Dot immunobinding assay method with chlorophyll removal for the detection of southern rice black-streaked dwarf virus
- PMID: 22669043
- PMCID: PMC6268761
- DOI: 10.3390/molecules17066886
Dot immunobinding assay method with chlorophyll removal for the detection of southern rice black-streaked dwarf virus
Abstract
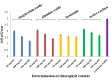
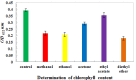
Southern rice black-streaked dwarf virus (SRBSDV), a new virus from Fiji, has seriously damaged rice crops in southern China and northern Vietnam in recent years. This virus is difficult to diagnose in the early stages of infection, and is very destructive at the late stage. In the present study, a dot immunobinding assay (DIBA) that has a high sensitivity for diagnosing SRBSDV was developed. Two kinds of treatment for the DIBA were evaluated to determine the most effective one for removing chlorophyll interferences via rice extraction. The first included several reagents to remove chlorophyll, namely, the alkaline reagents like magnesium oxide and alumina oxide, the adsorbent reagents like activated carbon and bentonite, as well as the extraction agent acetone. The second and third treatments, which were used to remove chlorophyll in blot membrane-nitrocellulose and polyvinylidene fluoride (PVDF), included several organic solvents containing methanol, ethanol, acetone, ethyl acetate, and diethyl ether. The results showed that activated carbon and methanol yielded the best contrasting purple color for the infected samples by decreasing the chlorophyll content.
Figures





References
-
- Zhuo G.H., Zhang S.G., Zou S.F. The characteristic of the occurrence and the analysis of the trend of the extension of SRBSDV-The new rice disease. Plant Protect. 2010;36:235–237.
-
- Zhuo G.H., Wen J.J., Cai D.J. A new kind of virus of Feiji genus of Reovirus family: Southern rice black-streaked dwarf virus. Chin. Sci. Ser. B. 2008;53:2500–2508.
-
- Chen Z., Song B.A. The Technology of Prevention and Control on Southern Rice Black-Streaked Dwarf Virus. Chemical Industrial Press; Beijing, China: 2011. pp. 1–3.
-
- Ha V.C., Nguyen V.H., Vu T.M., Masaru M. Rice dwarf disease in north Vietnam in 2009 is caused by southern rice black-streaked dwarf virus (SRBSDV) Bull. Inst. Trop. Agric. Kyushu Univ. 2009;32:85–92.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

