Adipose tissue microRNAs as regulators of CCL2 production in human obesity
- PMID: 22688341
- PMCID: PMC3402332
- DOI: 10.2337/db11-1508
Adipose tissue microRNAs as regulators of CCL2 production in human obesity
Abstract
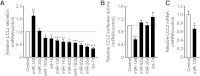
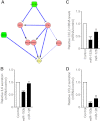
In obesity, white adipose tissue (WAT) inflammation is linked to insulin resistance. Increased adipocyte chemokine (C-C motif) ligand 2 (CCL2) secretion may initiate adipose inflammation by attracting the migration of inflammatory cells into the tissue. Using an unbiased approach, we identified adipose microRNAs (miRNAs) that are dysregulated in human obesity and assessed their possible role in controlling CCL2 production. In subcutaneous WAT obtained from 56 subjects, 11 miRNAs were present in all subjects and downregulated in obesity. Of these, 10 affected adipocyte CCL2 secretion in vitro and for 2 miRNAs (miR-126 and miR-193b), regulatory circuits were defined. While miR-126 bound directly to the 3'-untranslated region of CCL2 mRNA, miR-193b regulated CCL2 production indirectly through a network of transcription factors, many of which have been identified in other inflammatory conditions. In addition, overexpression of miR-193b and miR-126 in a human monocyte/macrophage cell line attenuated CCL2 production. The levels of the two miRNAs in subcutaneous WAT were significantly associated with CCL2 secretion (miR-193b) and expression of integrin, α-X, an inflammatory macrophage marker (miR-193b and miR-126). Taken together, our data suggest that miRNAs may be important regulators of adipose inflammation through their effects on CCL2 release from human adipocytes and macrophages.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

