Irregular Breathing in Mice following Genetic Ablation of V2a Neurons
- PMID: 22674265
- PMCID: PMC3652431
- DOI: 10.1523/JNEUROSCI.0445-12.2012
Irregular Breathing in Mice following Genetic Ablation of V2a Neurons
Abstract
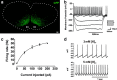
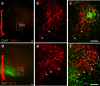
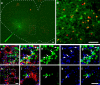
Neural networks called central pattern generators (CPGs) generate repetitive motor behaviors such as locomotion and breathing. Glutamatergic neurons are required for the generation and inhibitory neurons for the patterning of the motor activity associated with repetitive motor behaviors. In the mouse, glutamatergic V2a neurons coordinate the activity of left and right leg CPGs in the spinal cord enabling mice to generate an alternating gait. Here, we investigate the role of V2a neurons in the neural control of breathing, an essential repetitive motor behavior. We find that, following the ablation of V2a neurons, newborn mice breathe at a lower frequency. Recordings of respiratory activity in brainstem-spinal cord and respiratory slice preparations demonstrate that mice lacking V2a neurons are deficient in central respiratory rhythm generation. The absence of V2a neurons in the respiratory slice preparation can be compensated for by bath application of neurochemicals known to accelerate the breathing rhythm. In this slice preparation, V2a neurons exhibit a tonic firing pattern. The existence of direct connections between V2a neurons in the medial reticular formation and neurons of the pre-Bötzinger complex indicates that V2a neurons play a direct role in the function of the respiratory CPG in newborn mice. Thus, neurons of the embryonic V2a lineage appear to have been recruited to neural networks that control breathing and locomotion, two prominent CPG-driven, repetitive motor behaviors.
Figures
Similar articles
-
Electrophysiological characterization of V2a interneurons and their locomotor-related activity in the neonatal mouse spinal cord.J Neurosci. 2010 Jan 6;30(1):170-82. doi: 10.1523/JNEUROSCI.4849-09.2010. J Neurosci. 2010. PMID: 20053899 Free PMC article.
-
V2a Neurons Constrain Extradiaphragmatic Respiratory Muscle Activity at Rest.eNeuro. 2019 Aug 6;6(4):ENEURO.0492-18.2019. doi: 10.1523/ENEURO.0492-18.2019. Print 2019 Jul/Aug. eNeuro. 2019. PMID: 31324674 Free PMC article.
-
In mice lacking V2a interneurons, gait depends on speed of locomotion.J Neurosci. 2009 May 27;29(21):7098-109. doi: 10.1523/JNEUROSCI.1206-09.2009. J Neurosci. 2009. PMID: 19474336 Free PMC article.
-
Chx10+V2a interneurons in spinal motor regulation and spinal cord injury.Neural Regen Res. 2023 May;18(5):933-939. doi: 10.4103/1673-5374.355746. Neural Regen Res. 2023. PMID: 36254971 Free PMC article. Review.
-
Neuronal mechanisms of respiratory rhythm generation: an approach using in vitro preparation.Jpn J Physiol. 1997 Oct;47(5):385-403. doi: 10.2170/jjphysiol.47.385. Jpn J Physiol. 1997. PMID: 9504127 Review.
Cited by
-
Modular derivation of diverse, regionally discrete human posterior CNS neurons enables discovery of transcriptomic patterns.Sci Adv. 2022 Sep 30;8(39):eabn7430. doi: 10.1126/sciadv.abn7430. Epub 2022 Sep 30. Sci Adv. 2022. PMID: 36179024 Free PMC article.
-
V2a interneuron differentiation from mouse and human pluripotent stem cells.Nat Protoc. 2019 Nov;14(11):3033-3058. doi: 10.1038/s41596-019-0203-1. Epub 2019 Oct 18. Nat Protoc. 2019. PMID: 31628445 Free PMC article.
-
Lhx3-Chx10 reticulospinal neurons in locomotor circuits.J Neurosci. 2013 Sep 11;33(37):14681-92. doi: 10.1523/JNEUROSCI.5231-12.2013. J Neurosci. 2013. PMID: 24027269 Free PMC article.
-
Dual effects of 5-HT(1a) receptor activation on breathing in neonatal mice.J Neurosci. 2014 Jan 1;34(1):51-9. doi: 10.1523/JNEUROSCI.0864-13.2014. J Neurosci. 2014. PMID: 24381267 Free PMC article.
-
Measuring Breathing Patterns in Mice Using Whole-body Plethysmography.Bio Protoc. 2020 Sep 5;10(17):e3741. doi: 10.21769/BioProtoc.3741. eCollection 2020 Sep 5. Bio Protoc. 2020. PMID: 33659401 Free PMC article.
References
-
- Al-Mosawie A, Wilson JM, Brownstone RM. Heterogeneity of V2-derived interneurons in the adult mouse spinal cord. Eur J Neurosci. 2007;26:3003–3015. - PubMed
-
- Amiel J, Dubreuil V, Ramanantsoa N, Fortin G, Gallego J, Brunet JF, Goridis C. PHOX2B in respiratory control: lessons from congenital central hypoventilation syndrome and its mouse models. Respir Physiol Neurobiol. 2009;168:125–132. - PubMed
-
- Borday C, Chatonnet F, Thoby-Brisson M, Champagnat J, Fortin G. Neural tube patterning by Krox20 and emergence of a respiratory control. Respir Physiol Neurobiol. 2005;149:63–72. - PubMed
-
- Bouvier J, Thoby-Brisson M, Renier N, Dubreuil V, Ericson J, Champagnat J, Pierani A, Chédotal A, Fortin G. Hindbrain interneurons and axon guidance signaling critical for breathing. Nat Neurosci. 2010;13:1066–1074. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
