovine serum albumin and insulin, whichwere used as model target proteins. The composite aggregates of amelogenin and target protein were formed at neutral pH and could be reversibly solubilized at weakly acidic pH. Gelatin microparticles prepared in the presence of amelogenin, showed a modulated structure in response to pH change, when studied by scanning electron microscopy, compared to particles without amelogenin. At neutral pH amelogenin induced formation of pores in the particle surface, which were not present at acidic pH, or in particles lacking amelogenin.
Conclusions: The results from this study demonstrate that amelogenin can be a useful component in drug delivery systems in order to achieve a pH dependent response.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

