Modern age pathology of pulmonary arterial hypertension
- PMID: 22679007
- PMCID: PMC3886716
- DOI: 10.1164/rccm.201201-0164OC
Modern age pathology of pulmonary arterial hypertension
Abstract
Rationale: The impact of modern treatments of pulmonary arterial hypertension (PAH) on pulmonary vascular pathology remains unknown.
Objectives: To assess the spectrum of pulmonary vascular remodeling in the modern era of PAH medication.
Methods: Assessment of pulmonary vascular remodeling and inflammation in 62 PAH and 28 control explanted lungs systematically sampled.
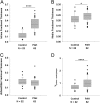
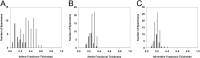
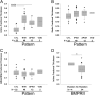
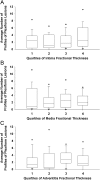
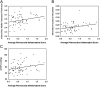
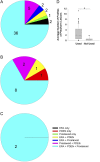
Measurements and main results: Intima and intima plus media fractional thicknesses of pulmonary arteries were increased in the PAH group versus the control lungs and correlated with pulmonary hemodynamic measurements. Despite a high variability of morphological measurements within a given PAH lung and among all PAH lungs, distinct pathological subphenotypes were detected in cohorts of PAH lungs. These included a subset of lungs lacking intima or, most prominently, media remodeling, which had similar numbers of profiles of plexiform lesions as those in lungs with more pronounced remodeling. Marked perivascular inflammation was present in a high number of PAH lungs and correlated with intima plus media remodeling. The number of profiles of plexiform lesions was significantly lower in lungs of male patients and those never treated with prostacyclin or its analogs.
Conclusions: Our results indicate that multiple features of pulmonary vascular remodeling are present in patients treated with modern PAH therapies. Perivascular inflammation may have an important role in the processes of vascular remodeling, all of which may ultimately lead to increased pulmonary artery pressure. Moreover, our study provides a framework to interpret and design translational studies in PAH.
Figures







Comment in
-
Progress in pulmonary arterial hypertension pathology: relighting a torch inside the tunnel.Am J Respir Crit Care Med. 2012 Aug 1;186(3):210-2. doi: 10.1164/rccm.201206-1049ED. Am J Respir Crit Care Med. 2012. PMID: 22855540 No abstract available.
References
-
- Humbert M, Sitbon O, Simonneau G. Treatment of pulmonary arterial hypertension. N Engl J Med 2004;351:1425–1436 - PubMed
-
- D’Alonzo GE, Barst RJ, Ayres SM, Bergofsky EH, Brundage BH, Detre KM, Fishman AP, Goldring RM, Groves BM, Kernis JT, et al. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann Intern Med 1991;115:343–349 - PubMed
-
- Heath D, Edwards JE. The pathology of hypertensive pulmonary vascular disease; a description of six grades of structural changes in the pulmonary arteries with special reference to congenital cardiac septal defects. Circulation 1958;18:533–547 - PubMed
-
- Wagenvoort CA, Wagenvoort N. Primary pulmonary hypertension: a pathologic study of the lung vessels in 156 clinically diagnosed cases. Circulation 1970;42:1163–1184
-
- Rabinovitch M, Haworth SG, Castaneda AR, Nadas AS, Reid LM. Lung biopsy in congenital heart disease: a morphometric approach to pulmonary vascular disease. Circulation 1978;58:1107–1122 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

