β-Catenin regulates hepatic mitochondrial function and energy balance in mice
- PMID: 22684045
- PMCID: PMC12045480
- DOI: 10.1053/j.gastro.2012.05.048
β-Catenin regulates hepatic mitochondrial function and energy balance in mice
Abstract
Background & aims: Wnt signaling regulates hepatic function and nutrient homeostasis. However, little is known about the roles of β-catenin in cellular respiration or mitochondria of hepatocytes.
Methods: We investigated β-catenin's role in the metabolic function of hepatocytes under homeostatic conditions and in response to metabolic stress using mice with hepatocyte-specific deletion of β-catenin and their wild-type littermates, given either saline (sham) or ethanol (as a model of binge drinking and acute ethanol intoxication).
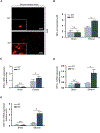
Results: Under homeostatic conditions, β-catenin-deficient hepatocytes demonstrated mitochondrial dysfunctions that included impairments to the tricarboxylic acid cycle and oxidative phosphorylation (OXPHOS) and decreased production of adenosine triphosphate (ATP). There was no evidence for redox imbalance or oxidative cellular injury in the absence of metabolic stress. In mice with β-catenin-deficient hepatocytes, ethanol intoxication led to significant redox imbalance in the hepatocytes and further deterioration in mitochondrial function that included reduced OXPHOS, fatty acid oxidation (FAO), and ATP production. Ethanol feeding significantly increased liver steatosis and oxidative damage, compared with wild-type mice, and disrupted the ratio of nicotinamide adenine dinucleotide. β-catenin-deficient hepatocytes also had showed disrupted signaling of Sirt1/peroxisome proliferator-activated receptor-α signaling.
Conclusions: β-catenin has an important role in the maintenance of mitochondrial homeostasis, regulating ATP production via the tricarboxylic acid cycle, OXPHOS, and fatty acid oxidation; β-catenin function in these systems is compromised under conditions of nutrient oxidative stress. Reagents that alter Wnt-β-catenin signaling might be developed as a useful new therapeutic strategy for treatment of liver disease.
Copyright © 2012 AGA Institute. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Disclosures:
The authors declare no conflict of interest.
Figures




References
-
- Rodgers JT, Lerin C, Haas W, Gygi SP, Spiegelman BM, Puigserver P. Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature 2005;434:113–8. - PubMed
-
- Calvisi DF, Factor VM, Ladu S, Conner EA, Thorgeirsson SS. Disruption of beta-catenin pathway or genomic instability define two distinct categories of liver cancer in transgenic mice. Gastroenterology 2004;126:1374–86. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

