The Chinese medicine formula HB01 reduces choroidal neovascularization by regulating the expression of vascular endothelial growth factor
- PMID: 22676316
- PMCID: PMC3479009
- DOI: 10.1186/1479-5876-10-118
The Chinese medicine formula HB01 reduces choroidal neovascularization by regulating the expression of vascular endothelial growth factor
Abstract
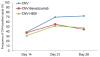
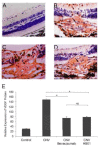
Background: Choroidal neovascularization (CNV) remains the leading cause of newly acquired blindness in the developed world. Currently anti-vascular endothelial growth factor (VEGF) therapies are broadly used to treat neovascular ocular disorders. Here we demonstrate the effect of a traditional Chinese medicine formula, HB01, on CNV.
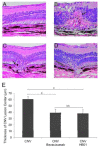
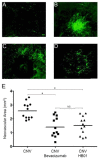
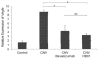
Methods: A rat model of laser-induced CNV was used to investigate the effect of HB01 in vivo. The CNV lesions in the eye were evaluated using fundus fluorescein angiography and visualized/quantified using confocal microscopy. Expression of VEGF in the choroidal and retinal tissues was measured using quantitative real-time PCR and immunohistochemistry.
Results: We demonstrated that a traditional Chinese Medicine formula, named HB01, significantly reduced neovascularization in a rat CNV model. The effect of HB01 on CNV was comparable to the intravitreal injection of bevacizumab (Avastin). Our results also suggested that HB01 may reduce CNV partially through inhibiting the expression of VEGF.
Conclusions: These data support HB01 as an alternative therapy for ocular neovascular disorders.
Figures

Similar articles
-
Inhibition of experimental choroidal neovascularization in mice by anti-VEGFA/VEGFR2 or non-specific siRNA.Exp Eye Res. 2010 Sep;91(3):433-9. doi: 10.1016/j.exer.2010.06.019. Epub 2010 Jul 1. Exp Eye Res. 2010. PMID: 20599960
-
Intravitreal itraconazole inhibits laser-induced choroidal neovascularization in rats.PLoS One. 2017 Jun 30;12(6):e0180482. doi: 10.1371/journal.pone.0180482. eCollection 2017. PLoS One. 2017. PMID: 28666022 Free PMC article.
-
Suppression of choroidal neovascularization by vasohibin-1, a vascular endothelium-derived angiogenic inhibitor.Invest Ophthalmol Vis Sci. 2011 May 17;52(6):3272-80. doi: 10.1167/iovs.10-6295. Invest Ophthalmol Vis Sci. 2011. PMID: 21345982
-
Intravitreal Stanniocalcin-1 Enhances New Blood Vessel Growth in a Rat Model of Laser-Induced Choroidal Neovascularization.Invest Ophthalmol Vis Sci. 2018 Feb 1;59(2):1125-1133. doi: 10.1167/iovs.17-23083. Invest Ophthalmol Vis Sci. 2018. PMID: 29490350 Free PMC article.
-
Annexin A2 promotes choroidal neovascularization by increasing vascular endothelial growth factor expression in a rat model of argon laser coagulation-induced choroidal neovascularization.Chin Med J (Engl). 2010 Mar 20;123(6):713-21. Chin Med J (Engl). 2010. PMID: 20368092
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

