Increased expression of importin13 in endometriosis and endometrial carcinoma
- PMID: 22648251
- PMCID: PMC3560734
- DOI: 10.12659/msm.882879
Increased expression of importin13 in endometriosis and endometrial carcinoma
Abstract
Background: Importin13 (IPO13) is a novel potential marker of corneal epithelial progenitor cells. We investigated the expression and localization of IPO13 in endometrial, endometriotic and endometrial carcinoma tissue.
Material/methods: IPO13 expression in endometrial, endometriotic and endometrial carcinoma tissue was examined by immunohistochemistry, qPCR and Western blot.
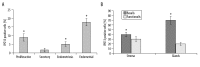
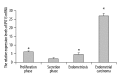
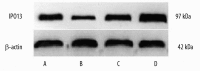
Results: Immunohistochemistry studies showed that IPO13 protein was expressed mainly in cytoplasm of glandular epithelial cell and stromal cells. The rate of importin13-positive cells in proliferative phase endometrium was higher (by about 6-fold) than that in secretory endometrium (P<0.05) and the rate of importin13-positive cells in endometriosis and endometrial carcinoma was higher than that in normal secretory phase endometrial tissues (by about 4- and 9-fold, respectively). Immunofluorescence microscopy revealed co-localization of IPO13 with CD34, CD45, c-kit, telomerase, CD90 and CD146. QPCR revealed significantly increased IPO13 mRNA in endometriosis and endometrial carcinoma versus secretory phase endometrium (by about 2- and 10-fold, respectively). Western blot analysis showed that IPO13 protein is enhanced in endometriosis and endometrial carcinoma versus secretory phase endometrium (p<0.05).
Conclusions: These results demonstrate an increased expression of IPO13 in endometriosis and endometrial carcinoma, which could be involved in the pathogenesis of endometriosis and endometrial carcinoma; IPO13 can serve as an endometrial progenitor/stem cell marker.
Figures



References
-
- Lucot JP, Coulon C, Collinet P, et al. Surgical therapeutic management for menorrhagia. J Gynecol Obstet Biol Reprod (Paris) 2008;37(Suppl 8):S398–404. - PubMed
-
- Chan RW, Gargett CE. Identification of label-retaining cells in mouse endometrium. Stem Cells. 2006;24(6):1529–38. - PubMed
-
- Kato K, Yoshimoto M, Kato K, et al. Characterization of side-population cells in human normal endometrium. Hum Reprod. 2007;22(5):1214–23. - PubMed
-
- Tsuji S, Yoshimoto M, Takahashi K, et al. Side population cells contribute to the genesis of human endometrium. Fertil Steril. 2008;90(Suppl 4):1528–37. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

