Time course pathogenesis of sulphur mustard-induced skin lesions in mouse model
- PMID: 22672652
- PMCID: PMC7950708
- DOI: 10.1111/j.1742-481X.2012.01003.x
Time course pathogenesis of sulphur mustard-induced skin lesions in mouse model
Abstract
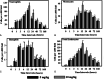
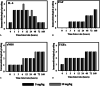
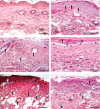
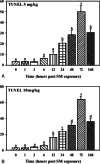
Sulphur mustard (SM) is a bifunctional alkylating agent that causes cutaneous blistering in humans and animals. In this study, we have presented closer views on pathogenesis of SM-induced skin injury in a mouse model. SM diluted in acetone was applied once dermally at a dose of 5 or 10 mg/kg to Swiss albino mice. Skin was dissected out at 0, 1, 3, 6, 12, 24, 48, 72 and 168 hours, post-SM exposure for studying histopathological changes and immunohistochemistry of inflammatory-reparative biomarkers, namely, transforming growth factor alpha (TGF-α), fibroblast growth factor (FGF), endothelial nitric oxide synthase (eNOS) and interlukin 6 (IL-6). Histopathological changes were similar to other mammalian species and basal cell damage resembled the histopathological signs observed with vesication in human skin. Inflammatory cell recruitment at the site of injury was supported by differential expressions of IL-6 at various stages. Time-dependent expressions of eNOS played pivotal roles in all the events of wound healing of SM-induced skin lesions. TGF-α and FGF were strongly associated with keratinocyte migration, re-epithelialisation, angiogenesis, fibroblast proliferation and cell differentiation. Furthermore, quantification of the tissue leukocytosis and DNA damage along with semiquantitative estimation of re-epithelialisation, fibroplasia and neovascularisation on histomorphologic scale could be efficiently used for screening the efficacy of orphan drugs against SM-induced skin injury.
Keywords: DNA damage; FGF; IL-6; Sulphur mustard; TGF-α; eNOS.
© 2012 The Authors. International Wound Journal © 2012 John Wiley & Sons Ltd and Medicalhelplines.com Inc.
Figures






Similar articles
-
Evaluation of wound-healing formulation against sulphur mustard-induced skin injury in mice.Hum Exp Toxicol. 2012 Jun;31(6):588-605. doi: 10.1177/0960327111429139. Epub 2011 Dec 5. Hum Exp Toxicol. 2012. PMID: 22144726
-
A novel decontaminant and wound healant formulation of N,N'-dichloro-bis[2,4,6-trichlorophenyl]urea against sulfur mustard-induced skin injury.Wound Repair Regen. 2014 Jan-Feb;22(1):85-95. doi: 10.1111/wrr.12126. Wound Repair Regen. 2014. PMID: 24393156
-
Designing of mouse model: a new approach for studying sulphur mustard-induced skin lesions.Burns. 2011 Aug;37(5):851-64. doi: 10.1016/j.burns.2010.12.010. Epub 2011 Feb 22. Burns. 2011. PMID: 21334815
-
Molecular toxicology of sulfur mustard-induced cutaneous inflammation and blistering.Toxicology. 2009 Sep 1;263(1):12-9. doi: 10.1016/j.tox.2009.01.019. Epub 2009 Jan 23. Toxicology. 2009. PMID: 19651324 Review.
-
Tissue injury and repair following cutaneous exposure of mice to sulfur mustard.Ann N Y Acad Sci. 2016 Aug;1378(1):118-123. doi: 10.1111/nyas.13125. Epub 2016 Jul 2. Ann N Y Acad Sci. 2016. PMID: 27371823 Free PMC article. Review.
Cited by
-
Skin Models Used to Define Mechanisms of Action of Sulfur Mustard.Disaster Med Public Health Prep. 2023 Oct 18;17:e551. doi: 10.1017/dmp.2023.177. Disaster Med Public Health Prep. 2023. PMID: 37849329 Free PMC article. Review.
-
In Vitro and In Vivo Characterization Methods for Evaluation of Modern Wound Dressings.Pharmaceutics. 2022 Dec 22;15(1):42. doi: 10.3390/pharmaceutics15010042. Pharmaceutics. 2022. PMID: 36678671 Free PMC article. Review.
References
-
- Saladi RN, Smith E, Persaud AN. Mustard: a potential agent of chemical warfare and terrorism. Clin Exp Dermatol 2006;31:1–5. - PubMed
-
- Smith KJ, Smith WJ, Hamilton T, Skelton HG, Graham JS, Okerberg C, Moeller R, Hackley BE Jr. Histopathologic and immunohistochemical features in human skin after exposure to nitrogen and sulfur mustard. Am J Dermatopathol 1998;20:22–8. - PubMed
-
- Kehe K, Balszuweit F, Steinritz D, Thiermann H. Molecular toxicology of sulphur mustard‐induced cutaneous inflammation and blistering. Toxicology 2009;263:12–9. - PubMed
-
- Debiak M, Kehe K, Bürkle A. Role of poly(ADP‐ribose) polymerase in sulfur mustard toxicity. Toxicology 2009;263:20–5. - PubMed
-
- Kehe K, Raithel K, Kreppel H, Jochum M, Worek F, Thiermann H. Inhibition of poly (ADP‐ribose) polymerase (PARP) influences the mode of sulfur mustard (SM)‐induced cell death in HaCaT cells. Arch Toxicol 2007;82:461–70. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

