Characterization of fusarium keratitis outbreak isolates: contribution of biofilms to antimicrobial resistance and pathogenesis
- PMID: 22669723
- PMCID: PMC3394686
- DOI: 10.1167/iovs.12-9848
Characterization of fusarium keratitis outbreak isolates: contribution of biofilms to antimicrobial resistance and pathogenesis
Abstract
Purpose: Fusarium is a major cause of microbial keratitis, and its ability to form biofilms was suggested as a contributing factor in recent outbreaks. We investigated the ability of outbreak Fusarium isolates (F. solani species complex [FSSC] and F. oxysporum species complex [FOSC]) to form biofilms in vitro and in vivo, and evaluated their antifungal susceptibilities.
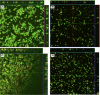
Methods: Biofilm formation was assessed using our in vitro contact lens model and in vivo murine model. Biofilm architecture was assessed using confocal laser scanning microscopy (CLSM). Susceptibility against amphotericin B (AmB), voriconazole (VCZ), and natamycin (NAT) was determined using the CLSI-M38-A2 method and XTT metabolic assay.
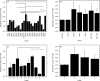
Results: FSSC strains formed more biofilms than FOSC, in a strain- and clade-dependent manner. CLSM analyses revealed that "high biofilm forming" (HBF) strains had denser and thicker biofilms than "low biofilm forming" (LBF) strains of both species (thickness 51 vs. 41 μm for FSSC and 61 vs. 45 μm for FOSC strains, P < 0.05 for both comparisons). Fusarium biofilms exhibited species-dependent antifungal susceptibilities (e.g., FSSC biofilms AmB minimal inhibitory concentrations [MIC] ≥16 μg/mL, while NAT or VCZ MICs were 2-8 μg/mL). FSSC-infected mice had severe corneal opacification independent of biofilm thickness, while FOSC infection resulted in moderate corneal opacification. Corneal fungal burden of mice infected with HBF strains was higher than those of the LBF strains. In contrast, the reference ATCC isolate was unable to cause infection.
Conclusions: The ability to form biofilms is a key pathogenicity determinant of Fusarium, irrespective of the thickness of these biofilms. Further studies are warranted to explore this association in greater detail.
Conflict of interest statement
Disclosure:
Figures

References
-
- Rosa RH, Jr, Miller D, Alfonso EC. The changing spectrum of fungal keratitis in south Florida. Ophthalmology. 1994;101:1005–1013 - PubMed
-
- Hu SBS, Fan VC, Koonapareddy C, Du TT, Asbell PA. Contact lens-related Fusarium infection: case series experience in New York City and review of fungal keratitis. Eye Contact Lens. 2007;33:322–328 - PubMed
-
- Iyer SA, Tuli SS, Wagoner RC. Fungal keratitis: emerging trends and treatment outcomes. Eye Contact Lens. 2006;32:267–271 - PubMed
-
- Bharathi MJ, Ramakrishnan R, Vasu S, Meenakshi R, Palaniappan R. Epidemiological characteristics and laboratory diagnosis of fungal keratitis. A three-year study. Indian J Ophthalmol. 2003;51:315–321 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

