Chondrocyte-intrinsic Smad3 represses Runx2-inducible matrix metalloproteinase 13 expression to maintain articular cartilage and prevent osteoarthritis
- PMID: 22674505
- PMCID: PMC3544176
- DOI: 10.1002/art.34566
Chondrocyte-intrinsic Smad3 represses Runx2-inducible matrix metalloproteinase 13 expression to maintain articular cartilage and prevent osteoarthritis
Abstract
Objective: To identify mechanisms by which Smad3 maintains articular cartilage and prevents osteoarthritis.
Methods: A combination of in vivo and in vitro approaches was used to test the hypothesis that Smad3 represses Runx2-inducible gene expression to prevent articular cartilage degeneration. Col2-Cre;Smad3(fl/fl) mice allowed study of the chondrocyte-intrinsic role of Smad3 independently of its role in the perichondrium or other tissues. Primary articular cartilage chondrocytes from Smad3(fl/fl) mice and ATDC5 chondroprogenitor cells were used to evaluate Smad3 and Runx2 regulation of matrix metalloproteinase 13 (MMP-13) messenger RNA (mRNA) and protein expression.
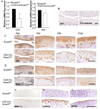
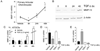
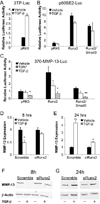
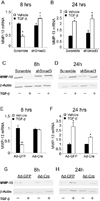
Results: Chondrocyte-specific reduction of Smad3 caused progressive articular cartilage degeneration due to imbalanced cartilage matrix synthesis and degradation. In addition to reduced type II collagen mRNA expression, articular cartilage from Col2-Cre;Smad3(fl/fl) mice was severely deficient in type II collagen and aggrecan protein due to excessive MMP-13-mediated proteolysis of these key cartilage matrix constituents. Normally, transforming growth factor β (TGFβ) signals through Smad3 to confer a rapid and dynamic repression of Runx2-inducible MMP-13 expression. However, we found that in the absence of Smad3, TGFβ signals through p38 and Runx2 to induce MMP-13 expression.
Conclusion: Our findings elucidate a mechanism by which Smad3 mutations in humans and mice cause cartilage degeneration and osteoarthritis. Specifically, Smad3 maintains the balance between cartilage matrix synthesis and degradation by inducing type II collagen expression and repressing Runx2-inducible MMP-13 expression. Selective activation of TGFβ signaling through Smad3, rather than p38, may help to restore the balance between matrix synthesis and proteolysis that is lost in osteoarthritis.
Copyright © 2012 by the American College of Rheumatology.
Figures
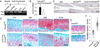

Similar articles
-
Deletion of the transforming growth factor β receptor type II gene in articular chondrocytes leads to a progressive osteoarthritis-like phenotype in mice.Arthritis Rheum. 2013 Dec;65(12):3107-19. doi: 10.1002/art.38122. Arthritis Rheum. 2013. PMID: 23982761 Free PMC article.
-
Deletion of Runx2 in Articular Chondrocytes Decelerates the Progression of DMM-Induced Osteoarthritis in Adult Mice.Sci Rep. 2017 May 24;7(1):2371. doi: 10.1038/s41598-017-02490-w. Sci Rep. 2017. PMID: 28539595 Free PMC article.
-
Chondrocyte-Specific RUNX2 Overexpression Accelerates Post-traumatic Osteoarthritis Progression in Adult Mice.J Bone Miner Res. 2019 Sep;34(9):1676-1689. doi: 10.1002/jbmr.3737. Epub 2019 Jun 12. J Bone Miner Res. 2019. PMID: 31189030 Free PMC article.
-
Endochondral ossification signals in cartilage degradation during osteoarthritis progression in experimental mouse models.Mol Cells. 2008 Feb 29;25(1):1-6. Mol Cells. 2008. PMID: 18319608 Review.
-
The role of the Notch pathway in healthy and osteoarthritic articular cartilage: from experimental models to ex vivo studies.Arthritis Res Ther. 2011 Mar 18;13(2):208. doi: 10.1186/ar3255. Arthritis Res Ther. 2011. PMID: 21457519 Free PMC article. Review.
Cited by
-
Runx2 and Runx3 differentially regulate articular chondrocytes during surgically induced osteoarthritis development.Nat Commun. 2022 Oct 19;13(1):6187. doi: 10.1038/s41467-022-33744-5. Nat Commun. 2022. PMID: 36261443 Free PMC article.
-
Exosomes Derived from Runx2-Overexpressing BMSCs Enhance Cartilage Tissue Regeneration and Prevent Osteoarthritis of the Knee in a Rabbit Model.Stem Cells Int. 2022 Nov 28;2022:6865041. doi: 10.1155/2022/6865041. eCollection 2022. Stem Cells Int. 2022. PMID: 39282499 Free PMC article.
-
TGFβ/BMP Signaling Pathway in Cartilage Homeostasis.Cells. 2019 Aug 24;8(9):969. doi: 10.3390/cells8090969. Cells. 2019. PMID: 31450621 Free PMC article. Review.
-
Effects of Balneotherapy in Jeju Magma-Seawater on Knee Osteoarthritis Model.Sci Rep. 2020 Apr 20;10(1):6620. doi: 10.1038/s41598-020-62867-2. Sci Rep. 2020. PMID: 32313003 Free PMC article.
-
Eosinophils-Induced Lumican Secretion by Synovial Fibroblasts Alleviates Cartilage Degradation via the TGF-β Pathway Mediated by Anxa1 Binding.Adv Sci (Weinh). 2025 Aug;12(29):e2416030. doi: 10.1002/advs.202416030. Epub 2025 Mar 24. Adv Sci (Weinh). 2025. PMID: 40125663 Free PMC article.
References
-
- Aigner T, Vornehm SI, Zeiler G, Dudhia J, von der Mark K, Bayliss MT. Suppression of cartilage matrix gene expression in upper zone chondrocytes of osteoarthritic cartilage. Arthritis Rheum. 1997;40(3):562–569. - PubMed
-
- Shlopov BV, Lie WR, Mainardi CL, Cole AA, Chubinskaya S, Hasty KA. Osteoarthritic lesions: involvement of three different collagenases. Arthritis Rheum. 1997;40(11):2065–2074. - PubMed
-
- Blaney Davidson EN, van der Kraan PM, van den Berg WB. TGF-beta and osteoarthritis. Osteoarthritis Cartilage. 2007;15(6):597–604. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

