Three-dimensional quantitative structure activity relationship (QSAR) of cytotoxic active 3,5-diaryl-4,5-dihydropyrazole analogs: a comparative molecular field analysis (CoMFA) revisited study
- PMID: 22647291
- PMCID: PMC3536690
- DOI: 10.1186/1752-153X-6-50
Three-dimensional quantitative structure activity relationship (QSAR) of cytotoxic active 3,5-diaryl-4,5-dihydropyrazole analogs: a comparative molecular field analysis (CoMFA) revisited study
Abstract
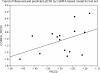
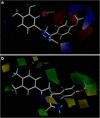
In vitro antitumor evaluation of the synthesized 46 compounds of 3,5-diaryl-4,5-dihydropyrazoles against EAC cell lines and 3D QSAR study using pharmacophore and Comparative Molecular Field Analysis (CoMFA) methods were described. CoMFA derived QSAR model shows a good conventional squared correlation coefficient r2 and cross validated correlation coefficient r2cv 0.896 and 0.568 respectively. In this analysis steric and electrostatic field contribute to the QSAR equation by 70% and 30% respectively, suggesting that variation in biological activity of the compounds is dominated by differences in steric (van der Waals) interactions. To visualize the CoMFA steric and electrostatic field from partial least squares (PLS) analysis, contour maps are plotted as percentage contribution to the QSAR equation and are associated with the differences in biological activity.
Background: Pyrazole derivatives exhibit a wide range of biological properties including promising antitumor activity. Furthermore, Aldol condensation assisted organic synthesis has delivered rapid routes to N-containing heterocycles, including pyrazoles. Combining these features, the use of chalconisation-assisted processes will provide rapid access to a targeted dihydropyrazoles library bearing a hydrazino 3D QSAR study using pharmacophore and Comparative Molecular Field Analysis (CoMFA) methods were described for evaluation of antioxidant properties.
Results: Chalcones promoted 1 of the 2 steps in a rapid, convergent synthesis of a small library of hydrazinyl pyrazole derivatives, all of which exhibited significant antitumor activity against Ehrlich Ascites Carcinoma (EAC) human tumor cell line comparable to that of the natural anticancer doxorubicin, as a reference standard during this study. In order to understand the observed pharmacological properties, quantitative structure-activity relationship (3D QSAR) study was initiated.
Conclusions: Chalcones heating provides a rapid and expedient route to a series of pyrazoles to investigate their chracterization scavenging properties. Given their favorable properties, in comparison with known anticancer, these pyrazole derivatives are promising leads for further development and optimization.
Figures




Similar articles
-
Microwave-assisted synthesis and antioxidant properties of hydrazinyl thiazolyl coumarin derivatives.Chem Cent J. 2012 Apr 17;6(1):32. doi: 10.1186/1752-153X-6-32. Chem Cent J. 2012. PMID: 22510146 Free PMC article.
-
CoMFA and CoMSIA 3D-QSAR analysis of DMDP derivatives as anti-cancer agents.Bioinformation. 2008 Jun 27;2(9):384-91. doi: 10.6026/97320630002384. Bioinformation. 2008. PMID: 18795111 Free PMC article.
-
CoMFA, CoMSIA and HQSAR Analysis of 3-aryl-3-ethoxypropanoic Acid Derivatives as GPR40 Modulators.Curr Drug Discov Technol. 2020;17(1):100-118. doi: 10.2174/1570163815666180829144431. Curr Drug Discov Technol. 2020. PMID: 30160214
-
AI-based 3D-QSAR model of FDA-approved repurposed drugs for inhibiting sclerostin.Technol Health Care. 2024;32(5):3007-3019. doi: 10.3233/THC-231358. Technol Health Care. 2024. PMID: 39031396
-
[Identification of environmental estrogens with a three-dimensional quantitative structure-activity relationship(3D-QSAR) analysis].Nihon Rinsho. 2000 Dec;58(12):2469-76. Nihon Rinsho. 2000. PMID: 11187739 Review. Japanese.
Cited by
-
A second polymorph of (Z)-3-amino-4-(2-phenyl-hydrazinyl-idene)-1H-pyrazol-5(4H)-one.Acta Crystallogr Sect E Struct Rep Online. 2014 Jan 15;70(Pt 2):o141-2. doi: 10.1107/S1600536814000427. eCollection 2014 Feb 1. Acta Crystallogr Sect E Struct Rep Online. 2014. PMID: 24764867 Free PMC article.
-
Ethyl 5-amino-1-[(4-methyl-phen-yl)sulfon-yl]-1H-pyrazole-4-carboxyl-ate.Acta Crystallogr Sect E Struct Rep Online. 2013 Aug 3;69(Pt 9):o1376. doi: 10.1107/S1600536813019326. eCollection 2013. Acta Crystallogr Sect E Struct Rep Online. 2013. PMID: 24427020 Free PMC article.
References
-
- Dai Y, Guo Y, Frey RR, Ji Z, Curtin ML, Ahmed AA, Albert DH, Arnold L, Arries SS, Barlozzari T, Bauch JL, Bouska JJ, Bousquet PF, Cunha GA, Glaser KB, Guo J, Li J, Marcotte PA, Marsh KC, Moskey MD, Pease LJ, Stewart KD, Stoll VS, Tapang P, Wishart N, Davidsen SK, Michaelides MR. Thienopyrimidine ureas as novel and potent multitargeted receptor tyrosine kinase inhibitors. J Med Chem. 2005;48(19):6066–6083. doi: 10.1021/jm050458h. - DOI - PubMed
-
- Manfredini S, Bazzanini R, Baraldi PG, Guarneri M, Simoni D, Marongiu ME, Pani A, Tramontano E, La Colla P. Pyrazole-related nucleosides. Synthesis and antiviral/antitumor activity of some substituted pyrazole and pyrazolo[4,3-d]-1,2,3-triazin-4-one nucleosides. J Med Chem. 1992;35(5):917–924. doi: 10.1021/jm00083a017. - DOI - PubMed
LinkOut - more resources
Full Text Sources

