Tbl3 regulates cell cycle length during zebrafish development
- PMID: 22659140
- PMCID: PMC4133786
- DOI: 10.1016/j.ydbio.2012.05.024
Tbl3 regulates cell cycle length during zebrafish development
Abstract
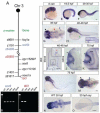
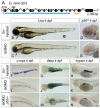
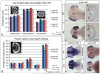
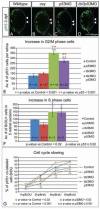
The regulation of cell cycle rate is essential for the correct timing of proliferation and differentiation during development. Changes to cell cycle rate can have profound effects on the size, shape and cell types of a developing organ. We previously identified a zebrafish mutant ceylon (cey) that has a severe reduction in T cells and hematopoietic stem/progenitor cells (HSPCs). Here we find that the cey phenotype is due to absence of the gene transducin (beta)-like 3 (tbl3). The tbl3 homolog in yeast regulates the cell cycle by maintaining rRNA levels and preventing p53-induced cell death. Zebrafish tbl3 is maternally expressed, but later in development its expression is restricted to specific tissues. Tissues expressing tbl3 are severely reduced in cey mutants, including HSPCs, the retina, exocrine pancreas, intestine, and jaw cartilage. Specification of these tissues is normal, suggesting the reduced size is due to a reduced number of differentiated cells. Tbl3 MO injection into either wild-type or p53-/- mutant embryos phenocopies cey, indicating that loss of tbl3 causes specific defects in cey. Progression of both hematopoietic and retinal development is delayed beginning at 3 day post fertilization due to a slowing of the cell cycle. In contrast to yeast, reduction of Tbl3 causes a slowing of the cell cycle without a corresponding increase in p53 induced cell death. These data suggest that tbl3 plays a tissue-specific role regulating cell cycle rate during development.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Depletion of minichromosome maintenance protein 5 in the zebrafish retina causes cell-cycle defect and apoptosis.Proc Natl Acad Sci U S A. 2005 Dec 20;102(51):18467-72. doi: 10.1073/pnas.0506187102. Epub 2005 Dec 8. Proc Natl Acad Sci U S A. 2005. PMID: 16339308 Free PMC article.
-
Loss of zygotic NUP107 protein causes missing of pharyngeal skeleton and other tissue defects with impaired nuclear pore function in zebrafish embryos.J Biol Chem. 2012 Nov 2;287(45):38254-64. doi: 10.1074/jbc.M112.408997. Epub 2012 Sep 10. J Biol Chem. 2012. PMID: 22965233 Free PMC article.
-
Nestin is essential for zebrafish brain and eye development through control of progenitor cell apoptosis.PLoS One. 2010 Feb 19;5(2):e9318. doi: 10.1371/journal.pone.0009318. PLoS One. 2010. PMID: 20174467 Free PMC article.
-
dazed gene is necessary for late cell type development and retinal cell maintenance in the zebrafish retina.Dev Dyn. 2005 Jun;233(2):680-94. doi: 10.1002/dvdy.20375. Dev Dyn. 2005. PMID: 15844196
-
Analysis of cell proliferation, senescence, and cell death in zebrafish embryos.Methods Cell Biol. 2011;101:19-38. doi: 10.1016/B978-0-12-387036-0.00002-5. Methods Cell Biol. 2011. PMID: 21550438 Free PMC article. Review.
Cited by
-
Exploring hematopoiesis in zebrafish using forward genetic screening.Exp Mol Med. 2024 Feb;56(1):51-58. doi: 10.1038/s12276-023-01138-2. Epub 2024 Jan 4. Exp Mol Med. 2024. PMID: 38172599 Free PMC article. Review.
-
The Application of Methylprednisolone Nanoscale Zirconium-Porphyrin Metal-Organic Framework (MPS-NPMOF) in the Treatment of Photoreceptor Degeneration.Int J Nanomedicine. 2019 Dec 10;14:9763-9776. doi: 10.2147/IJN.S225992. eCollection 2019. Int J Nanomedicine. 2019. PMID: 31849467 Free PMC article.
-
Genetic Analysis in a Familial Case With High Bone Mineral Density Suggests Additive Effects at Two Loci.JBMR Plus. 2022 Feb 18;6(4):e10602. doi: 10.1002/jbm4.10602. eCollection 2022 Apr. JBMR Plus. 2022. PMID: 35434450 Free PMC article.
-
The Roles of Histamine Receptor 1 (hrh1) in Neurotransmitter System Regulation, Behavior, and Neurogenesis in Zebrafish.Mol Neurobiol. 2023 Nov;60(11):6660-6675. doi: 10.1007/s12035-023-03447-z. Epub 2023 Jul 20. Mol Neurobiol. 2023. PMID: 37474883 Free PMC article.
-
Effect of electromagnetic field radiation on transcriptomic profile and DNA methylation level in pig conceptuses during the peri-implantation period.Sci Rep. 2025 Apr 23;15(1):14025. doi: 10.1038/s41598-025-98918-9. Sci Rep. 2025. PMID: 40269194 Free PMC article.
References
-
- Agathocleous M, Harris WA. From progenitors to differentiated cells in the vertebrate retina. Annual review of cell and developmental biology. 2009;25:45–69. - PubMed
-
- Aifantis I, Raetz E, Buonamici S. Molecular pathogenesis of T-cell leukaemia and lymphoma. Nat Rev Immunol. 2008;8:380–390. - PubMed
-
- Alexiades MR, Cepko C. Quantitative analysis of proliferation and cell cycle length during development of the rat retina. Dev Dyn. 1996;205:293–307. - PubMed
-
- Austin CP, Feldman DE, Ida JA, Jr., Cepko CL. Vertebrate retinal ganglion cells are selected from competent progenitors by the action of Notch. Development. 1995;121:3637–3650. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

