Chronic hypoxia suppresses pregnancy-induced upregulation of large-conductance Ca2+-activated K+ channel activity in uterine arteries
- PMID: 22665123
- PMCID: PMC3562497
- DOI: 10.1161/HYPERTENSIONAHA.112.196097
Chronic hypoxia suppresses pregnancy-induced upregulation of large-conductance Ca2+-activated K+ channel activity in uterine arteries
Abstract
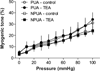
Our previous study demonstrated that increased Ca(2+)-activated K(+) (BK(Ca)) channel activity played a key role in the normal adaptation of reduced myogenic tone of uterine arteries in pregnancy. The present study tested the hypothesis that chronic hypoxia during gestation inhibits pregnancy-induced upregulation of BK(Ca) channel function in uterine arteries. Resistance-sized uterine arteries were isolated from nonpregnant and near-term pregnant sheep maintained at sea level (≈ 300 m) or exposed to high-altitude (3801 m) hypoxia for 110 days. Hypoxia during gestation significantly inhibited pregnancy-induced upregulation of BK(Ca) channel activity and suppressed BK(Ca) channel current density in pregnant uterine arteries. This was mediated by a selective downregulation of BK(Ca) channel β1 subunit in the uterine arteries. In accordance, hypoxia abrogated the role of the BK(Ca) channel in regulating pressure-induced myogenic tone of uterine arteries that was significantly elevated in pregnant animals acclimatized to chronic hypoxia. In addition, hypoxia abolished the steroid hormone-mediated increase in the β1 subunit and BK(Ca) channel current density observed in nonpregnant uterine arteries. Although the activation of protein kinase C inhibited BK(Ca) channel current density in pregnant uterine arteries of normoxic sheep, this effect was ablated in the hypoxic animals. The results demonstrate that selectively targeting BK(Ca) channel β1 subunit plays a critical role in the maladaption of uteroplacental circulation caused by chronic hypoxia, which contributes to the increased incidence of preeclampsia and fetal intrauterine growth restriction associated with gestational hypoxia.
Figures



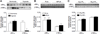


Similar articles
-
Pregnancy upregulates large-conductance Ca(2+)-activated K(+) channel activity and attenuates myogenic tone in uterine arteries.Hypertension. 2011 Dec;58(6):1132-9. doi: 10.1161/HYPERTENSIONAHA.111.179952. Epub 2011 Oct 31. Hypertension. 2011. PMID: 22042813 Free PMC article.
-
Gestational hypoxia increases reactive oxygen species and inhibits steroid hormone-mediated upregulation of Ca(2+)-activated K(+) channel function in uterine arteries.Hypertension. 2014 Aug;64(2):415-22. doi: 10.1161/HYPERTENSIONAHA.114.03555. Epub 2014 May 27. Hypertension. 2014. PMID: 24866137 Free PMC article.
-
Chronic hypoxia inhibits sex steroid hormone-mediated attenuation of ovine uterine arterial myogenic tone in pregnancy.Hypertension. 2010 Oct;56(4):750-7. doi: 10.1161/HYPERTENSIONAHA.110.155812. Epub 2010 Jul 26. Hypertension. 2010. PMID: 20660818 Free PMC article.
-
Reduction of large-conductance Ca²(+) -activated K(+) channel with compensatory increase of nitric oxide in insulin resistant rats.Diabetes Metab Res Rev. 2011 Jul;27(5):461-9. doi: 10.1002/dmrr.1196. Diabetes Metab Res Rev. 2011. PMID: 21425425 Review.
-
Hypoxic Regulation of the Large-Conductance, Calcium and Voltage-Activated Potassium Channel, BK.Front Physiol. 2021 Dec 22;12:780206. doi: 10.3389/fphys.2021.780206. eCollection 2021. Front Physiol. 2021. PMID: 35002762 Free PMC article. Review.
Cited by
-
Chronic hypoxia inhibits pregnancy-induced upregulation of SKCa channel expression and function in uterine arteries.Hypertension. 2013 Aug;62(2):367-74. doi: 10.1161/HYPERTENSIONAHA.113.01236. Epub 2013 May 28. Hypertension. 2013. PMID: 23716582 Free PMC article.
-
Developmental acceleration of bradykinin-dependent relaxation by prenatal chronic hypoxia impedes normal development after birth.Am J Physiol Lung Cell Mol Physiol. 2016 Feb 1;310(3):L271-86. doi: 10.1152/ajplung.00340.2015. Epub 2015 Dec 4. Am J Physiol Lung Cell Mol Physiol. 2016. PMID: 26637638 Free PMC article.
-
Long-term high altitude hypoxia during gestation suppresses large conductance Ca2+ -activated K+ channel function in uterine arteries: a causal role for microRNA-210.J Physiol. 2018 Dec;596(23):5891-5906. doi: 10.1113/JP276058. Epub 2018 Jul 1. J Physiol. 2018. PMID: 29869786 Free PMC article.
-
Altered placental ion channel gene expression in preeclamptic high-altitude pregnancies.Physiol Genomics. 2023 Sep 1;55(9):357-367. doi: 10.1152/physiolgenomics.00013.2023. Epub 2023 Jul 17. Physiol Genomics. 2023. PMID: 37458464 Free PMC article.
-
Direct effect of chronic hypoxia in suppressing large conductance Ca(2+)-activated K(+) channel activity in ovine uterine arteries via increasing oxidative stress.J Physiol. 2016 Jan 15;594(2):343-56. doi: 10.1113/JP271626. Epub 2015 Dec 21. J Physiol. 2016. PMID: 26613808 Free PMC article.
References
-
- Ni Y, May V, Braas K, Osol G. Pregnancy augments uteroplacental vascular endothelial growth factor gene expression and vasodilator effects. Am J Physiol. 1997;273:H938–H944. - PubMed
-
- Nelson SH, Steinsland OS, Suresh MS, Lee NM. Pregnancy augments nitric oxide-dependent dilator response to acetylcholine in the human uterine artery. Hum Reprod. 1998;13:1361–1367. - PubMed
-
- Gangula PR, Zhao H, Supowit S, Wimalawansa S, DiPette D, Yallampalli C. Pregnancy and steroid hormones enhance the vasodilation responses to CGRP in rats. Am J Physiol. 1999;276:H284–H288. - PubMed
-
- Xiao D, Pearce WJ, Zhang L. Pregnancy enhances endothelium-dependent relaxation of ovine uterine artery: role of NO and intracellular Ca2+ Am J Physiol Heart Circ Physiol. 2001;281:H183–H190. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

