Rapid early innate control of hepatitis C virus during IFN-α treatment compromises adaptive CD4+ T-cell immunity
- PMID: 22653709
- PMCID: PMC3781703
- DOI: 10.1002/eji.201142072
Rapid early innate control of hepatitis C virus during IFN-α treatment compromises adaptive CD4+ T-cell immunity
Abstract
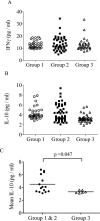
The ability to control HCV with IFN-α-based treatments provides an opportunity in humans to study how the rate of viral clearance in vivo impinges on the development of antiviral responses. Ex vivo (IFN-γ-producing) and cultured antiviral CD4(+) T cells, serum cytokines, and viral loads were measured repeatedly in a cohort of chronically HCV-infected subjects (n = 33) receiving IFN-α. Rapid control of virus indicated by an increased calculated rate of virus clearance, occurred in those subjects demonstrating absent/minimal T-cell responses (p < 0.0006). Surprisingly, in subjects who demonstrated the most robust T-cell responses (and reduced serum IL-10), there was actually a reduced rate of early virus clearance. A subsequent analysis of NK-cell function in available subjects (n = 8) revealed an inverse correlation between pretreatment NK-cell expression of NKp46 and the potential to upregulate cytotoxic function on exposure to IFN-α (p < 0.004), as well as the subsequent measured rate of viral clearance (p = 0.045). Thus, the CD4(+) T-cell response during IFN-α treatment appears to be shaped by the rate of innate virus suppression. These data suggest that individuals who respond most effectively to immune intervention may be most in need of subsequent vaccination to prevent reinfection.
© 2012 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures



References
-
- Randall RE, Goodbourn S. Interferons and viruses: an interplay between induction, signalling, antiviral responses and virus countermeasures. J. Gen. Virol. 2008;89:1–47. - PubMed
-
- Thursz M, Yallop R, Goldin R, Trepo C, Thomas H. Influence of MHC class II genotype on outcome of infection with hepatitis C virus. Lancet. 1999;354:2119–2124. - PubMed
-
- Botarelli P, Brunetto M, Minutello M, Calvo P, Unutmaz D, Weiner A, Choo Q, et al. T-lymphocyte response to hepatitis C virus in different clinical courses of infection. Gastroenterology. 1993;104:580–587. - PubMed
-
- Cramp M, Rossol S, Chokshi S, Carucci P, Williams R, Naoumov N. Hepatitis C virus-specific T-cell reactivity during interferon and ribavirin treatment in chronic hepatitis C. Gastroenterology. 2000;118:346–355. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

