Electron transfer-based combination therapy of cisplatin with tetramethyl-p-phenylenediamine for ovarian, cervical, and lung cancers
- PMID: 22685209
- PMCID: PMC3387118
- DOI: 10.1073/pnas.1203451109
Electron transfer-based combination therapy of cisplatin with tetramethyl-p-phenylenediamine for ovarian, cervical, and lung cancers
Abstract
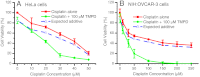
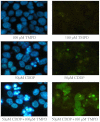
The platinum-based chemotherapy is the standard treatment for several types of cancer. However, cancer cells often become refractory with time and most patients with serious cancers die of drug resistance. Recently, we have discovered a unique dissociative electron-transfer mechanism of action of cisplatin, the first and most widely used platinum-based anticancer drug. Here, we show that the combination of cisplatin with an exemplary biological electron donor, N,N,N',N'-tetramethyl-p-phenylenediamine (TMPD), may overcome the resistance of cancer cells to cisplatin. Our steady-state absorption and fluorescence spectroscopic measurements confirm the effective dissociative electron-transfer reaction between TMPD and cisplatin. More significantly, we found that the combination of 100 μM TMPD with cisplatin enhances double-strand breaks of plasmid DNA by a factor of approximately 3.5 and dramatically reduces the viability of cisplatin-sensitive human cervical (HeLa) cancer cells and highly cisplatin-resistant human ovarian (NIH:OVCAR-3) and lung (A549) cancer cells. Furthermore, this combination enhances apoptosis and DNA fragmentation by factors of 2-5 compared with cisplatin alone. These results demonstrate that this combination treatment not only results in a strong synergetic effect, but also makes resistant cancer cells sensitive to cisplatin. Because cisplatin is the cornerstone agent for the treatment of a variety of human cancers (including testicular, ovarian, cervical, bladder, head/neck, and lung cancers), our results show both the potential to improve platinum-based chemotherapy of various human cancers and the promise of femtomedicine as an emerging frontier in advancing cancer therapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
New combination chemotherapy of cisplatin with an electron-donating compound for treatment of multiple cancers.Sci Rep. 2021 Jan 12;11(1):788. doi: 10.1038/s41598-020-80876-z. Sci Rep. 2021. PMID: 33436996 Free PMC article.
-
Cisplatin inhibits paclitaxel-induced apoptosis in cisplatin-resistant ovarian cancer cell lines: possible explanation for failure of combination therapy.Cancer Res. 1999 May 15;59(10):2425-32. Cancer Res. 1999. PMID: 10344753
-
Cisplatin resistance in human cervical, ovarian and lung cancer cells.Cancer Chemother Pharmacol. 2015 Jun;75(6):1217-27. doi: 10.1007/s00280-015-2739-2. Epub 2015 Apr 18. Cancer Chemother Pharmacol. 2015. PMID: 25894720
-
Cisplatin in Ovarian Cancer Treatment-Known Limitations in Therapy Force New Solutions.Int J Mol Sci. 2023 Apr 20;24(8):7585. doi: 10.3390/ijms24087585. Int J Mol Sci. 2023. PMID: 37108749 Free PMC article. Review.
-
Recent updates in the clinical use of platinum compounds for the treatment of gynecologic cancers.Semin Oncol. 2004 Dec;31(6 Suppl 14):17-24. doi: 10.1053/j.seminoncol.2004.11.007. Semin Oncol. 2004. PMID: 15726530 Review.
Cited by
-
miR34a/GOLPH3 Axis abrogates Urothelial Bladder Cancer Chemoresistance via Reduced Cancer Stemness.Theranostics. 2017 Oct 17;7(19):4777-4790. doi: 10.7150/thno.21713. eCollection 2017. Theranostics. 2017. PMID: 29187903 Free PMC article.
-
Preclinical Evaluation of 1,2-Diamino-4,5-Dibromobenzene in Genetically Engineered Mouse Models of Pancreatic Cancer.Cells. 2019 Jun 9;8(6):563. doi: 10.3390/cells8060563. Cells. 2019. PMID: 31181844 Free PMC article.
-
Antioxidant Defenses: A Context-Specific Vulnerability of Cancer Cells.Cancers (Basel). 2019 Aug 20;11(8):1208. doi: 10.3390/cancers11081208. Cancers (Basel). 2019. PMID: 31434226 Free PMC article. Review.
-
Neuroprotective activities of curcumin and quercetin with potential relevance to mitochondrial dysfunction induced by oxaliplatin.Protoplasma. 2016 Mar;253(2):417-30. doi: 10.1007/s00709-015-0821-6. Epub 2015 May 29. Protoplasma. 2016. PMID: 26022087
-
Dual targeting of heat shock proteins 90 and 70 promotes cell death and enhances the anticancer effect of chemotherapeutic agents in bladder cancer.Oncol Rep. 2014 Jun;31(6):2482-92. doi: 10.3892/or.2014.3132. Epub 2014 Apr 9. Oncol Rep. 2014. PMID: 24718854 Free PMC article.
References
-
- Kavarnos GJ, Turro NJ. Photosensitization by reversible electron-transfer-theories, experimental-evidence, and examples. Chem Rev. 1986;86:401–449.
-
- Marcus RA. Electron transfer reactions in chemistry: Theory and experiment (Nobel Lecture) Rev Mod Phys. 1993;65:599–610.
-
- Gray HB, Winkler JR. Electron transfer in proteins. Ann Rev Biochem. 1996;65:537–561. - PubMed
-
- Lu QB. Cosmic-ray-driven electron-induced reactions of halogenated molecules adsorbed on ice surfaces: Implications for atmospheric ozone depletion and global climate change. Phys Rep. 2010;487:141–167.

