Ceramide kinase-like (CERKL) interacts with neuronal calcium sensor proteins in the retina in a cation-dependent manner
- PMID: 22678504
- PMCID: PMC3394741
- DOI: 10.1167/iovs.12-9770
Ceramide kinase-like (CERKL) interacts with neuronal calcium sensor proteins in the retina in a cation-dependent manner
Abstract
Purpose: CERKL encodes for a ceramide kinase (CERK)-like protein. CERKL mutations are associated with severe retinal degeneration. Several studies have been conducted to prove a biochemical similarity between CERK and CERKL enzymatic activities. However, so far there has been no evidence that CERKL phosphorylates ceramide or any other lipid substrate in vitro or in vivo. The purpose of this work was to characterize CERKL's function by identification of CERKL-interacting proteins in the mammalian retina.
Methods: CERKL-interacting proteins were identified implementing the Ras-recruitment system (RRS) on a bovine retina cDNA library. Co-immunoprecipitation (co-IP) in transfected cells and in photoreceptor outer segments was used to verify the identified interactions. Serial deletion constructs were used to map the interacting sites. CERKL's kinase activity was tested by a CERK activity assay.
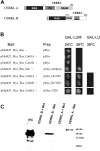
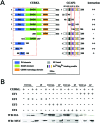
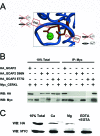
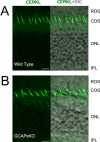
Results: We identified an interaction between CERKL and several neuronal calcium sensor (NCS) proteins, including guanylate cyclase activating protein 1 (GCAP1), GCAP2, and recoverin. These interactions were confirmed by co-IP experiments in transfected mammalian cells. Moreover, the interaction between endogenous CERKL and GCAP2 was confirmed by co-IP in photoreceptor outer segments. We found that CERKL-GCAP interaction is cation dependent and is mediated by CERKL's N-terminal region and by GCAPs cation-binding domains (EF-hands 2-4).
Conclusions: This study, which is the first to describe the interactions of CERKL with other retinal proteins, links CERKL to proteins involved in the photoresponse and Ca(2+) signaling, providing important clues for future research required in this direction.
Conflict of interest statement
Disclosure:
Figures


Similar articles
-
Wild-type levels of ceramide and ceramide-1-phosphate in the retina of ceramide kinase-like-deficient mice.Biochem Biophys Res Commun. 2008 Aug 15;373(1):159-63. doi: 10.1016/j.bbrc.2008.06.002. Epub 2008 Jun 12. Biochem Biophys Res Commun. 2008. PMID: 18555012
-
Regulatory function of the C-terminal segment of guanylate cyclase-activating protein 2.Biochim Biophys Acta. 2015 Oct;1854(10 Pt A):1325-37. doi: 10.1016/j.bbapap.2015.05.005. Epub 2015 May 19. Biochim Biophys Acta. 2015. PMID: 26001899
-
Expression and localization of CERKL in the mammalian retina, its response to light-stress, and relationship with NeuroD1 gene.Exp Eye Res. 2013 Jan;106:24-33. doi: 10.1016/j.exer.2012.10.014. Epub 2012 Nov 8. Exp Eye Res. 2013. PMID: 23142158 Free PMC article.
-
Molecular tuning of calcium dependent processes by neuronal calcium sensor proteins in the retina.Biochim Biophys Acta Mol Cell Res. 2023 Aug;1870(6):119491. doi: 10.1016/j.bbamcr.2023.119491. Epub 2023 May 23. Biochim Biophys Acta Mol Cell Res. 2023. PMID: 37230154 Review.
-
Dimerization of Neuronal Calcium Sensor Proteins.Front Mol Neurosci. 2018 Nov 2;11:397. doi: 10.3389/fnmol.2018.00397. eCollection 2018. Front Mol Neurosci. 2018. PMID: 30450035 Free PMC article. Review.
Cited by
-
CERKL knockdown causes retinal degeneration in zebrafish.PLoS One. 2013 May 9;8(5):e64048. doi: 10.1371/journal.pone.0064048. Print 2013. PLoS One. 2013. PMID: 23671706 Free PMC article.
-
Hyperautofluorescent Dots are Characteristic in Ceramide Kinase Like-associated Retinal Degeneration.Sci Rep. 2019 Jan 29;9(1):876. doi: 10.1038/s41598-018-37578-4. Sci Rep. 2019. PMID: 30696906 Free PMC article.
-
CERKL, a Retinal Dystrophy Gene, Regulates Mitochondrial Transport and Dynamics in Hippocampal Neurons.Int J Mol Sci. 2022 Sep 30;23(19):11593. doi: 10.3390/ijms231911593. Int J Mol Sci. 2022. PMID: 36232896 Free PMC article.
-
Sphingolipids as Emerging Mediators in Retina Degeneration.Front Cell Neurosci. 2019 Jun 11;13:246. doi: 10.3389/fncel.2019.00246. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31244608 Free PMC article. Review.
-
CERKL, a retinal disease gene, encodes an mRNA-binding protein that localizes in compact and untranslated mRNPs associated with microtubules.PLoS One. 2014 Feb 3;9(2):e87898. doi: 10.1371/journal.pone.0087898. eCollection 2014. PLoS One. 2014. PMID: 24498393 Free PMC article.
References
-
- Aleman TS, Soumittra N, Cideciyan AV, et al. CERKL mutations cause an autosomal recessive cone-rod dystrophy with inner retinopathy. Invest Ophthalmol Vis Sci. 2009;50:5944–5954 - PubMed
-
- Auslender N, Sharon D, Abbasi AH, Garzozi HJ, Banin E, Ben-Yosef T. A common founder mutation of CERKL underlies autosomal recessive retinal degeneration with early macular involvement among Yemenite Jews. Invest Ophthalmol Vis Sci. 2007;48:5431–5438 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

