Balancing of sulfur storage in maize seed
- PMID: 22646812
- PMCID: PMC3420316
- DOI: 10.1186/1471-2229-12-77
Balancing of sulfur storage in maize seed
Abstract
Background: A balanced composition of amino acids in seed flour is critical because of the demand on essential amino acids for nutrition. However, seed proteins in cereals like maize, the crop with the highest yield, are low in lysine, tryptophan, and methionine. Although supplementation with legumes like soybean can compensate lysine deficiency, both crops are also relatively low in methionine. Therefore, understanding the mechanism of methionine accumulation in the seed could be a basis for breeding cultivars with superior nutritional quality.
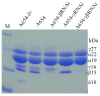
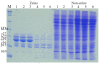
Results: In maize (Zea mays), the 22- and 19-kDa α-zeins are the most prominent storage proteins, nearly devoid of lysine and methionine. Although silencing synthesis of these proteins through RNA interference (RNAi) raises lysine levels in the seed, it fails to do so for methionine. Computational analysis of annotated gene models suggests that about 57% of all proteins exhibit a lysine content of more than 4%, whereas the percentage of proteins with methionine above 4% is only around 8%. To compensate for this low representation, maize seeds produce specialized storage proteins, the 15-kDa β-, 18-kDa and 10-kDa δ-zeins, rich in methionine. However, they are expressed at variant levels in different inbred lines. A654, an inbred with null δ-zein alleles, methionine levels are significantly lower than when the two intact δ-zein alleles are introgressed. Further silencing of β-zein results in dramatic reduction in methionine levels, indicating that β- and δ-zeins are the main sink of methionine in maize seed. Overexpression of the 10-kDa δ-zein can increase the methionine level, but protein analysis by SDS-PAGE shows that the increased methionine levels occur at least in part at the expense of cysteines present in β- and γ-zeins. The reverse is true when β- and γ-zein expression is silenced through RNAi, then 10-kDa δ-zein accumulates to higher levels.
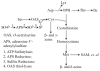
Conclusions: Because methionine receives the sulfur moiety from cysteine, it appears that when seed protein synthesis of cysteine-rich proteins is blocked, the synthesis of methionine-rich seed proteins is induced, probably at the translational level. The same is true, when methionine-rich proteins are overexpressed, synthesis of cysteine-rich proteins is reduced, probably also at the translational level. Although we only hypothesize a translational control of protein synthesis at this time, there are well known paradigms of how amino acid concentration can play a role in differential gene expression. The latter we think is largely controlled by the flux of reduced sulfur during plant growth.
Figures


References
-
- Pedersen K, Argos P, Naravana SV, Larkins BA. Sequence analysis and characterization of amaize gene encoding a high-sulfur zein protein of Mr 15,000. J Biol Chem. 1986;261(14):6279–6284. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

