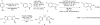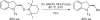Multigram Synthesis of a Chiral Substituted Indoline Via Copper-Catalyzed Alkene Aminooxygenation
- PMID: 22639473
- PMCID: PMC3358786
- DOI: 10.1055/s-0031-1289762
Multigram Synthesis of a Chiral Substituted Indoline Via Copper-Catalyzed Alkene Aminooxygenation
Abstract
(S)-5-Fluoro-2-(2,2,6,6-tetramethylpiperidin-1-yloxymethyl)-1-tosylindoline, a 2-methyleneoxy-substituted chiral indoline, was synthesized on multigram scale using an efficient copper-catalyzed enantioselective intramolecular alkene aminooxygenation. The synthesis is accomplished in four steps and the indoline is obtained in 89% ee (>98% after one recrystallization). Other highlights include efficient gram-scale synthesis of the (4R,5S)-di-Ph-box ligand and efficient separation of a monoallylaniline from its bis(allyl)aniline by-product by distillation under reduced pressure.
Figures
References
-
- Anas S, Kagan HB. Tetrahedron: Asymmetry. 2009;20:2193.
-
- Seaman W, Norton AR, Woods JT, Bank HN. J. Am. Chem. Soc. 1945;67:1571.
- Goetz-Luthy N. J. Chem. Educ. 1949;26:271.
- Bannister WW, Pennace JR, Smith DW, Chelton CF, Wareing JA, Curby WA. J. Chem. Educ. 1973;50:578.
-
-
Original synthesis of ligand 3: Masamune S, Lowenthal RE. Chem. Abstr. ??? 1994 US Patent 5298623 A 19940329, The method used in the present work: Denmark SE, Stiff CM. J. Org. Chem. 2000;65:5875.
-
-
-
a We believe loss of conversion upon scale-up is due to self-quenching of the TEMPO radical (disproportionation), which O2 might prevent. TEMPO reactivity: Vogler T, Studer A. Synthesis. 2008:1979.
-
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous