Role of reactive metabolites in the circulation in extrahepatic toxicity
- PMID: 22681489
- PMCID: PMC3423547
- DOI: 10.1517/17425255.2012.695347
Role of reactive metabolites in the circulation in extrahepatic toxicity
Abstract
Introduction: Reactive metabolite-mediated toxicity is frequently limited to the organ where the electrophilic metabolites are generated. Some reactive metabolites, however, might have the ability to translocate from their site of formation. This suggests that for these reactive metabolites, investigations into the role of organs other than the one directly affected could be relevant to understanding the mechanism of toxicity.
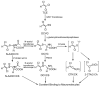
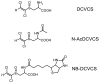
Areas covered: The authors discuss the physiological and biochemical factors that can enable reactive metabolites to cause toxicity in an organ distal from the site of generation. Furthermore, the authors present a case study which describes studies that demonstrate that S-(1,2-dichlorovinyl)-L-cysteine sulfoxide (DCVCS) and N-acetyl-S-(1,2-dichlorovinyl-L-cysteine sulfoxide (N-AcDCVCS), reactive metabolites of the known trichloroethylene metabolites S-(1,2-dichlorovinyl)-L-cysteine (DCVC), and N-acetyl-S-(1,2-dichlorovinyl)-L-cysteine (N-AcDCVC), are generated in the liver and translocate through the circulation to the kidney to cause nephrotoxicity.
Expert opinion: The ability of reactive metabolites to translocate could be important to consider when investigating mechanisms of toxicity. A mechanistic approach, similar to the one described for DCVCS and N-AcDCVCS, could be useful in determining the role of circulating reactive metabolites in extrahepatic toxicity of drugs and other chemicals. If this is the case, intervention strategies that would not otherwise be feasible might be effective for reducing extrahepatic toxicity.
Figures
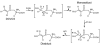

References
-
- Einarson TR. Drug-related hospital admissions. Ann Pharmacother. 1993;27:832–40. - PubMed
-
- Lazarou J, Pomeranz BH, Corey PN. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA. 1998;279:1200–5. - PubMed
-
- Lasser KE, Allen PD, Woolhandler SJ, et al. Timing of new black box warnings and withdrawals for prescription medications. JAMA. 2002;287:2215–20. - PubMed
-
- Brock N, Hohorst HJ. Metabolism of cyclophosphamide. Cancer. 1967;20:900–4. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
