PAK4 kinase activity and somatic mutation promote carcinoma cell motility and influence inhibitor sensitivity
- PMID: 22689056
- PMCID: PMC3446866
- DOI: 10.1038/onc.2012.233
PAK4 kinase activity and somatic mutation promote carcinoma cell motility and influence inhibitor sensitivity
Abstract
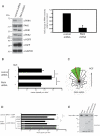
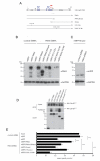
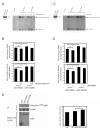
Hepatocyte growth factor (HGF) and its receptor (c-Met) are associated with cancer cell motility and invasiveness. p21-activated kinase 4 (PAK4), a potential therapeutic target, is recruited to and activated by c-Met. In response, PAK4 phosphorylates LIM kinase 1 (LIMK1) in an HGF-dependent manner in metastatic prostate carcinoma cells. PAK4 overexpression is known to induce increased cell migration speed but the requirement for kinase activity has not been established. We have used a panel of PAK4 truncations and mutations in a combination of overexpression and RNAi rescue experiments to determine the requirement for PAK4 kinase activity during carcinoma cell motility downstream of HGF. We find that neither the kinase domain alone nor a PAK4 mutant unable to bind Cdc42 is able to fully rescue cell motility in a PAK4-deficient background. Nevertheless, we find that PAK4 kinase activity and associated LIMK1 activity are essential for carcinoma cell motility, highlighting PAK4 as a potential anti-metastatic therapeutic target. We also show here that overexpression of PAK4 harbouring a somatic mutation, E329K, increased the HGF-driven motility of metastatic prostate carcinoma cells. E329 lies within the glycine-rich loop region of the kinase. Our data suggest that E329K mutation leads to a modest increase in kinase activity, conferring resistance to competitive ATP inhibitors in addition to promoting cell migration. The existence of such a mutation may have implications for the development of PAK4-specific competitive ATP inhibitors should PAK4 be further explored for clinical inhibition.
Figures

Similar articles
-
A PAK4-LIMK1 pathway drives prostate cancer cell migration downstream of HGF.Cell Signal. 2008 Jul;20(7):1320-8. doi: 10.1016/j.cellsig.2008.02.021. Epub 2008 Mar 12. Cell Signal. 2008. PMID: 18424072
-
Overexpression of P21-activated kinase 4 is associated with poor prognosis in non-small cell lung cancer and promotes migration and invasion.J Exp Clin Cancer Res. 2015 May 15;34(1):48. doi: 10.1186/s13046-015-0165-2. J Exp Clin Cancer Res. 2015. PMID: 25975262 Free PMC article.
-
Pak4, a novel Gab1 binding partner, modulates cell migration and invasion by the Met receptor.Mol Cell Biol. 2009 Jun;29(11):3018-32. doi: 10.1128/MCB.01286-08. Epub 2009 Mar 16. Mol Cell Biol. 2009. PMID: 19289496 Free PMC article.
-
PAK4: a pluripotent kinase that regulates prostate cancer cell adhesion.J Cell Sci. 2010 May 15;123(Pt 10):1663-73. doi: 10.1242/jcs.055707. Epub 2010 Apr 20. J Cell Sci. 2010. PMID: 20406887 Free PMC article.
-
Drug discovery targeting p21-activated kinase 4 (PAK4): a patent review.Expert Opin Ther Pat. 2021 Nov;31(11):977-987. doi: 10.1080/13543776.2021.1944100. Epub 2021 Aug 9. Expert Opin Ther Pat. 2021. PMID: 34369844 Review.
Cited by
-
Mutational drivers of cancer cell migration and invasion.Br J Cancer. 2021 Jan;124(1):102-114. doi: 10.1038/s41416-020-01149-0. Epub 2020 Nov 18. Br J Cancer. 2021. PMID: 33204027 Free PMC article. Review.
-
MicroRNA-Based Prophylaxis in a Mouse Model of Cirrhosis and Liver Cancer.Mol Ther Nucleic Acids. 2019 Mar 1;14:239-250. doi: 10.1016/j.omtn.2018.11.018. Epub 2018 Dec 6. Mol Ther Nucleic Acids. 2019. PMID: 30641476 Free PMC article.
-
Fisetin Modulates Human Oral Squamous Cell Carcinoma Proliferation by Blocking PAK4 Signaling Pathways.Drug Des Devel Ther. 2020 Feb 25;14:773-782. doi: 10.2147/DDDT.S229270. eCollection 2020. Drug Des Devel Ther. 2020. PMID: 32158195 Free PMC article.
-
Structure, biochemistry, and biology of PAK kinases.Gene. 2017 Mar 20;605:20-31. doi: 10.1016/j.gene.2016.12.014. Epub 2016 Dec 19. Gene. 2017. PMID: 28007610 Free PMC article. Review.
-
Potassium channel activity controls breast cancer metastasis by affecting β-catenin signaling.Cell Death Dis. 2019 Feb 21;10(3):180. doi: 10.1038/s41419-019-1429-0. Cell Death Dis. 2019. PMID: 30792401 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

