Transition metals and mitochondrial metabolism in the heart
- PMID: 22668786
- PMCID: PMC3597232
- DOI: 10.1016/j.yjmcc.2012.05.014
Transition metals and mitochondrial metabolism in the heart
Abstract
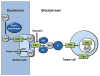
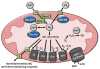
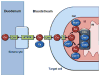
Transition metals are essential to many biological processes in almost all organisms from bacteria to humans. Their versatility, which arises from an ability to undergo reduction-oxidation chemistry, enables them to act as critical cofactors of enzymes throughout the cell. Accumulation of metals, however, can also lead to oxidative stress and cellular damage. The importance of metals to both enzymatic reactions and oxidative stress makes them key players in mitochondria. Mitochondria are the primary energy-generating organelles of the cell that produce ATP through a chain of enzymatic complexes that require transition metals, and are highly sensitive to oxidative damage. Moreover, the heart is one of the most mitochondrially-rich tissues in the body, making metals of particular importance to cardiac function. In this review, we focus on the current knowledge about the role of transition metals (specifically iron, copper, and manganese) in mitochondrial metabolism in the heart. This article is part of a Special Issue entitled "Focus on Cardiac Metabolism".
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
Metabolic Derangement of Essential Transition Metals and Potential Antioxidant Therapies.Int J Mol Sci. 2024 Jul 18;25(14):7880. doi: 10.3390/ijms25147880. Int J Mol Sci. 2024. PMID: 39063122 Free PMC article. Review.
-
Yeast, a model organism for iron and copper metabolism studies.Biometals. 2003 Mar;16(1):185-97. doi: 10.1023/a:1020771000746. Biometals. 2003. PMID: 12572678 Review.
-
[Transition metal accumulation and cellular senescence].Sheng Li Xue Bao. 2024 Jun 25;76(3):418-428. Sheng Li Xue Bao. 2024. PMID: 38939936 Review. Chinese.
-
Metals, oxidative stress and neurodegeneration: a focus on iron, manganese and mercury.Neurochem Int. 2013 Apr;62(5):575-94. doi: 10.1016/j.neuint.2012.12.006. Epub 2012 Dec 21. Neurochem Int. 2013. PMID: 23266600 Free PMC article. Review.
-
SR and mitochondria: calcium cross-talk between kissing cousins.J Mol Cell Cardiol. 2013 Feb;55:42-9. doi: 10.1016/j.yjmcc.2012.07.015. Epub 2012 Aug 2. J Mol Cell Cardiol. 2013. PMID: 22902320 Review.
Cited by
-
Associations Between Metal-Metal Interaction and the Risk of Hypertension: A Case-Control Study in Chinese Community-Dwelling Elderly.Cardiovasc Toxicol. 2025 Jul;25(7):955-968. doi: 10.1007/s12012-025-10015-5. Epub 2025 May 28. Cardiovasc Toxicol. 2025. PMID: 40434610
-
Mitochondrial membrane transporters and metabolic switch in heart failure.Heart Fail Rev. 2019 Mar;24(2):255-267. doi: 10.1007/s10741-018-9756-2. Heart Fail Rev. 2019. PMID: 30535838 Review.
-
Mitochondrial Dysfunction in Heart Failure: From Pathophysiological Mechanisms to Therapeutic Opportunities.Int J Mol Sci. 2024 Feb 25;25(5):2667. doi: 10.3390/ijms25052667. Int J Mol Sci. 2024. PMID: 38473911 Free PMC article. Review.
-
Genetic Disorders Associated with Metal Metabolism.Cells. 2019 Dec 9;8(12):1598. doi: 10.3390/cells8121598. Cells. 2019. PMID: 31835360 Free PMC article. Review.
-
Role of copper homeostasis and cuproptosis in heart failure pathogenesis: implications for therapeutic strategies.Front Pharmacol. 2025 Jan 9;15:1527901. doi: 10.3389/fphar.2024.1527901. eCollection 2024. Front Pharmacol. 2025. PMID: 39850564 Free PMC article. Review.
References
-
- Aisen P, Enns C, Wessling-Resnick M. Chemistry and biology of eukaryotic iron metabolism. Int J Biochem Cell Biol. 2001;33:940–59. - PubMed
-
- Wardman P, Candeias LP. Fenton chemistry: an introduction. Radiat Res. 1996;145:523–31. - PubMed
-
- Thannickal VJ, Fanburg BL. Reactive oxygen species in cell signaling. Am J Physiol Lung Cell Mol Physiol. 2000;279:L1005–28. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

